This website uses cookies to ensure you get the best experience on our website.
- Table of Contents
Facts about Myeloid cell nuclear differentiation antigen.
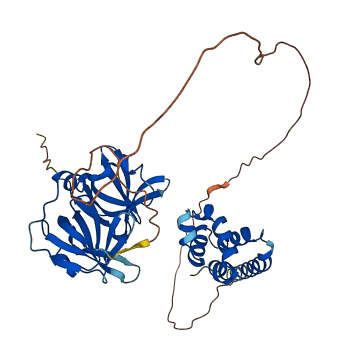
Stimulates the DNA binding of the transcriptional repressor protein YY1. .
| Human | |
|---|---|
| Gene Name: | MNDA |
| Uniprot: | P41218 |
| Entrez: | 4332 |

| Belongs to: |
|---|
| No superfamily |

myeloid cell nuclear differentiation antigen; PYHIN3
Mass (kDA):
45.836 kDA

| Human | |
|---|---|
| Location: | 1q23.1 |
| Sequence: | 1; NC_000001.11 (158831351..158849502) |
Expressed constitutively in cells of the myeloid lineage. Found in promyelocyte stage cells as well as in all other stage cells including peripheral blood monocytes and granulocytes. Also appear in myeloblast cells in some cases of acute myeloid Leukemia.
Nucleus. Cytoplasm. Uniformly distributed throughout the interphase cell nucleus. Associates with chromatin.





PMID: 1644857 by Briggs J.A., et al. Cloning and expression of the human myeloid cell nuclear differentiation antigen: regulation by interferon alpha.
PMID: 1377701 by Burrus G.R., et al. Characterization of the human myeloid cell nuclear differentiation antigen: relationship to interferon-inducible proteins.