This website uses cookies to ensure you get the best experience on our website.
- Table of Contents
Every flow cytometry (or FACS) experiment begins with sample preparation. Check out our flow cytometry sample preparation guide to learn how to prepare your samples for flow cytometric analysis.
Proper sample preparation is critical for avoiding instrument clogs, reducing background noise, and preserving cell viability. Whether working with cultured cells, cryopreserved samples, or primary cells from tissues, the goal is to produce a homogeneous single-cell suspension at the correct concentration in a suitable staining buffer. High-quality preparations improve staining efficiency, reduce variability, and provide reliable results for downstream analysis.
The following protocols provide a general framework for preparing samples for flow cytometry. While these steps are widely applicable, individual investigators should adapt and optimise them to suit their specific cell type, experimental goals, and staining requirements.
Cells for flow cytometry analysis are usually derived from four main sources:
Regardless of the source, the final cell preparation should be:
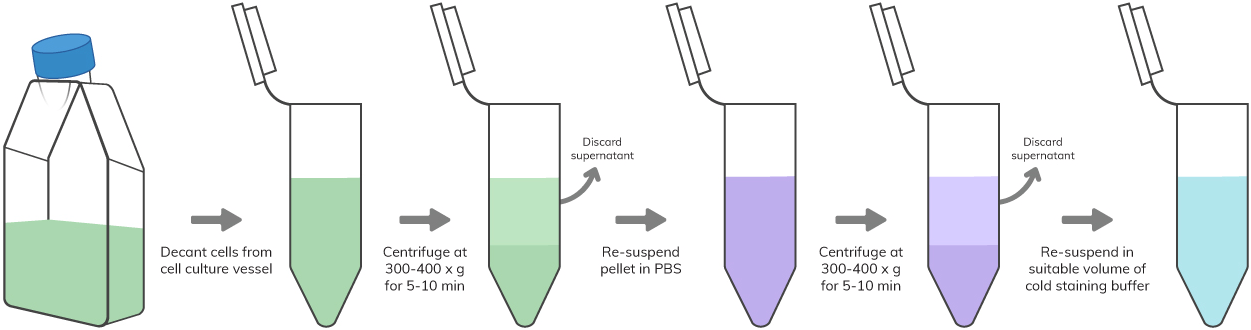
a homogenous single-cell suspension free of clumps and dead cell debris at a density of 106-107 cell per ml suspended in a suitable staining buffer.
PBMCs isolated from whole blood through Ficoll gradient centrifugation or RBC lysed whole blood or non-adherent cultured cells are readily available for flow cytometric analysis. Adherent cultured cells or cells present in the solid organs should be first made into a single cell suspension before flow analysis by using enzymatic digestion or mechanical dissociation of the tissue, respectively. Mechanical filtration should be followed to avoid unwanted instrument clogs or lower quality flow data. Use the following sample preparation protocols based on your appropriate starting materials.
Key Reagents – PBS, staining buffer
Key Reagents – PBS, staining buffer, 0.25% trypsin
Key reagents – PBS, staining buffer, culture medium with 10% FBS
Key reagents – PBS, staining buffers, suitable gradients medium like Ficoll or Histopaque
Key reagents – PBS, staining buffer (see appendix), RBC lysis buffer [check Appendix for recipe or use a commercially available buffer]
This guide will show you all the nuts and bolts for Flow Cytometry and FACS, including expert review of principle, optimized protocol that really works, and more.
Improve flow cytometry sample quality with practical guidance on cell handling, viability, and preparation methods before analysis.
Identify common flow cytometry issues such as high background, weak signal, and poor population separation with practical troubleshooting tips.
Learn the fundamental principle of flow cytometry and FACS, including how cells are counted, profiled, and sorted in a fluid mixture.
Review step-by-step flow cytometry protocols for routine workflow setup, experiment planning, and day-to-day lab reference.
Explore optimization tips for panel setup, staining conditions, and signal quality to improve overall flow cytometry performance.
Browse flow cytometry resources in one place, including technical guides, sample preparation help, and supporting educational content.
Access the downloadable guide covering flow cytometry and FACS basics, optimized protocols, and troubleshooting support in one place.
Optimize fixation and permeabilization methods for intracellular staining to improve consistency and preserve target detection.
Learn how cell-free antibody synthesis works and explore its potential advantages for rapid antibody generation and screening workflows.