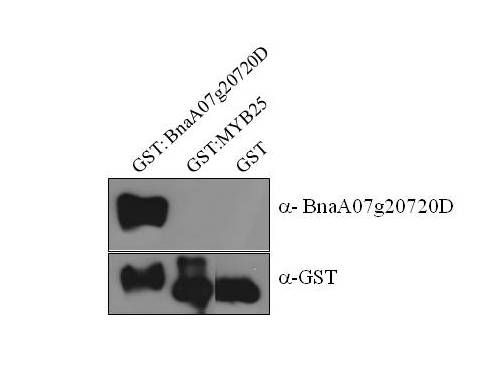
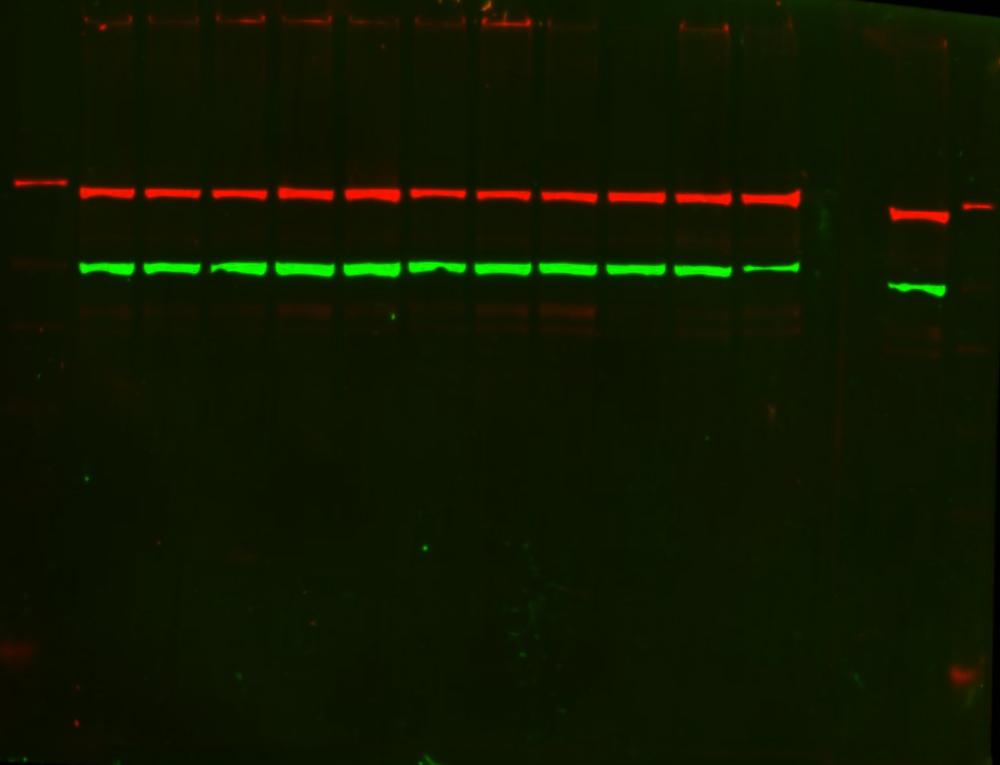
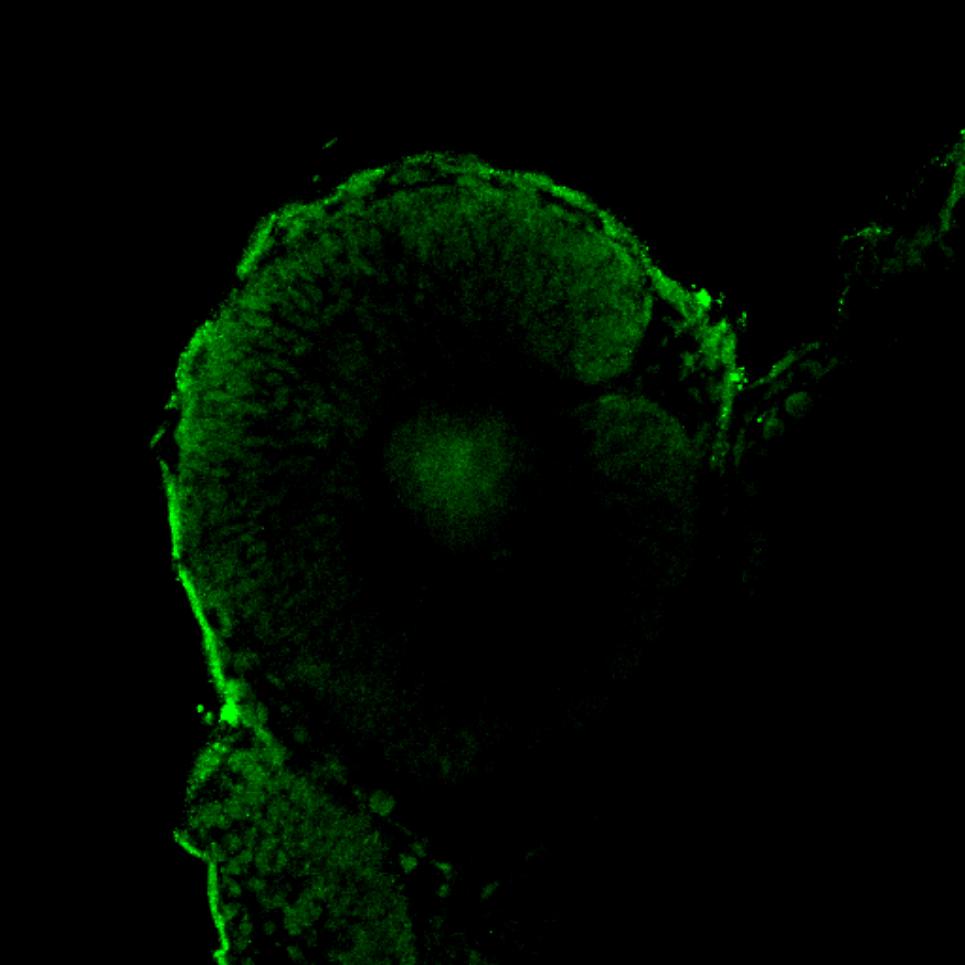
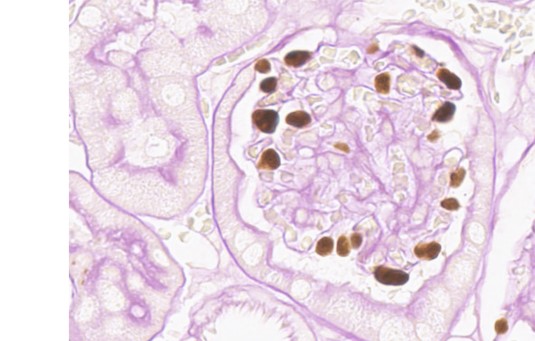
How are primary antibodies made?
At their origins, all primary antibodies are made from building blocks from
immunesystems of the hosts. These building blocks are called complementarity-determining regions
(CDRs). CDR is a complex topic which we will not go into in this article so for more information on
CDRs you can visit this Wikipedia page.
Traditionally, scientists made polyclonal primary antibodies by immunizing the host
animals, typically rabbit, goat, chicken, mice and variety of mammalian and avian species, with
antigens, inciting the immune responses which produce antibodies, and extracting (by a method called
immune-affinity purification chromatography) the resulting antibodies from the host’s sera or eggs.
In 1975, Georges Köhler and César Milstein developed the monoclonal antibody
technology. In 1985, George P Smith developed the method called phage-display, which marks
the start of the antibody engineering era where antibody development moves from hijacking hosts’
adaptive immune systems towards rational design.
However the majority of antibodies used today, in the 2020s, are still made from host
animals at some point in its production. A truly animal free primary antibody development solution
has yet to be invented, primarily because scientists have not found an animal-free mechanism that
can replace the somatic maturation of B-cells. Without such mechanism, it is impossible to achieve
the same level of affinity and specificity of naturally matured antibodies. This opinion is from the
author of this article and is up for debate, if you believe otherwise you can send your arguments to
[email
protected] or post on our Facebook page.



.png)