This website uses cookies to ensure you get the best experience on our website.
- Table of Contents
Discover the highest affinity/specificity Clones
With our patented microfluidics plasma cell discovery (PCD) platform, you can get fully validated, affinity matured rabbit monoclonal antibodies:
The scale of phage display meets the power of in vivo affinity maturation--antibody discovery has never been so robust.
Standard workflows can miss the clones that matter most.
|
Recommended
Boster Bio PCD Rabbit Monoclonal
|
Hybridoma Rabbit Monoclonal
|
Mouse Monoclonal
|
Rabbit Polyclonal
|
Phage Display
|
|
| Affinity | +++ High | ++ Hit-or-miss | + Lower | ++ Hit-or-miss | ++ Can be higher |
|---|---|---|---|---|---|
| CDR Pool Size | +++ Large | +++ Large | + Small | +++ Large | +++ Large |
| Cost | +++ High | +++ High | ++ Mid | + Very low | +++ High |
| Scalability | +++ High | +++ High | +++ High | + Low | +++ High |
| Turnaround | + Slow | + Slow | + Slow | +++ Fast | + Slow |
| Best Fit | Diagnostics & Therapeutics | Diagnostics & Therapeutics | Diagnostics & Research | Research | Diagnostics & Therapeutics |
Built for antibody discovery programs where target difficulty, epitope sensitivity, or downstream development requirements call for a more selective rabbit monoclonal workflow.
Best-fit use cases
Common target classes
Boster also highlights therapeutic target classes such as ion channels, GPCRs, and other difficult biomarker targets as part of its rabbit monoclonal development positioning.
Boster’s plasma cell discovery (PCD) platform is designed to expand rabbit monoclonal screening earlier in the workflow, helping uncover rare clones for difficult and specificity-critical targets. The current service positions this approach for diagnostic and therapeutic antibody development.
Instead of limiting discovery to a narrow pool of candidates, the PCD workflow uses flow cytometry to isolate antigen-specific plasma cells from millions of splenocytes, followed by secondary screening and recombinant expression. Boster also states that this approach increases the number of screenable clones by 500-fold.
Screening reach
Millions
Clone expansion
500-fold
Best aligned with
Dx + Tx
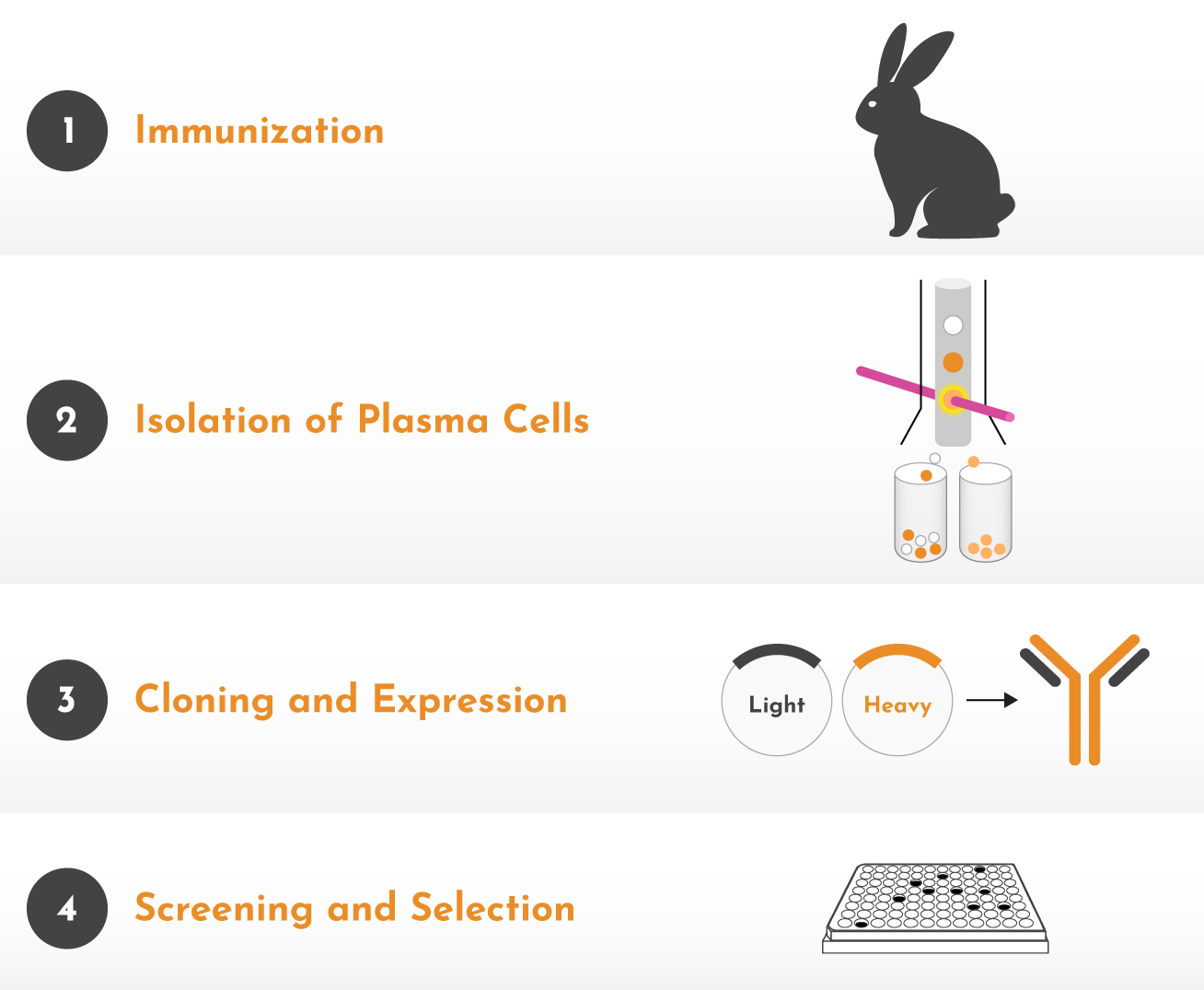
A simplified view of Boster’s rabbit monoclonal workflow: immunization, plasma cell isolation, cloning and expression, followed by screening and selection.
See the Service in 1 Minute or Dive Deeper
Start with a quick PCD overview, then move into the full rabbit monoclonal presentation or slide deck for a deeper look at workflow, target scope, and delivery logic.
Ready to discuss your target and project scope?
From immunization through clone expression, Boster’s workflow supports structured rabbit monoclonal development for difficult and specificity-critical targets.
