Product Info Summary
| SKU: | A02285-2 |
|---|---|
| Size: | 100 μg/vial |
| Reactive Species: | Human, Monkey, Mouse, Rat |
| Host: | Rabbit |
| Application: | ELISA, IF, IHC, WB |
Customers Who Bought This Also Bought
Product info
Product Name
Anti-Mannose Receptor/MRC1 Picoband® Antibody
SKU/Catalog Number
A02285-2
Size
100 μg/vial
Form
Lyophilized
Description
Boster Bio Anti-Mannose Receptor/MRC1 Picoband® Antibody catalog # A02285-2. Tested in ELISA, IF, IHC, WB applications. This antibody reacts with Human, Monkey, Mouse, Rat. The brand Picoband indicates this is a premium antibody that guarantees superior quality, high affinity, and strong signals with minimal background in Western blot applications. Only our best-performing antibodies are designated as Picoband, ensuring unmatched performance.
Storage & Handling
Store at -20˚C for one year from date of receipt. After reconstitution, at 4˚C for one month. It can also be aliquotted and stored frozen at -20˚C for six months. Avoid repeated freeze-thaw cycles.
Cite This Product
Anti-Mannose Receptor/MRC1 Picoband® Antibody (Boster Biological Technology, Pleasanton CA, USA, Catalog # A02285-2)
Host
Rabbit
Contents
Each vial contains 4 mg Trehalose, 0.9 mg NaCl and 0.2 mg Na2HPO4.
Clonality
Polyclonal
Isotype
Rabbit IgG
Immunogen
E.coli-derived human Mannose Receptor/MRC1 recombinant protein (Position: D21-A1140).
Cross-reactivity
No cross-reactivity with other proteins.
Reactive Species
A02285-2 is reactive to MRC1 in Human, Monkey, Mouse, Rat
Observed Molecular Weight
190-200 kDa
Calculated molecular weight
166.0 kDa
Background of MRC1
The mannose receptor (Cluster of Differentiation 206, CD206) is a C-type lectin primarily present on the surface of macrophages, immature dendritic cells and liver sinusoidal endothelial cells, but is also expressed on the surface of skin cells such as human dermal fibroblasts and keratinocytes. It is mapped to 10p12.33. The recognition of complex carbohydrate structures on glycoproteins is an important part of several biological processes, including cell-cell recognition, serum glycoprotein turnover, and neutralization of pathogens. The protein encoded by this gene is a type I membrane receptor that mediates the endocytosis of glycoproteins by macrophages. The protein has been shown to bind high-mannose structures on the surface of potentially pathogenic viruses, bacteria, and fungi so that they can be neutralized by phagocytic engulfment.
Antibody Validation
Boster validates all antibodies on WB, IHC, ICC, Immunofluorescence, and ELISA with known positive control and negative samples to ensure specificity and high affinity, including thorough antibody incubations.
Application & Images
Applications
A02285-2 is guaranteed for ELISA, IF, IHC, WB Boster Guarantee
Assay Dilutions Recommendation
The recommendations below provide a starting point for assay optimization. The actual working concentration varies and should be decided by the user.
Western blot, 0.25-0.5μg/ml, Human, Monkey, Mouse, Rat
Immunohistochemistry (Paraffin-embedded Section), 2-5μg/ml, Human, Mouse, Rat
Immunofluorescence, 5 μg/ml, Human, Mouse
ELISA, 0.1-0.5μg/ml, -
Positive Control
WB: rat lung tissue, mouse lung tissue, human liver tissue, monkey lung tissue
IHC: human liver cancer tissue, human tonsil tissue, mouse liver tissue, rat liver tissue
IF: human tonsil tissue, mouse liver tissue
Validation Images & Assay Conditions
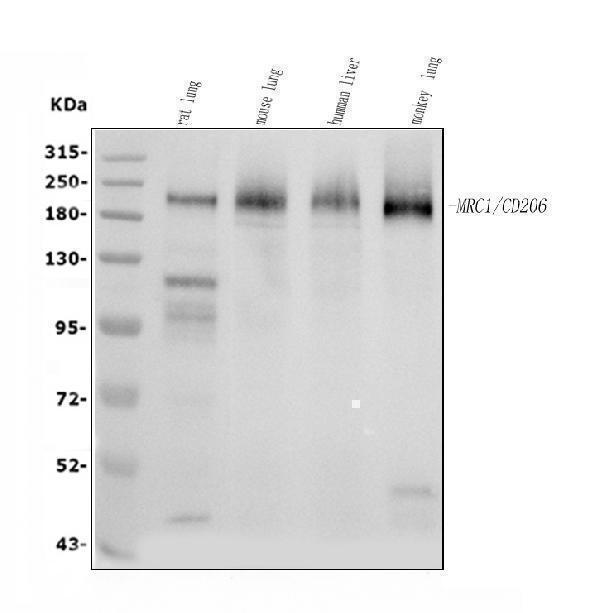
Click image to see more details
Western blot analysis of MRC1 using anti-MRC1 antibody (A02285-2).
Electrophoresis was performed on a 5-20% SDS-PAGE gel at 70V (Stacking gel) / 90V (Resolving gel) for 2-3 hours. The sample well of each lane was loaded with 30 ug of sample under reducing conditions.
