This website uses cookies to ensure you get the best experience on our website.
- Table of Contents
Need your gene of interest packaged into AAV? Our AAV production service delivers research-grade recombinant AAV (rAAV) across common serotypes with documented quality control, including qPCR titer, sterility testing, and capsid purity by SDS-PAGE. Typical titers reach up to ~1×10¹³ vg/mL (serotype and cargo dependent). Request a quote to match serotype, purification method, and scale to your study.
Understand the advantages that make adeno-associated virus a powerful, low-risk choice for research and therapeutic applications.
AAV vectors are derived from a naturally occurring virus that causes no known disease in humans. This inherent safety profile reduces experimental risk and translates well to therapeutic development, giving you confidence in both in vitro and in vivo studies.
AAV triggers minimal immune responses compared to other viral vectors, enabling longer transgene expression in animal models and reducing confounding variables in your experiments. This characteristic is especially valuable for chronic gene therapy studies.
Multiple AAV serotypes provide natural tropism for specific tissues including CNS, muscle, liver, and retina. This diversity lets you select the right vector for your target cell type without extensive optimization, saving time and experimental resources.
Gene Synthesis
Sequence-verified
Clone Gene into Vector
Plasmid map + Sanger confirmation
AAV Production + Purification
HEK293 triple transfection AAV packaging protocol
Quality Control
qPCR titer + SDS-PAGE + sterility (+ optional endotoxin)
As easy as 1, 2, 3.
Important: Tropism varies by species, route (IV/IT/IM/subretinal), dose, and promoter; use tables as starting points.
| Serotype | Tissue Tropism |
|---|---|
| AAV1 | Smooth muscle, skeletal muscle, CNS, brain, lung, retina, inner ear, pancreas, heart, liver |
| AAV2 | Smooth muscle, CNS, brain, liver, pancreas, kidney, retina, inner ear, testes |
| AAV2.7m8 | Retina, inner ear |
| AAV2-retro | Spinal nerves |
| AAV3 | Smooth muscle, liver, lung |
| AAV4 | CNS, retina, lung, kidney, heart |
| AAV5 | Smooth muscle, CNS, brain, lung, retina, heart, immune system |
| AAV6 | Smooth muscle, heart, lung, pancreas, adipose, liver, immune system |
| AAV7 | Smooth muscle, retina, CNS, brain, liver |
| AAV8 | Smooth muscle, CNS, brain, retina, inner ear, liver, pancreas, heart, kidney, adipose |
| AAV9 | Smooth muscle, skeletal muscle, lung, liver, heart, pancreas, CNS, retina, inner ear, testes, kidney, adipose |
| AAVShH10 | Retina |
| AAVrh10 | Retina, liver, brain |
| AAV-DJ | Liver, heart, kidney, spleen, retina |
| AAV-PHP.eB | CNS |
| AAV10 | Kidney, Uterus, heart, liver, lung, skeletal muscle |
| AAV11 | Kidney, spleen, stomach, heart, lung skeletal muscle |
| Tissue Type | Recommended AAV Serotype |
|---|---|
| Smooth Muscle | AAV1, AAV2, AAV3, AAV5, AAV6, AAV7, AAV8, AAV9 |
| Skeletal Muscle | AAV1, AAV9, AAV10, AAV11 |
| CNS | AAV1, AAV2, AAV4, AAV5, AAV7, AAV8, AAV9, AAV-PHP.eB |
| Brain | AAV1, AAV2, AAV5, AAV7, AAV8 |
| Retina | AAV1, AAV2, AAV2.7m8, AAV4, AAV5, AAV7, AAV8, AAV9, AAVShH10, AAVrh10, AAV-DJ |
| Inner Ear | AAV1, AAV2, AAV6.2, AAV8, AAV9, AAV2.7m8 |
| Lung | AAV1, AAV3, AAV4, AAV5, AAV6, AAV6.2, AAV9, AAV10, AAV11 |
| Liver | AAV1, AAV2, AAV3, AAV6, AAV6.2, AAV7, AAV8, AAV9, AAVrh10, AAV-DJ, AAV10 |
| Pancreas | AAV1, AAV2, AAV6, AAV8, AAV9 |
| Heart | AAV1, AAV4, AAV5, AAV6, AAV8, AAV9, AAV-DJ, AAV10, AAV11 |
| Kidney | AAV2, AAV4, AAV8, AAV9, AAV-DJ, AAV10, AAV11 |
| Adipose | AAV6, AAV8, AAV9 |
| Testes | AAV2, AAV9 |
| Spleen | AAV-DJ, AAV11 |
| Spinal Nerves | AAV2-retro |
We may be able to source additional serotypes upon request. Please contact us for more information.
