Product Info Summary
| SKU: | EK0327 |
|---|---|
| Size: | 96 wells/kit, with removable strips. |
| Reactive Species: | Human |
| Application: | ELISA |
| Sample Types: | cell culture supernatants, serum, plasma (heparin, EDTA) and human milk. |
Product info
Product Name
Human soluble EGFR ELISA Kit PicoKine®
SKU/Catalog Number
EK0327
Size
96 wells/kit, with removable strips.
*Question: How many samples can I assay/run in this kit?
Description
Human soluble EGFR ELISA Kit PicoKine® (96 Tests). Quantitate Human EGFR in cell culture supernatants, serum, plasma (heparin, EDTA) and human milk. Sensitivity: 1pg/ml. The brand Picokine indicates this is a premium quality ELISA kit. Each Picokine kit delivers precise quantification, high sensitivity, and excellent reproducibility. Only our most reliable and effective kits qualify as Picokine, guaranteeing top-tier results for your assays.
Storage & Handling
Store at 4°C for 6 months, at -20°C for 12 months. Avoid multiple freeze-thaw cycles (Ships with gel ice, can store for up to 3 days in room temperature. Freeze upon receiving.)
Cite This Product
Human soluble EGFR ELISA Kit PicoKine® (Boster Biological Technology, Pleasanton CA, USA, Catalog # EK0327)
Clonality of Antibodies
See Datasheet for details
Standard Protein
Expression system for standard: NS0; Immunogen sequence: M1-S645
Sensitivity
<1 pg/ml
Assay Range
156 pg/ml - 10,000 pg/ml
Standard Dilution Instructions

See datasheet of EK0327 for more details
Cross-reactivity
There is no detectable cross-reactivity with other relevant proteins.
Reactive Species
EK0327 is reactive to EGFR in Human samples
Validated Sample Types
cell culture supernatants, serum, plasma (heparin, EDTA) and human milk.
Application Guarantee
EK0327 is guaranteed for ELISA in Human by Boster Guarantee
See how Boster Bio validate our ELISA kits: ELISA Validation Information
Background of EGFR
The epidermal growth factor receptor (EGFR; ErbB-1; HER1 in humans) is the cell-surface receptor for members of the epidermal growth factor family (EGF-family) of extracellular protein ligands.1 It is a member of the ErbB family of receptors, a subfamily of four closely related receptor tyrosine kinases: EGFR (ErbB-1), HER2/c-neu (ErbB-2), Her 3 (ErbB-3) and Her 4 (ErbB-4). EGFR exists on the cell surface and is activated by binding of its specific ligands, including epidermal growth factor and transforming growth factor alpha (TGFalpha). EGFR and its ligands are cell signaling molecules involved in diverse cellular functions, including cell proliferation, differentiation, motility, and survival, and in tissue development. Mutations that lead to EGFR overexpression (known as upregulation) or overactivity have been associated with a number of cancers, including lung cancer and glioblastoma multiforme. In this latter case a more or less specific mutation of EGFR, called EGFRvIII is often observed.
Kit Components
| Catalog Number | Description | Quantity |
|---|---|---|
| EK0327-CAP | Anti-Human EGFR Pre-coated 96-well strip microplate | 1 |
| EK0327-ST | Human EGFR Standard | 2 vials, 10 ng/tube |
| EK0327-DA | Human EGFR Biotinylated antibody (100x) | 100ul |
| AR1103 | Avidin-Biotin-Peroxidase Complex (100x) | 100ul |
| AR1106-1 | Sample Diluent | 30ml |
| AR1106-2 | Antibody Diluent | 12ml |
| AR1106-3 | Avidin-Biotin-Peroxidase Diluent | 12ml |
| AR1104 | Color Developing Reagent (TMB) | 10ml |
| AR1105 | Stop Solution | 10ml |
| AR1106-5 | Wash Buffer (25x) | 20ml |
| PLA-SEA | Adhesive plate sealers | 4 |
*The kit components are not available for individual purchase.
Materials Required But Not Included In Kit
- Microplate Reader capable of reading absorbance at 450nm.
- Incubator.