Lane 1: rat lung tissue lysates,
Lane 2: mouse lung tissue lysates,
Lane 3: human liver tissue lysates,
Lane 4: monkey lung tissue lysates.
After electrophoresis, proteins were transferred to a nitrocellulose membrane at 150 mA for 50-90 minutes. Blocked the membrane with 5% non-fat milk/TBS for 1.5 hour at RT. The membrane was incubated with rabbit anti-MRC1 antigen affinity purified polyclonal antibody (Catalog # A02285-2) at 0.5 μg/mL overnight at 4°C, then washed with TBS-0.1%Tween 3 times with 5 minutes each and probed with a goat anti-rabbit IgG-HRP secondary antibody at a dilution of 1:5000 for 1.5 hour at RT. The signal is developed using an Enhanced Chemiluminescent detection (ECL) kit (Catalog # EK1002) with Tanon 5200 system. A specific band was detected for MRC1 at approximately 190-200 kDa. The expected band size for MRC1 is at 166 kDa.
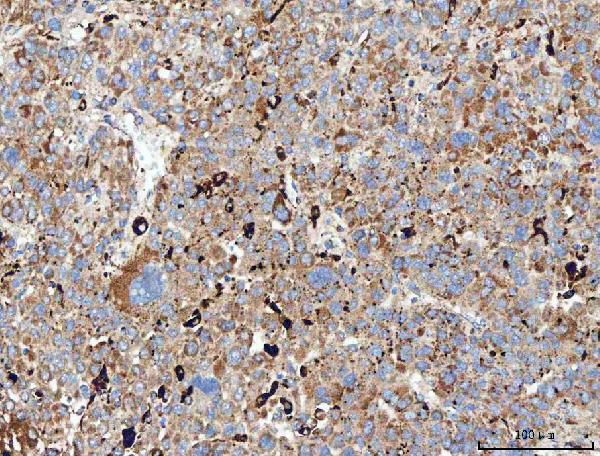
Click image to see more details
IHC analysis of MRC1 using anti-MRC1 antibody (A02285-2).
MRC1 was detected in a paraffin-embedded section of human liver cancer tissue. Heat mediated antigen retrieval was performed in EDTA buffer (pH 8.0, epitope retrieval solution). The tissue section was blocked with 10% goat serum. The tissue section was then incubated with 2 μg/ml rabbit anti-MRC1 Antibody (A02285-2) overnight at 4°C. Peroxidase Conjugated Goat Anti-rabbit IgG was used as secondary antibody and incubated for 30 minutes at 37°C. The tissue section was developed using HRP Conjugated Rabbit IgG Super Vision Assay Kit (Catalog # SV0002) with DAB as the chromogen.
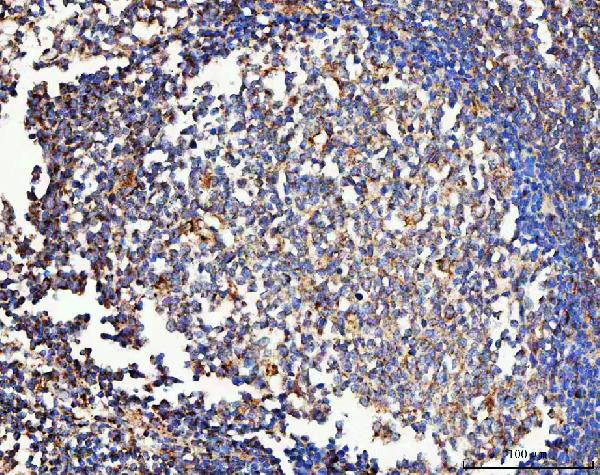
Click image to see more details
IHC analysis of MRC1 using anti-MRC1 antibody (A02285-2).
MRC1 was detected in a paraffin-embedded section of human tonsil tissue. Heat mediated antigen retrieval was performed in EDTA buffer (pH 8.0, epitope retrieval solution). The tissue section was blocked with 10% goat serum. The tissue section was then incubated with 2 μg/ml rabbit anti-MRC1 Antibody (A02285-2) overnight at 4°C. Peroxidase Conjugated Goat Anti-rabbit IgG was used as secondary antibody and incubated for 30 minutes at 37°C. The tissue section was developed using HRP Conjugated Rabbit IgG Super Vision Assay Kit (Catalog # SV0002) with DAB as the chromogen.
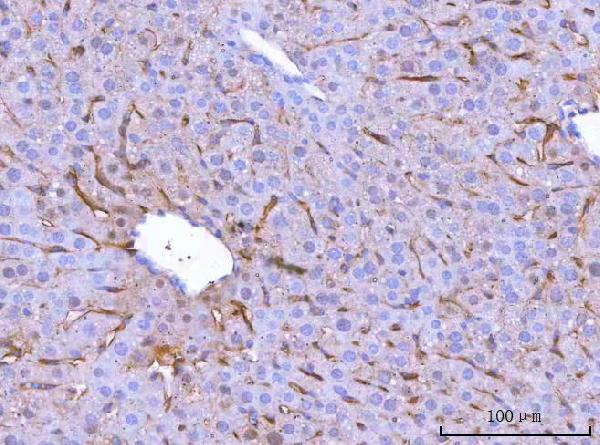
Click image to see more details
IHC analysis of MRC1 using anti-MRC1 antibody (A02285-2).