Contact UsKnow the size constraints and design principles for efficient packaging.
AAV can package about 4.7 kilobases of single-stranded DNA between the two ITR sequences. Constructs exceeding this limit will package inefficiently or not at all.
Your total insert size, including promoter, transgene, regulatory elements, and polyadenylation signal, must fit within ~4.7 kb. For reference, a CMV promoter is about 0.6 kb, a standard polyA is 0.2 kb, leaving approximately 3.9 kb for your gene of interest and any additional elements.
Design Rule: The ITR sequences are NOT counted toward the 4.7 kb limit. Plan your construct elements accordingly and contact us if your design approaches the size maximum.
Maximum Cargo: 4.7 kb
Typical Components:
Need help optimizing your construct size? Our team can suggest compact promoters and regulatory elements.
From gene therapy to CRISPR editing, see how AAV vectors support diverse experimental workflows.
Deliver functional copies of genes to correct genetic deficiencies in disease models. AAV's long-term expression makes it ideal for evaluating therapeutic efficacy in monogenic disorders affecting liver, muscle, CNS, and retinal tissues.
Package Cas9, base editors, or prime editors alongside sgRNAs for targeted genome modification. AAV's episomal nature reduces off-target integration risk compared to lentiviral delivery, providing cleaner editing outcomes for validation studies.
Express channelrhodopsins, calcium indicators, or fluorescent reporters in specific cell populations using serotype-specific tropism. AAV enables stable, long-term expression for chronic imaging and behavior studies without repeated injections.
Deliver shRNA or miRNA expression cassettes for sustained gene knockdown. AAV-mediated RNAi provides months-long suppression in vivo, offering an alternative to CRISPR knockout when reversibility or dose titration is needed.
Map neural circuits using retrograde AAV serotypes and fluorescent proteins. Combine with Cre-dependent constructs for intersectional genetic targeting in complex brain region studies.
Transduce T cells with chimeric antigen receptors for adoptive cell therapy research. AAV's lower integration rate compared to lentivirus may reduce insertional mutagenesis risk in certain CAR-T protocols.
Start small with research-grade AAV, then scale seamlessly to preclinical and GMP production as your program advances.
1-5 mL at 1x1012 GC/mL
Ideal for in vitro validation, construct screening, and small animal pilot studies. Fast turnaround for iterative design optimization.
Typical use: Cell culture transduction, 5-10 mice
10-50 mL at 1x1013 GC/mL
Sufficient for multi-dose efficacy studies, biodistribution analysis, and early toxicity assessment in rodents or NHP.
Typical use: Dose-response studies, 50-100 animals
100+ mL, CDMO partnerships
Boster will not handle GMP in-house but will transition seamlessly to a GMP and AAV CDMO partner. We provide tech transfer support and process validation documentation.
Typical use: IND-enabling studies, clinical trials
Complete your gene delivery workflow with complementary viral vector services designed to work together.
If your research-grade AAV shows promise, you'll eventually need GMP manufacturing for clinical development.
Our preclinical AAV protocols are designed with GMP compatibility in mind. We document processes, maintain batch records, and can provide tech transfer packages to reduce friction during scale-up.
Ask about our CDMO partnerships and CMC consulting services.
