This website uses cookies to ensure you get the best experience on our website.
- Table of Contents
17 Q&As
1 Citations
Facts about ADP-ribosylation factor 6.
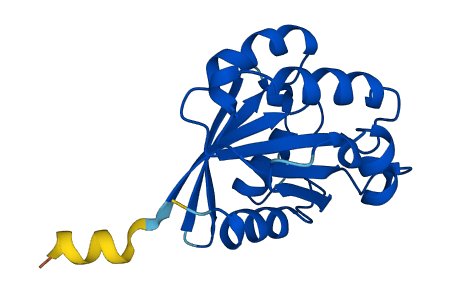
Involved in the regulation of dendritic spine growth, contributing to the regulation of dendritic branching and filopodia extension (PubMed:14978216). Plays an essential role in membrane trafficking, during junctional remodeling and epithelial polarization.
| Human | |
|---|---|
| Gene Name: | ARF6 |
| Uniprot: | P62330 |
| Entrez: | 382 |

| Belongs to: |
|---|
| small GTPase superfamily |

ADP-ribosylation factor 6; DKFZp564M0264
Mass (kDA):
20.082 kDA

| Human | |
|---|---|
| Location: | 14q21.3 |
| Sequence: | 14; NC_000014.9 (49893082..49897054) |
Ubiquitous, with higher levels in heart, substantia nigra, and kidney.
Cytoplasm, cytosol. Cell membrane; Lipid-anchor. Endosome membrane; Lipid-anchor. Recycling endosome membrane; Lipid-anchor. Cell projection, filopodium membrane; Lipid-anchor. Cell projection, ruffle. Cleavage furrow. Midbody, Midbody ring. Early endosome membrane; Lipid-anchor. Golgi apparatus, trans-Golgi network membrane; Lipid-anchor. Distributed uniformly on the plasma membrane, as well as throughout the cytoplasm during metaphase. Subsequently concentrated at patches in the equatorial region at the onset of cytokinesis, and becomes distributed in the equatorial region concurrent with cl






PMID: 1993656 by Tsuchiya M., et al. Molecular identification of ADP-ribosylation factor mRNAs and their expression in mammalian cells.
PMID: 14659046 by Lebeda R.A., et al. Sequence, genomic organization, and expression of the human ADP- ribosylation factor 6 (ARF6) gene: a class III ARF.