MRC1 was detected in a paraffin-embedded section of mouse liver tissue. Heat mediated antigen retrieval was performed in EDTA buffer (pH 8.0, epitope retrieval solution). The tissue section was blocked with 10% goat serum. The tissue section was then incubated with 2 μg/ml rabbit anti-MRC1 Antibody (A02285-2) overnight at 4°C. Peroxidase Conjugated Goat Anti-rabbit IgG was used as secondary antibody and incubated for 30 minutes at 37°C. The tissue section was developed using HRP Conjugated Rabbit IgG Super Vision Assay Kit (Catalog # SV0002) with DAB as the chromogen.
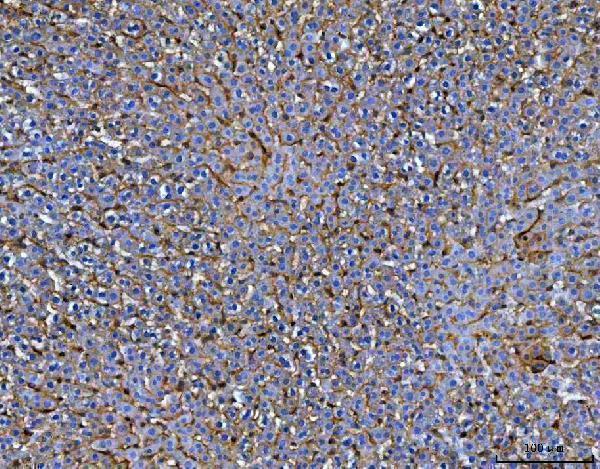
Click image to see more details
IHC analysis of MRC1 using anti-MRC1 antibody (A02285-2).
MRC1 was detected in a paraffin-embedded section of rat liver tissue. Heat mediated antigen retrieval was performed in EDTA buffer (pH 8.0, epitope retrieval solution). The tissue section was blocked with 10% goat serum. The tissue section was then incubated with 2 μg/ml rabbit anti-MRC1 Antibody (A02285-2) overnight at 4°C. Peroxidase Conjugated Goat Anti-rabbit IgG was used as secondary antibody and incubated for 30 minutes at 37°C. The tissue section was developed using HRP Conjugated Rabbit IgG Super Vision Assay Kit (Catalog # SV0002) with DAB as the chromogen.
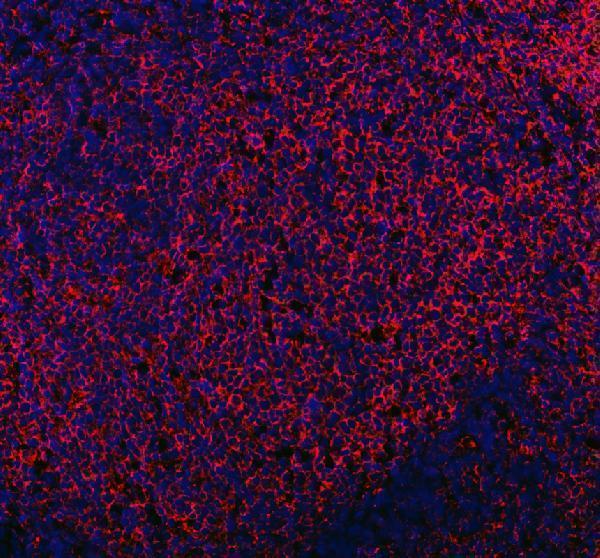
Click image to see more details
IF analysis of MRC1 using anti-MRC1 antibody (A02285-2).
MRC1 was detected in a paraffin-embedded section of human tonsil tissue. Heat mediated antigen retrieval was performed in EDTA buffer (pH 8.0, epitope retrieval solution). The tissue section was blocked with 10% goat serum. The tissue section was then incubated with 5 μg/mL rabbit anti-MRC1 Antibody (A02285-2) overnight at 4°C. DyLight®550 Conjugated Goat Anti-Rabbit IgG (BA1135) was used as secondary antibody at 1:500 dilution and incubated for 30 minutes at 37°C. The section was counterstained with DAPI. Visualize using a fluorescence microscope and filter sets appropriate for the label used.
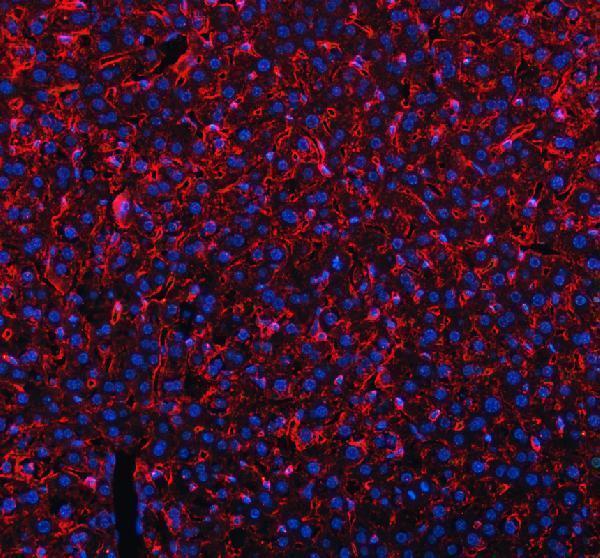
Click image to see more details
IF analysis of MRC1 using anti-MRC1 antibody (A02285-2).
