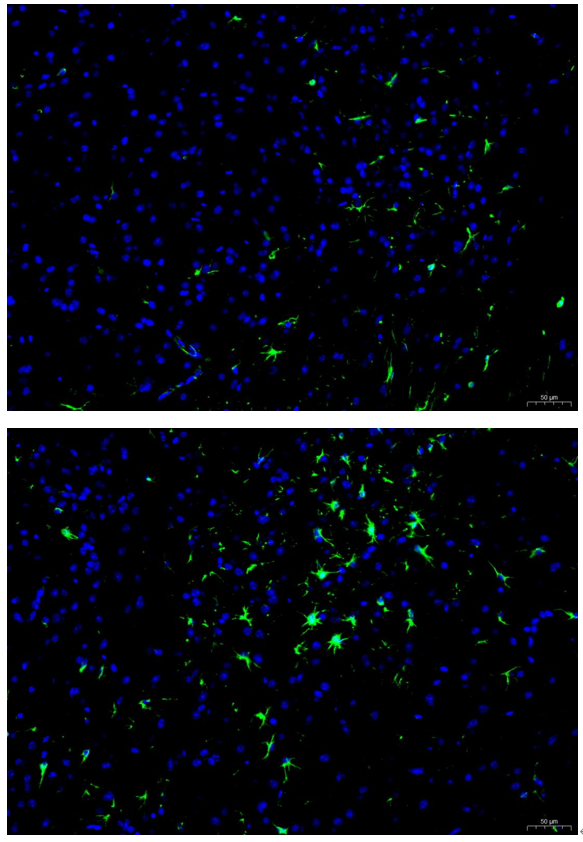
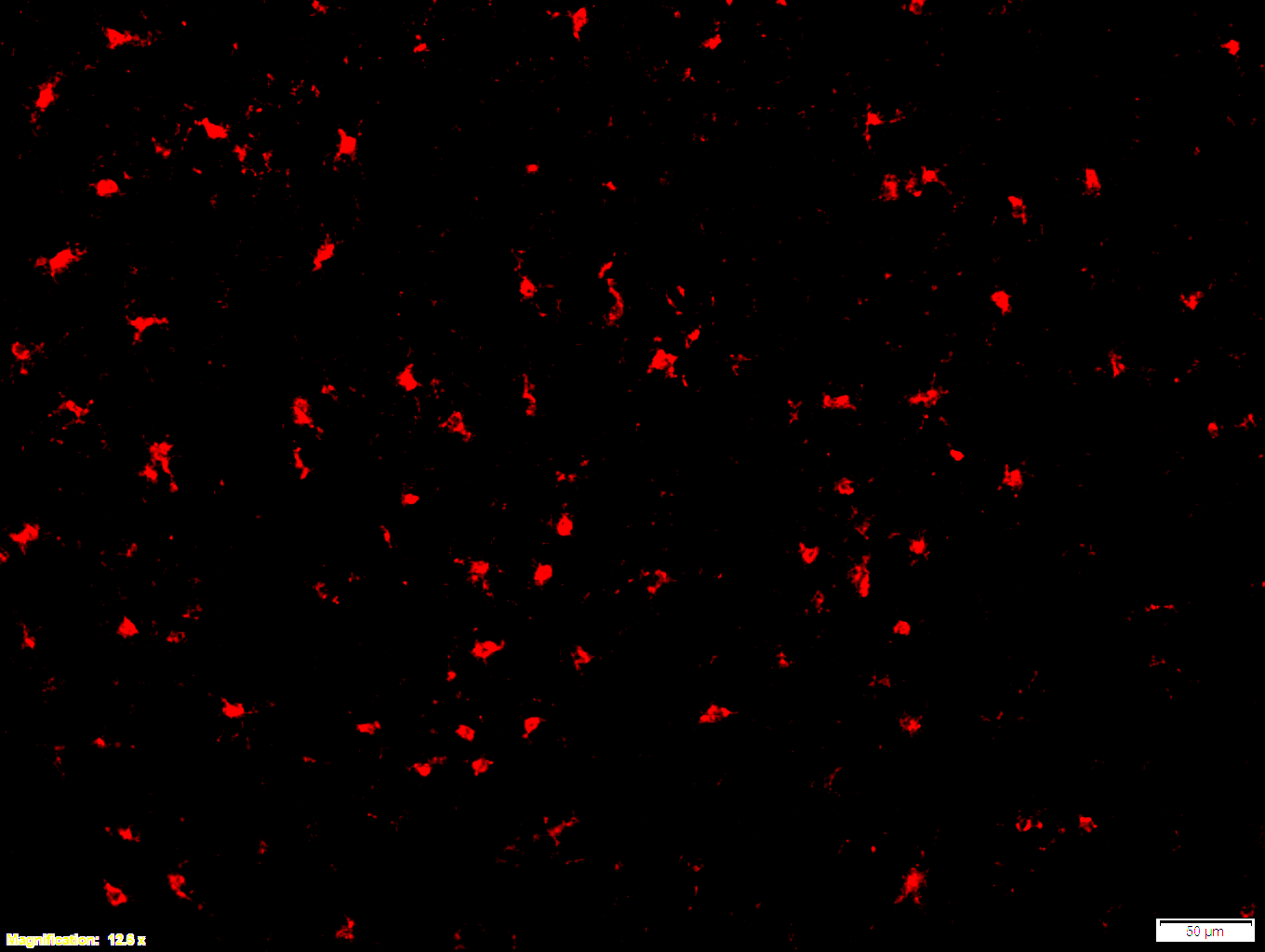
Boster bio Iba1 1:100 PFA (3-4) - cortex 12x - with scale bar

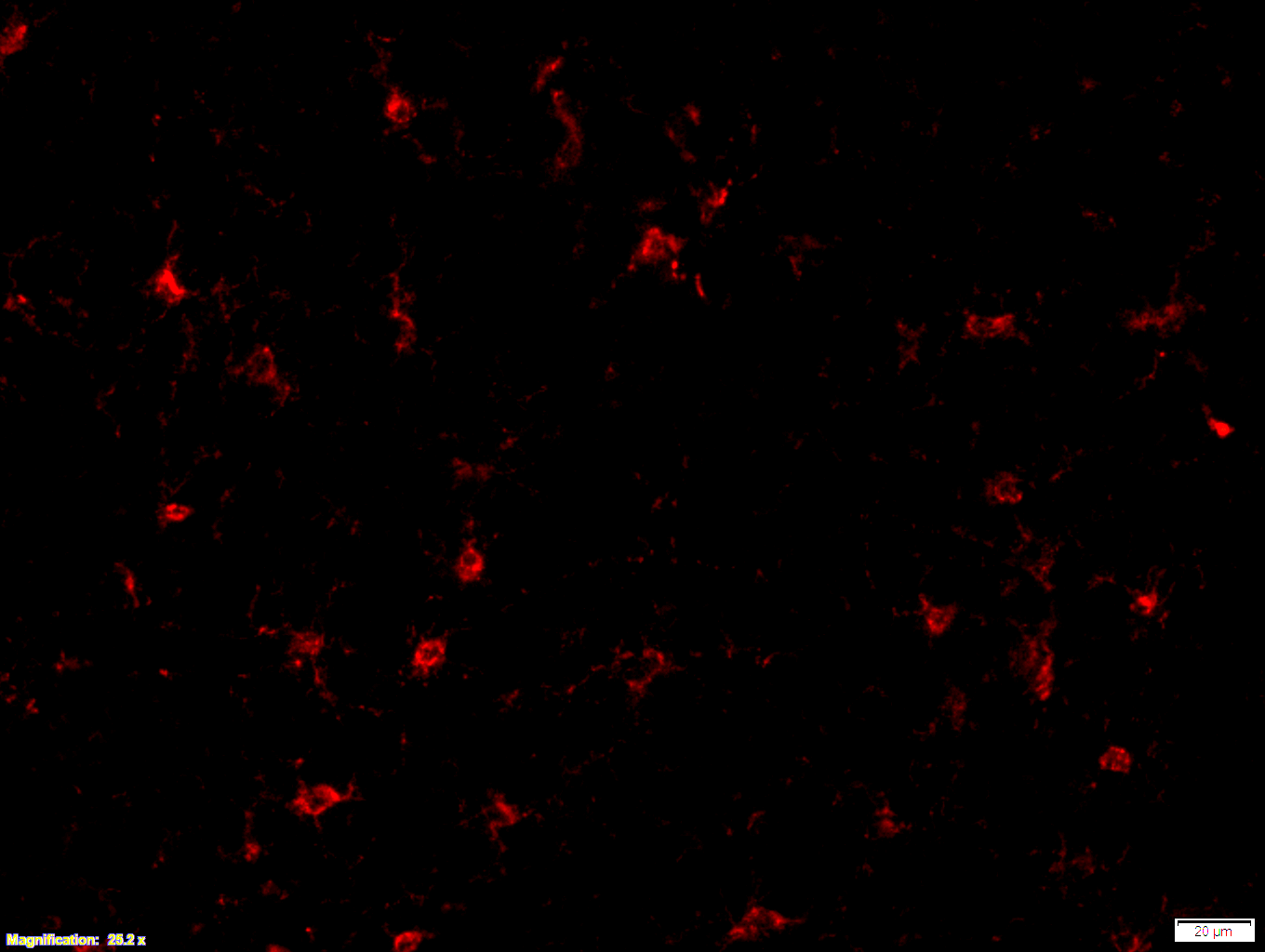
Boster bio Iba1 1:100 PFA (3-4) - cortex 25x - with scale bar
| SKU | A01394 |
|---|
| Application | Immunofluorescence |
|---|
| Sample | Mouse brain |
|---|
| Primary Antibody Dilution | 1:100 |
|---|
Images that were made from fresh frozen cryostat sections, were thaw-mounted onto microscope slides. Mounted sections were later post-fixed with 300 µL of 4% paraformaldehyde (PFA) in PBS at room temperature for 10 minutes. After fixation and prior to incubation with antibodies, slides were washed 3 times with 300 µL of 1X PBS with 0.01% sodium azide for 3 minutes per rinse, followed by permeabilization with 300 µL of 0.3% Triton X-100 in 1X PBS with 0.01% sodium azide for 30 minutes at room temperature, then blocking with 700 µL of 4% donkey serum diluted in 1X PBS with 0.01% sodium azide + Triton X-100 (blocking buffer) for 30 minutes at room temperature. Slides were incubated with 325 µL of Iba1 (1:100). Concentrations of 1:100 were achieved by diluting 10 µL of antibody in 1,000 µL of blocking buffer; concentrations of 1:250 were achieved by diluting 4 µL of antibody in 1,000 µL of blocking buffer. Sections were incubated overnight at 4 ˚C. On the next day, slides were washed 3 times with 300 µL of 1X PBS with 0.01% sodium azide for 3 minutes per rinse, then incubated with the secondary antibodies at room temperature, in the dark, for 1 hour. Washing step was repeated then slides were left in the dark to dry. Mounting media was added to cover slip the sections. Slides were kept in the dark at 4 ˚C prior to imaging.