A typical rabbit monoclonal development project runs about 4–6 months, covering immunization, FACS-based plasma cell screening, secondary ELISA screening, clone selection, and recombinant expression.
Typical total project time
4–6 months
Phase 1
Peptide Preparation, Conjugation, and Immunization
Timeline
2–3 months
Deliverable: Titer data
Phase 2
FACS Screening and Secondary ELISA Screening
First Screening with FACS (> 1 million B cells)
Timeline
2 weeks
Supernatant Production and Secondary ELISA
Supernatant production at 0.25 mL scale, followed by secondary screening with ELISA.
Timeline
1 week
Deliverable: ELISA data
Phase 3
Clone Review, Deconvolution, and Recombinant Expression
Customer further screening with supernatant if necessary, followed by selection of the three best clones.
Timeline
Project-dependent
Clone Deconvolution and 30 mL-Scale Production
Clone deconvolution, cloning, expression, ELISA confirmation, and 30 mL-scale production with about 0.5 mg antibody yield per clone.
Timeline
3–4 weeks
Deliverable: Sequences
Upon successful completion of the development phases, Boster provides a practical delivery package for downstream review, testing, and next-step project decisions.
Screening data
ELISA data
Delivered from both sera and supernatant assays.
Sequence information
Final clone sequences
Sequence information is included for the final delivered clones.
Vector map
Expression vector map
The final clone package includes the expression vector map.
Purified antibody
~0.5 mg Ab
About 0.5 mg purified antibody is typically delivered, with the original page noting a range of 0.1–1 mg.
Plasmid DNA
100 ng DNA
About 100 ng of heavy and light chain plasmid DNA is delivered for the final chosen clone.
Summary report
Final project report
A final summary report documents the delivered package and project outputs.
Boster’s rabbit monoclonal service can also extend beyond initial clone discovery, supporting downstream development needs such as engineering, characterization, validation, and larger-scale production.
Affinity maturation
For projects that need improved binding performance or further antibody optimization.
Humanization
For non-human antibodies moving toward translational or therapeutic development paths.
Epitope mapping
For deeper target characterization, clone differentiation, and downstream decision-making.
Scale-up production
For projects that require larger production volumes beyond the standard delivery package.
Serum-free production
For workflows that need a more advanced recombinant production format.
Custom application validation
For customers who need additional validation in assay-specific workflows.
Boster can also support downstream steps such as affinity maturation, humanization, epitope mapping, validation, and scale-up planning.