MRC1 was detected in a paraffin-embedded section of mouse liver tissue. Heat mediated antigen retrieval was performed in EDTA buffer (pH 8.0, epitope retrieval solution). The tissue section was blocked with 10% goat serum. The tissue section was then incubated with 5 μg/mL rabbit anti-MRC1 Antibody (A02285-2) overnight at 4°C. DyLight®550 Conjugated Goat Anti-Rabbit IgG (BA1135) was used as secondary antibody at 1:500 dilution and incubated for 30 minutes at 37°C. The section was counterstained with DAPI. Visualize using a fluorescence microscope and filter sets appropriate for the label used.
Click image to see more details
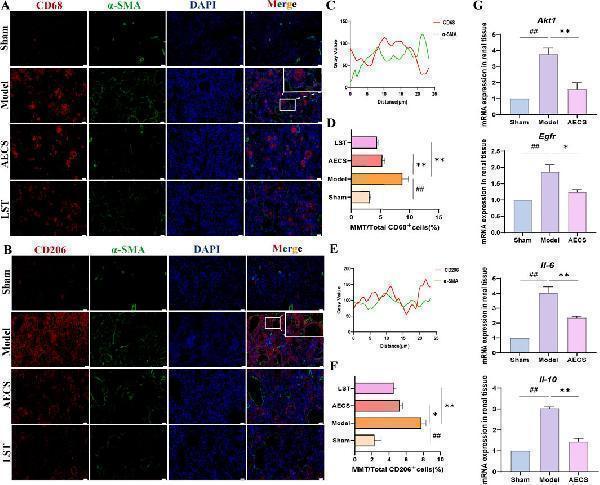
CS attenuates MMT in RIF rats. (A) Co-staining of the α-SMA (green) with CD68 (red) by Double Immunofluorescence. (B) Co-localization analysis of CD68+α-SMA + MMT cells. (C) Staining intensity of CD68+α-SMA + MMT cells was quantified. (D) Co-staining of the α-SMA (green) with CD206 (red) by Double Immunofluorescence. (E) Co-localization analysis of CD206+α-SMA + MMT cells. (F) Staining intensity of CD206+α-SMA + MMT cells was quantified (n = 4). (G) mRNA Expression of AKT1, EGFR, IL-6, and IL-10 in RIF rats kidney (n = 3). ## p < 0.01 vs. the sham group; * p < 0.05 vs. the model group; ** p < 0.01 vs. the model group. Scale bars (40 μm, 10 μm).
Index in PubMed under a CC BY license. PMID: 41142245
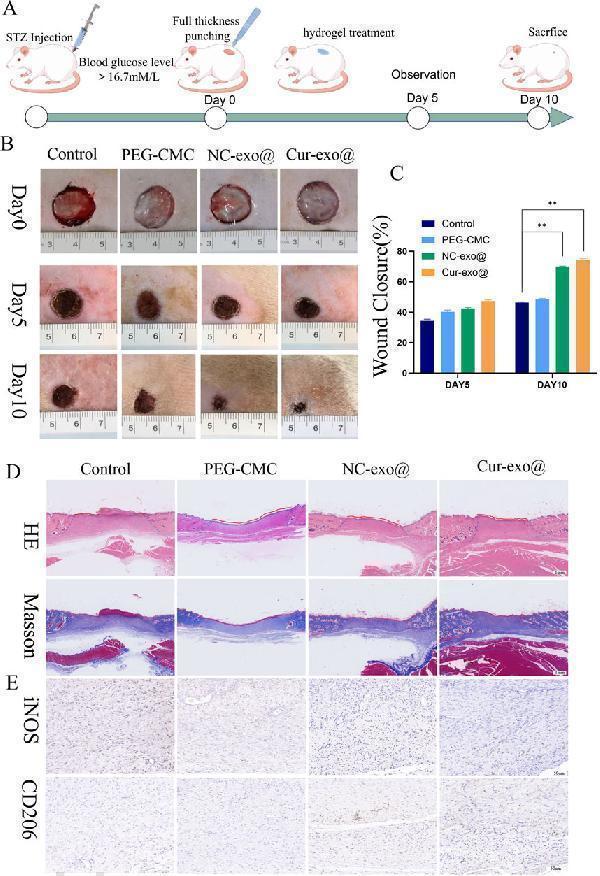
Click image to see more details
Wound healing with different hydrogels in vivo . (A) Preparation and observation of diabetic chronic wounds related to orthopedics. (B) Photographs of the wound treated with the hydrogel in different hydrogel groups. (C) Wound recovery curve of different hydrogel groups. (D) H&E staining and Masson staining of wound tissue on day 10 in different hydrogel groups. (E) IHC staining of wound tissue in iNOS and CD206.