- Automated plate washer (optional).
- Pipettes and pipette tips capable of precisely dispensing 0.5 µl through 1 ml volumes of aqueous solutions.
- Multichannel pipettes are recommended for large amount of samples.
- Deionized or distilled water.
- 500ml graduated cylinders.
- Test tubes for dilution.
Data Examples, Quality Control Data & Sample Dilution
Validation Standard Curve O.D. At 450nm
| Concentration (pg/ml) | 0 | 156 | 312 | 625 | 1250 | 2500 | 5000 | 10000 |
| O.D. | 0.033 | 0.099 | 0.149 | 0.246 | 0.431 | 0.735 | 1.26 | 1.91 |
Data Example Images
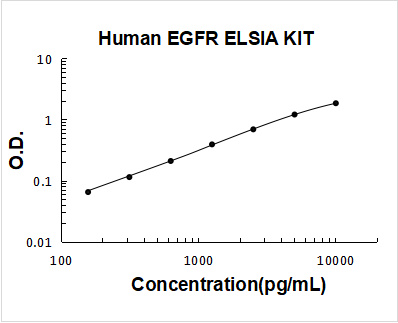
Click image to see more details
Human EGFR PicoKine ELISA Kit standard curve
Recommended Sample Dilution Ratios
According to our internal validation assays using this ELISA kit, to detect EGFR, Dilution ratio of 1:20, concentration in serum and plasma is 41-93 ng/ml..
Some articles we found to cite concentrations of EGFR in samples: 17679463 (Pubmed IDs).
Intra Assay Consistency & Inter Assay Consistency
We measured random samples of Human soluble EGFR ELISA Kit PicoKine® within the same batch/lot to ensure the consistency of the kits' performances. ELISA intra assay consistency is measured using wells from the same plate/assay kit. ELISA inter assay consistency is measured using wells from different plates from the same batch production/lot.
| Intra-Assay Precision | Inter-Assay Precision | |||||
|---|---|---|---|---|---|---|
| Sample | 1 | 2 | 3 | 1 | 2 | 3 |
| n | 16 | 16 | 16 | 24 | 24 | 24 |
| Mean (pg/ml) | 289 | 1634 | 5526 | 313 | 1731 | 5572 |
| Standard deviation | 13.58 | 81.7 | 408.92 | 18.78 | 102.12 | 417.9 |
| CV (%) | 4.7% | 5% | 7.4% | 6% | 5.9% | 7.5% |
Specific Publications For Human soluble EGFR ELISA Kit PicoKine® (EK0327)
Loading publications
Recommended Resources
Here are featured tools and databases that you might find useful.
- Boster's Pathways Library
- Protein Databases
- Bioscience Research Protocol Resources
- Data Processing & Analysis Software
- Photo Editing Software
- Scientific Literature Resources
- Research Paper Management Tools
- Molecular Biology Software
- Primer Design Tools
- Bioinformatics Tools
- Phylogenetic Tree Analysis
Customer Reviews
0 Reviews For Human soluble EGFR ELISA Kit PicoKine®
Customer Q&As
Have a question?
Find answers in Q&As, reviews.
Can't find your answer?
Submit your question
8 Customer Q&As for Human soluble EGFR ELISA Kit PicoKine®
Question
Q: can I use citrate plasma as samples in Human EGFR Picokine® ELISA Kit (Catalog # EK0327)?
Verified Customer
Verified customer
Asked: 2020-08-13
Answer
A: Chelating agents such as EDTA, Heparin and Citrate can attach metal ions from the functional domain of EGFR causing disruption of its protein structure. EGFR may be denatured as a result and may compromise the assay's measurements. The chilating sites could also be too close to the epitopes required for detection and block the antigen antibody reaction. We have tested the EGFR ELISA, treating samples with various anticoagulants and decided that heparin or EDTA can be used for treatment of blood/plasma samples. Do not use other anticoagulents when collecting samples.
Boster Scientific Support
Answered: 2020-08-13
Question
Q: what is the protocol regarding preparation of cell lysates prepared for use in Picokine® ELISA kits?
