This website uses cookies to ensure you get the best experience on our website.
- Table of Contents
2 Citations 8 Q&As
Facts about Alpha-2-macroglobulin.
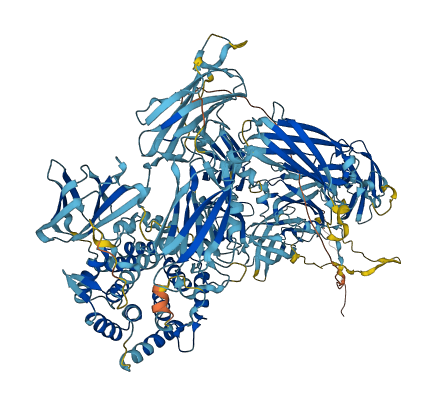
When a proteinase cleaves the bait region, a conformational change is induced in the protein which traps the proteinase. The entrapped enzyme remains active against low molecular weight substrates (activity against high molecular weight substrates is greatly reduced).
| Human | |
|---|---|
| Gene Name: | A2M |
| Uniprot: | P01023 |
| Entrez: | 2 |

| Belongs to: |
|---|
| protease inhibitor I39 (alpha-2-macroglobulin) family |

A2M; alpha 2Macroglobulin; alpha 2-Macroglobulin; alpha-2-M; alpha-2-macroglobulin; C3 and PZP-like alpha-2-macroglobulin domain-containing protein 5; CPAMD5; CPAMD5DKFZp779B086; FWP007; S863-7
Mass (kDA):
163.291 kDA

| Human | |
|---|---|
| Location: | 12p13.31 |
| Sequence: | 12; NC_000012.12 (9067708..9116229, complement) |
Secreted in plasma.
Secreted.




PMID: 2581245 by Kan C.-C., et al. Nucleotide sequence of cDNA encoding human alpha 2-macroglobulin and assignment of the chromosomal locus.
PMID: 15611997 by Lin V.K., et al. Alpha(2) macroglobulin, a PSA-binding protein, is expressed in human prostate stroma.
*More publications can be found for each product on its corresponding product page