Clear deliverables, timelines, and pricing structure so you can plan your project budget and schedule.
| Service Tier | Scale | Titer | Purity | Turnaround | Typical Applications |
|---|---|---|---|---|---|
| Research Grade | 1-5 mL | ≥1x1012 GC/mL | Standard purification | 3-4 weeks | In vitro studies, construct screening, small pilot experiments |
| Preclinical Grade | 10-50 mL | ≥1x1013 GC/mL | Ultracentrifugation + ion exchange | 4-6 weeks | Animal efficacy studies, biodistribution, proof-of-concept in vivo |
| High Purity | 10-50 mL | ≥1x1013 GC/mL | >95% by SDS-PAGE, empty capsid removal | 5-7 weeks | Toxicity studies, IND-enabling research, demanding applications |
| Custom Scale | 100+ mL | Custom | Custom purification protocol | 8-12 weeks | Large animal studies, manufacturing process development |
Not sure which serotype? We offer serotype screening panels to test 6-8 variants in your target tissue.
High
efficiency
High Purity
high Potency
Guaranteed
titer
Proprietary
technologies
Serving Science
since 1993
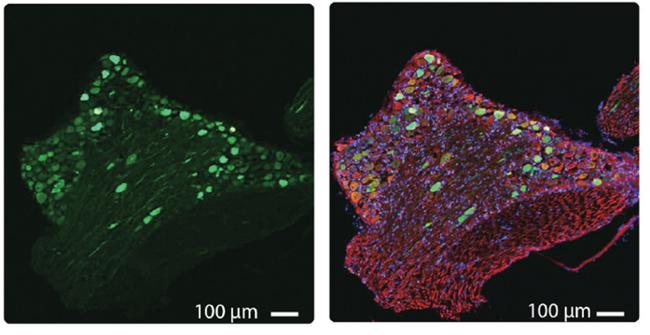
Left: EGFP expression (Green) in lumbar neuronal cells 4 weeks after intrathecal injection of AAV-EGFP Serotype 9 (Cat. # iAAV01509) into mice.
Right: Overlay with β-tubulin (red) and DAPI (blue).
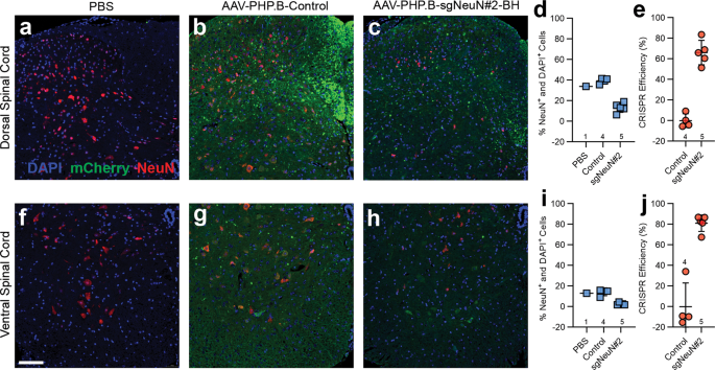
Immunohistochemical staining reveals robust NeuN gene disruption in the spinal cord.
Speak with our technical team about your construct design, timeline requirements, and application needs. We provide free design reviews and detailed quotes within 24 hours.
services@bosterbio.com
(888)466-3604
Still got questions about our AAV packaging services?
Q1. What AAV Volumes Do You Offer?
Q2. What Is Your Expected Turnaround Time?
Q3. How Do I Choose The Right AAV Packaging Service For My Needs?
Q4. What Experiments Can I Perform With AAVs?
Q6. What disadvantages are there in using AAV?
Q7. Is high yield or high purity of more importance?
In general, the titer is of more importance because it is a measurement of the number of viral particles per volume.
Purity will be a more important consideration for in vivo studies. If the virus is not pure, it could cause undesired responses in experimental animals. Our high titer AAVs are suitable for in vivo studies.
For more information on why AAV titer is important click here.
Q8. Is the small cellular immune response going to be an issue?
Q9. Storage, ordering and shipping
Our storage solution for AAVs produced via triple transfection is PBS with 5% Glycerol, which is suitable for in vivo injection. AAV stability at 4°C allows for short-term use and room temperature for immediate use. For long-term storage, AAV should be stored at -80°C for best quality and longevity. Although AAV is a very stable virus, multiple freeze-thaw cycles may reduce the titer of the product.
Shipments are typically shipped in dry ice overnight, however alternative shipping methods are available at an additional cost. Please get in contact to discuss your requirements.
Q10. What sequences do you not accept for AAV packaging?