This website uses cookies to ensure you get the best experience on our website.
- Table of Contents
Facts about Spondin-2.
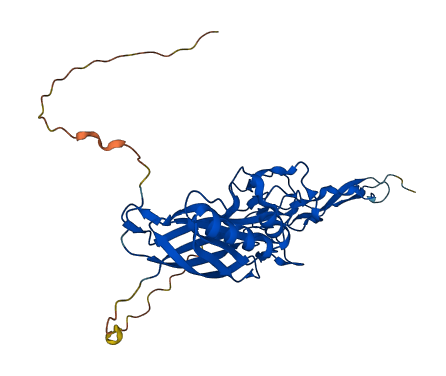
Critical in the initiation of the innate immune response and represents a special pattern- recognition molecule in the ECM for microbial pathogens (By similarity). Binds bacterial lipopolysaccharide (LPS).
| Human | |
|---|---|
| Gene Name: | SPON2 |
| Uniprot: | Q9BUD6 |
| Entrez: | 10417 |

| Belongs to: |
|---|
| No superfamily |

Differentially expressed in cancerous and non-cancerous lung cells 1; DIL1; DIL-1; DIL1FLJ34460; DKFZp686G21139; FLJ16313; FLJ22401; Mindin; M-SPONDIN; RG-1; SPON2; Spondin 2; spondin 2, extracellular matrix protein; spondin-2
Mass (kDA):
35.846 kDA

| Human | |
|---|---|
| Location: | 4p16.3 |
| Sequence: | 4; NC_000004.12 (1166932..1208962, complement) |
Secreted, extracellular space, extracellular matrix.





PMID: 10512675 by Manda R., et al. Identification of genes (SPON2 and C20orf2) differentially expressed between cancerous and noncancerous lung cells by mRNA differential display.
PMID: 19153605 by Li Y., et al. Structure of the F-spondin domain of mindin, an integrin ligand and pattern recognition molecule.