Discuss Next-Step OptionsCyclophosphamide (Cyc) is a small-molecule anti-cancer drug and immunosuppressant. In this example, Boster used its PCD workflow to develop rabbit monoclonal antibodies for cyclophosphamide monitoring and compared the resulting clones with a commercially available mouse monoclonal standard.
Screened
2M splenocytes
Isolated
94 plasma cells
ELISA-positive
61 clones (93.8%)
Study design
After 3 booster shots, rabbit splenocytes were screened with PCD technology. Antigen-specific plasma cells were isolated, heavy and light chain genes were cloned, and 4 randomly selected rabbit anti-Cyc clones were compared with a commercially available mouse hybridoma line.
Key result
All 4 rabbit anti-Cyc clones showed stronger affinity than the commercial mouse hybridoma standard.
This result highlights the value of rabbit monoclonals for small-molecule and specificity-sensitive targets.
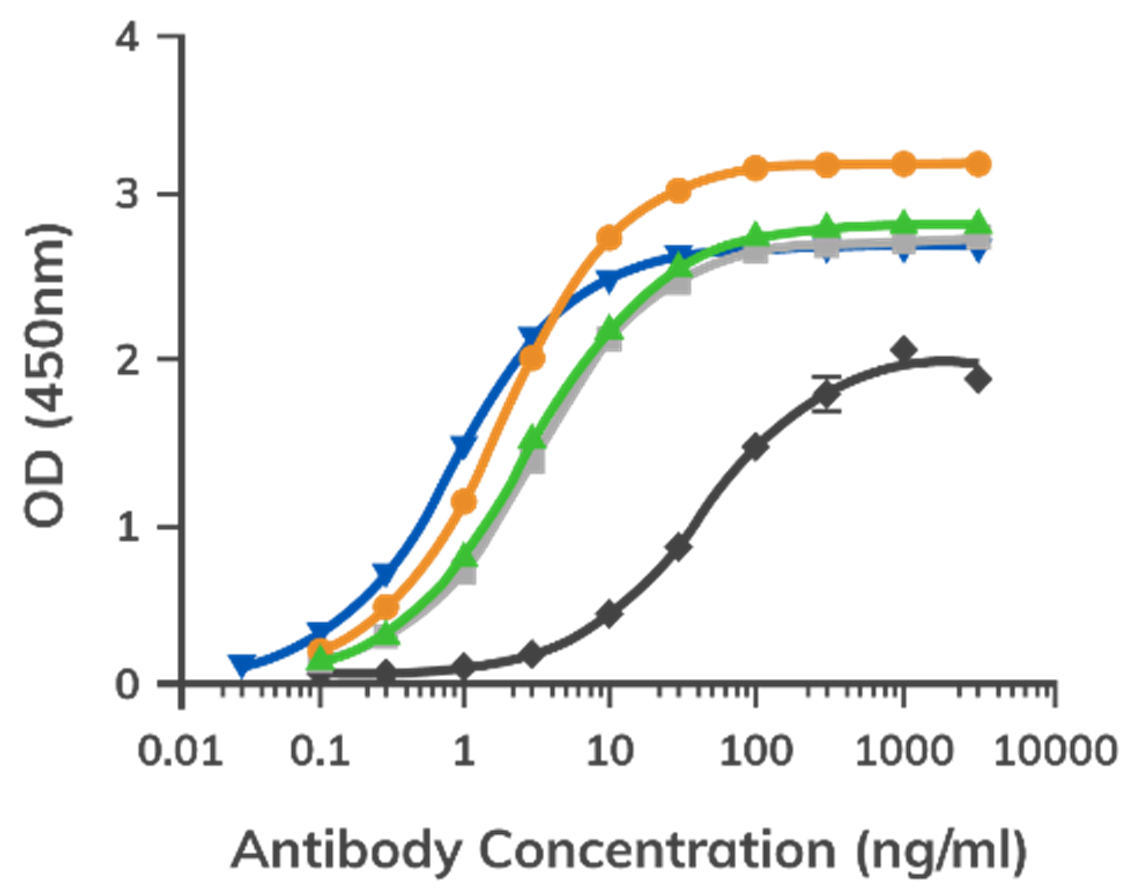
Figure. EC-50 curves of 4 randomly selected rabbit anti-cyclophosphamide monoclonal antibody-producing clones compared with a positive control antibody from a commercially available mouse hybridoma line.
Key questions about project scope, timeline, deliverables, scale-up, and post-delivery handling.
Before starting a project
After clone delivery
Tell us your target, intended application, and development goals. Our team can review project fit and recommend the next step.
Begin Inquiry1000+
PCD Projects
Making Antibodies
since 1993
70,000+
publications
2000+
RabMabs Available