Index in PubMed under a CC BY license. PMID: 41190287
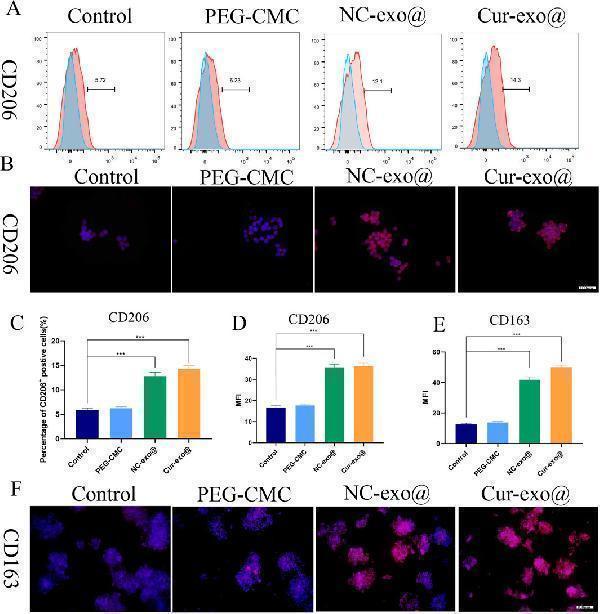
Click image to see more details
Hydrogel-induced macrophage M2 polarization in vitro . (A) Flow cytometry analysis of the macrophage surface marker CD206. (B) Immunofluorescence of CD206 in macrophages treated with different hydrogels. (C) Quantitative analysis of flow cytometry in CD206. (D) Mean fluorescence intensity of CD206 in macrophages treated with different hydrogels. (E, F) Quantitative analysis and immunofluorescence of CD163 in macrophages treated with different hydrogels.
Index in PubMed under a CC BY license. PMID: 41190287
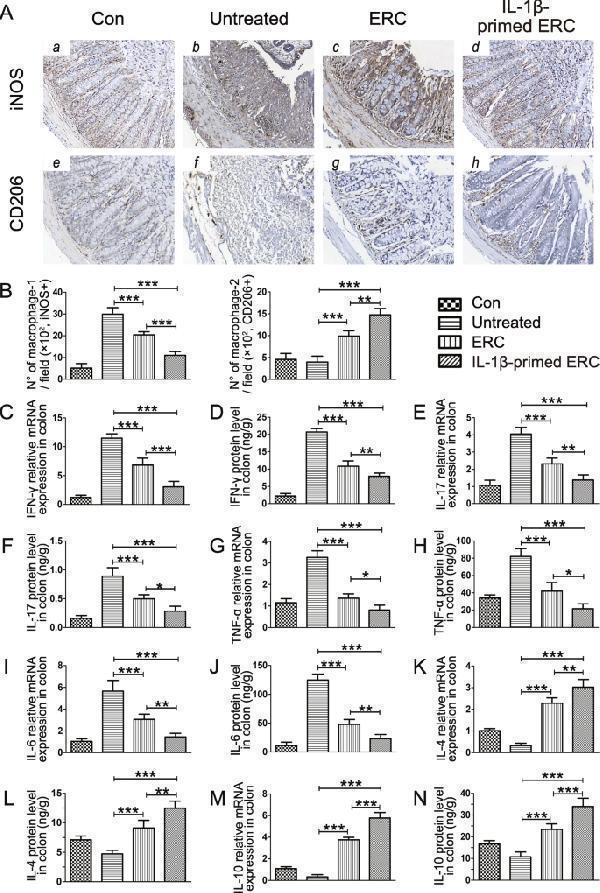
Click image to see more details
IL-1β-primed ERCs reduced macrophage infiltration and inflammatory cytokine expressions in colons. Intra-colon macrophage infiltration was evaluated by immunohistochemical staining. Specifically, we stained iNOS for detecting M1 cell infiltration, and CD206 for measuring M2 cell infiltration. The represent IHC images of mouse colons and quantitive data for cell counts of each group are shown in A (× 200) and B , respectively. The concentrations of inflammatory cytokine productions in colonic tissues were determined by ELISA kit and the relative mRNA expression changes were performed by real-time PCR. IFN-γ ( C , D ), IL-17 ( E , F ), TNF-α ( G , H ), IL-6 ( I , J ), IL-4 ( K , L ), and IL-10 ( M , N ) were shown, respectively, which are majorly secreted by Th1, Th2, Th17 cells, or CD206 + macrophages, and closely associated with the development of UC. Data were presented as mean ± standard deviation, and p values were calculated by using one-way ANOVA followed by the least significant difference (LSD) test ( n = 6, * p < 0.05, ** p < 0.01, *** p < 0.001)
Index in PubMed under a CC BY license. PMID: 34090510
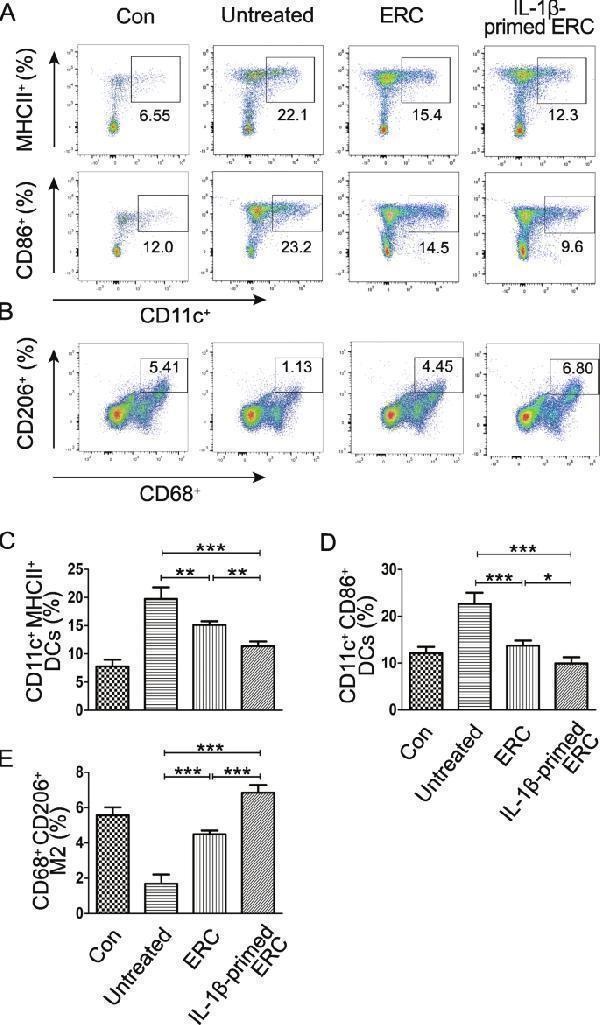
Click image to see more details
