This website uses cookies to ensure you get the best experience on our website.
- Table of Contents
1 Citations
Facts about NPC intracellular cholesterol transporter 1.
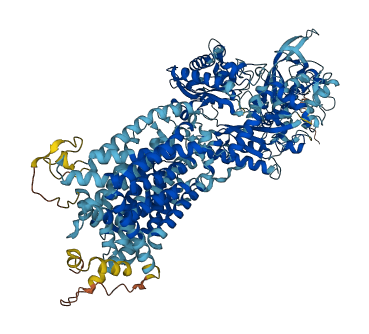
Cholesterol binds to NPC1 with the hydroxyl group buried in the binding pocket (PubMed:19563754). Binds oxysterol with greater affinity than cholesterol.
| Human | |
|---|---|
| Gene Name: | NPC1 |
| Uniprot: | O15118 |
| Entrez: | 4864 |

| Belongs to: |
|---|
| patched family |

FLJ98532; Niemann-Pick C1 protein; Niemann-Pick disease, type C1; Niemann-Pick Type C1; NPC; NPC1; SLC65A1
Mass (kDA):
142.167 kDA

| Human | |
|---|---|
| Location: | 18q11.2 |
| Sequence: | 18; NC_000018.10 (23506184..23586506, complement) |
Late endosome membrane; Multi-pass membrane protein. Lysosome membrane; Multi-pass membrane protein.






PMID: 9211849 by Carstea E.D., et al. Niemann-Pick C1 disease gene: homology to mediators of cholesterol homeostasis.
PMID: 10425213 by Morris J.A., et al. The genomic organization and polymorphism analysis of the human Niemann-Pick C1 gene.