This website uses cookies to ensure you get the best experience on our website.
- Table of Contents
2 Citations 6 Q&As
Facts about Serpin B5.
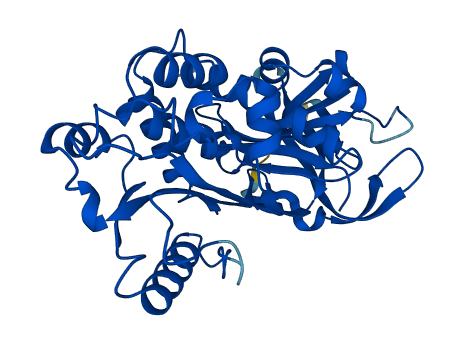
As it does not undergo the S (stressed) to R (relaxed) conformational transition characteristic of active serpins, it exhibits no serine protease inhibitory activity. .
| Human | |
|---|---|
| Gene Name: | SERPINB5 |
| Uniprot: | P36952 |
| Entrez: | 5268 |

| Belongs to: |
|---|
| serpin family |

Maspin; Peptidase inhibitor 5; PI5; PI-5; PI5protease inhibitor 5 (maspin); serine (or cysteine) proteinase inhibitor, clade B (ovalbumin), member 5; Serpin B5; serpin peptidase inhibitor, clade B (ovalbumin), member 5
Mass (kDA):
42.1 kDA

| Human | |
|---|---|
| Location: | 18q21.33 |
| Sequence: | 18; NC_000018.10 (63476958..63505085) |
Normal mammary epithelial cells.
Secreted, extracellular space.






PMID: 8290962 by Zou Z., et al. Maspin, a serpin with tumor-suppressing activity in human mammary epithelial cells.
PMID: 7797587 by Pemberton P.A., et al. The tumor suppressor maspin does not undergo the stressed to relaxed transition or inhibit trypsin-like serine proteases. Evidence that maspin is not a protease inhibitory serpin.
*More publications can be found for each product on its corresponding product page