IL-1β-primed ERCs reduced the population of mature DCs, but increased M2 macrophages in colitis mice. To determine whether each treatment has an influence on regulating DC and macrophage phenotypes, anti-CD11c antibody and antigen presenting-related antibodies (anti-MHCII, anti-CD86) were used to measure mature DCs, while anti-CD68 antibody and anti-CD206 antibody were used for M2 phenotype macrophages in spleens. A Representative dot plots of CD11c + MHCII + DCs and CD11c + CD86 + DCs in spleens. B Dot plots of CD68 + CD206 + macrophages. C – E Percentage of CD11c + MHCII + DCs, CD11c + CD86 + DCs, and CD68 + CD206 + macrophages, respectively. Data were mean ± SD ( n = 6, * p < 0.05, ** p < 0.01, *** p < 0.001). p values were analyzed by one-way ANOVA followed by the LSD test
Index in PubMed under a CC BY license. PMID: 34090510
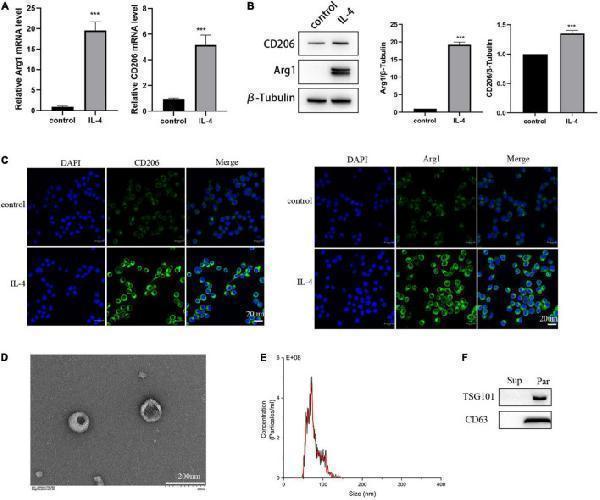
Click image to see more details
Identification of the isolated polarized M2 microglia and exosomes. (A) The expression of M2 microglia markers Arg1 and CD206 detected by RT-PCR. (B) The expression of M2 microglia markers Arg1 and CD206 detected by Western blot analysis. (C) The expression of M2 microglia markers Arg1 and CD206 detected by immunofluorescence. (D) Images of M2-EXOs detected using TEM. (E) Measurements of particle sizes range of the isolated exosomes using NTA. (F) Detection of exosomal marker proteins CD63 and TSG101 using western blot. All experiments were repeated three times. *** P < 0.001 vs. control.
Index in PubMed under a CC BY license. PMID: 35573832
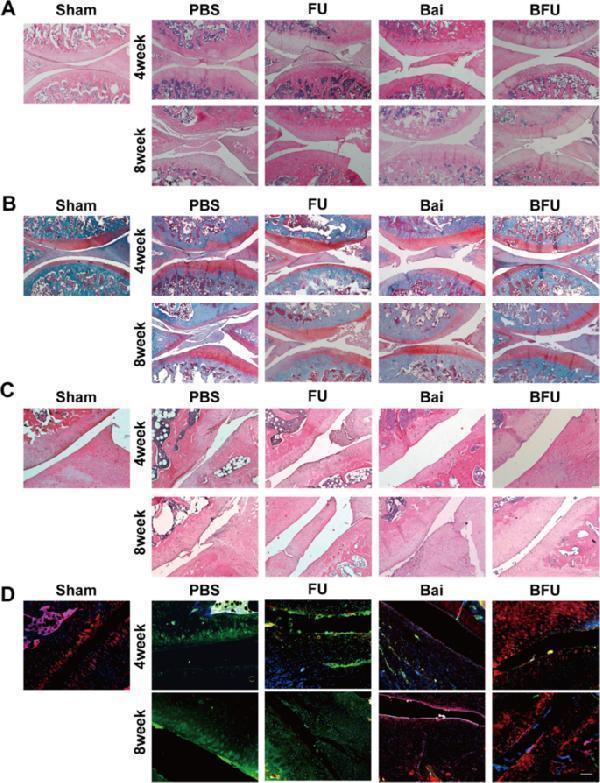
Click image to see more details
Histological staining evaluation. FU (FA-UIO-66-NH 2 ), BFU (Bai@FA-UIO-66-NH 2 ). ( A ) H&E staining and ( B ) Safranin O staining of the knee joint of SD rats and ( C ) partial magnified detail (bar = 200 μm). ( D ) Immunofluorescence staining of iNOS/CD206/DAPI of the synovial of SD rats (scale bar = 200 μm)
Index in PubMed under a CC BY license. PMID: 38724958