Y. Jones
Verified customer
Asked: 2020-07-15
Answer
A: in those Picokine® ELISAs where cell or tissue lysate is a validated sample type, sample preparation instructions for lysate are included in the product insert. Components in lysate and lysis buffer may affect immunoreactivity, so if lysate is not a validated sample type, care must be taken in sample preparation and validation.
Boster Scientific Support
Answered: 2020-07-15
Question
Q: Can EGFR ELISA Kits be used with tissue homogenates (or other non-validated sample types)?
L. Jha
Verified customer
Asked: 2020-07-01
Answer
A: Unfortunately, Boster Bio has not routinely validated tissue homogenates as a sample type for ELISA kits. This does not mean that ELISA kits are not valid for other sample types than we have tested: it means further investigation is required. One will need to perform a spike and recovery study to determine if an unvalidated sample type will work with a particular kit. To perform a spike and recovery experiment, one should divide a sample into two aliquots. In one of the aliquots, the user should spike in a known amount of the kit standard. a dilution series is performed comparing the spiked versus the unspiked sample. Generally, samples with expected recovery and linearity between 80-120% are considered acceptable. This method can be used to validate any sample type that has not been assessd by Boster Bio. for a more detailed spike and recovery protocol, please contact technical support.
Note: acceptable ranges should be determined individually by each laboratory. Additionally, technical support can help determine if a buffer component is not compatible with a given ELISA kit. please see the Citations tab on the product webpage for peer-reviewed papers utilizing a wide range of sample types. We also have an innovator's reward program where if the user validates our ELISA kits in applications or samples previously not validated by Boster Bio or other users, and share such information with us by submit a review, we will reward the user's efforts with a free antibody or ELISA kit from our catalog. Biocompare.com will also give $20 Amazon giftcard as an additional reward, if the review is submitted there as well.
Boster Scientific Support
Answered: 2020-07-01
Question
Q: how many samples can be assayed in a Picokine® ELISA Kit?
Verified Customer
Verified customer
Asked: 2019-11-29
Answer
A: The Picokine® ELISA Kits will generally run a 7-point standard curve, non-specific binding wells, and 39 samples in duplicate. this may depends upon the kit used so please refer to each datasheet for details.
Boster Scientific Support
Answered: 2019-11-29
Question
Do you have a non-cross reactivity validation data for the EK0327 kit with mouse EGFR? Does it react with mouse EGFR?
Verified customer
Asked: 2019-10-10
Answer
Our lab did not test the Human soluble EGFR ELISA Kit PicoKine EK0327 for the cross reactivity with mouse EGFR. Currently, we do not have the specific recombinant mouse EGFR protein for testing.
Boster Scientific Support
Answered: 2019-10-11
Question
Q: how can I thaw whole blood sample for EGFR ELISA after freezing?
Verified Customer
Verified customer
Asked: 2019-06-29
Answer
A: we do not recommend freezing and thaw whole blood. erythrocytes are fragile and, if frozen and thawed, will undergo hemolysis rendering the samples useless. To keep your blood samples to test EGFR for a later time, you should let the blood clot in glass tubes and collect the serum to freeze for later use.
Boster Scientific Support
Answered: 2019-06-29
Question
Q: Are Boster Bio recombinant proteins and antibodies sterile?
B. Evans
Verified customer
Asked: 2018-04-05
Answer
A: although the vials are bottled using aseptic techniques, heat-treated vials, and sterile stock solutions, they are not considered or guaranteed to be sterile. If sterile material is required for an experiment, the material can be filtered through a 0.2 micron filter designed for use with biological fluids.
Boster Scientific Support
Answered: 2018-04-05
Question
Q: if the enzyme conjugated EGFR antibodies are mixed with the substrate, will that convert the substrate into the enzymatic reaction product? Or the enzyme function is only activated when the antibody is attached with the EGFR antigen?
Verified Customer
Verified customer
Asked: 2017-03-17
Answer
A: The enzyme is always active. Avoid contaminating the substrate with enzyme prior to the incubation otherwise it compromises the assay with false positive signal.
Boster Scientific Support
Answered: 2017-03-17