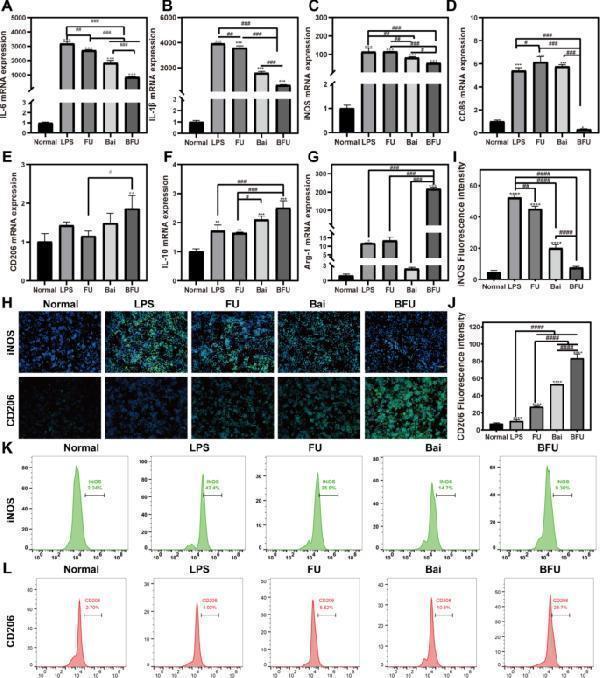
Click image to see more details
In vitro anti-inflammatory effect produced by driving the polarization of macrophage M1 towards M2. FU (FA-UIO-66-NH 2 ), BFU (Bai@FA-UIO-66-NH 2 ). (A-G) In vitro mRNA expression of IL-1β, IL-6, iNOS, Arg-1, CD86, IL-10 and CD206 . H-J) Immunofluorescence staining of iNOS and CD206 after different treatments. (H) (scale bar = 100 μm) and Corresponding fluorescence intensity quantification (I-J). Flow cytometry analysis of specific marker of M1 macrophage iNOS (K) and M2 macrophage CD206 (L). ( n = 3, mean ± SD, “*” symbol compared with normal group, * P < 0.05, ** P < 0.01, *** P < 0.001, **** P < 0.0001 and “” symbol compared between groups, # P < 0.05, ## P < 0.01, ### P < 0.001, #### P < 0.0001)
Index in PubMed under a CC BY license. PMID: 38724958
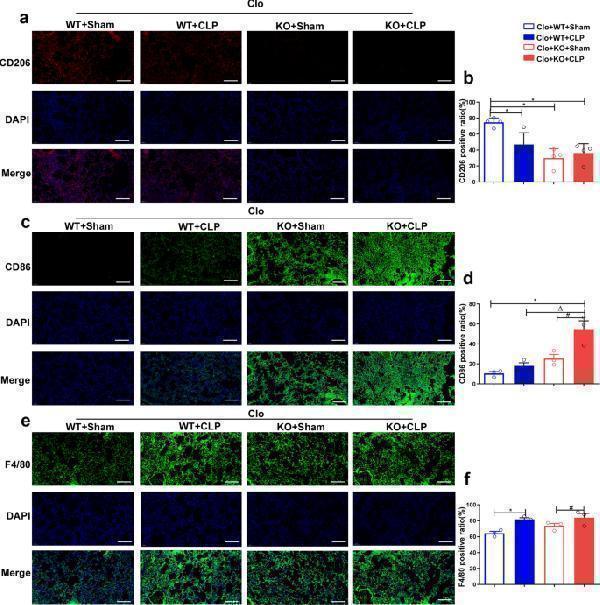
Click image to see more details
Mice reconstituted with FGF2 KO macrophages and subjected to CLP demonstrate increased M1 polarization in lung tissue. a - f The presence and levels of CD206, CD86, and F4/80 markers on macrophages within lung tissue were identified and quantitatively assessed using immunofluorescence staining. Bar is 20 μm. * p < 0.05, vs. WT; Δ p < 0.05 vs. WT + LPS; # p < 0.05 vs. FGF2 KO
Index in PubMed under a CC BY license. PMID: 39436561
Click image to see more details
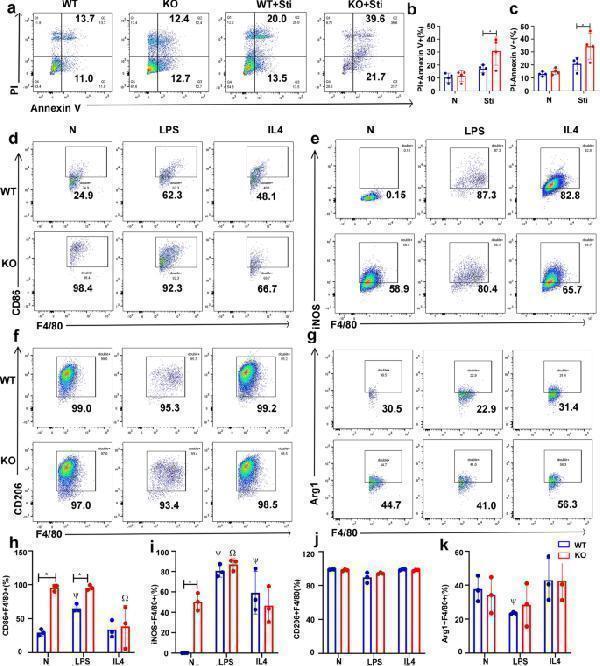
Effect of FGF2 deficiency on BMDM apoptosis and polarization. a – c FGF2 deletion increased BMDM apoptosis. a Apoptosis in BMDM deprived of FBS for 24 h was assessed by flow cytometry ( n = 4). b - c Percentage of PI + Annexin V + and PI- Annexin V + BMDM after starvation. d - k FGF2 deletion in BMDM promoted M1 polarization. d - g Flow cytometric analysis of macrophage markers in BMDM treated with LPS or IL4, including CD86, iNOS, CD206, and Arg1 ( n = 3). h - k The levels of CD86, iNOS, CD206 and Arg1 in BMDM after treatment with LPS or IL4. N represents no treatment; * p < 0.05, vs. WT; Ψ p < 0.05, vs. N + WT; Ω p < 0.05, vs. N + FGF2 KO
Index in PubMed under a CC BY license. PMID: 39436561
Click image to see more details
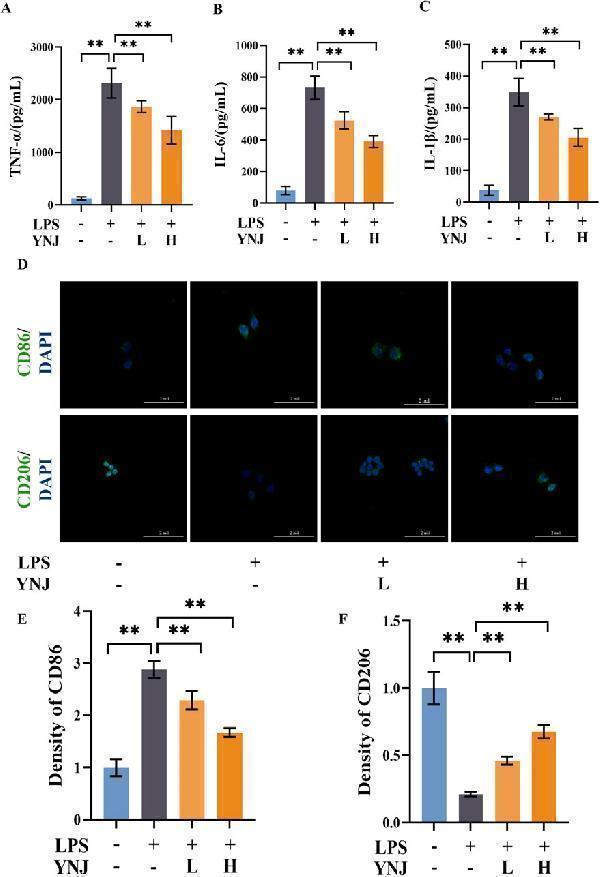
YNJ reduces the secretion of inflammatory factors and promotes M2 macrophage polarization. (A–C) ELISA of TNF-α (A) , IL-6 (B) , and IL-1β (C) contents in the supernatant of cultured RAW264.7 macrophages. (D) IF images of CD86 and CD206 detection in cultured RAW264.7 macrophages. (E, F) Statistical analysis of CD86 and CD206 expression by IF. The data are presented as the mean ± SD (n = 6). ** P < 0.01.
Index in PubMed under a CC BY license. PMID: 39925847

Click image to see more details
PF-127/hADSCs-Exos complex treatment inhibits inflammatory reaction. a Representative images of TNF-α immunostaining at 4, 7, and 10 days after treatment. Scale bar = 20 µm. b Quantification of TNF-α + IHC stained tissues. c Representative images illustrating IHC results of IL-6 at 4, 7, and 10 days after surgery. Scale bar = 20 µm. d Quantification of IL-6 + IHC stained tissues. e IHC images of wound sections stained with CD68 on days 4, 7, and 10 post-wounding. Scale bar = 20 µm. f Quantification of the number of CD68 positive cells in the wound area on days 4, 7, and 10. g IHC images of wound sections stained with CD206 at days 4, 7, and 10 post-wounding. Scale bar = 20 µm. h Quantification of the number of CD206 positive cells in the wound area on days 4, 7, and 10. In b, d, and f , data are shown as mean ± SEM; n = 6 for each group. * p < 0.05, ** p < 0.01, *** p < 0.001, and **** p < 0.0001 versus vehicle control group
Index in PubMed under a CC BY license. PMID: 35941707
Specific Publications For Anti-Mannose Receptor/MRC1 Picoband® Antibody (A02285-2)
Loading publications
Recommended Resources
Here are featured tools and databases that you might find useful.
- Boster's Pathways Library
- Protein Databases
- Bioscience Research Protocol Resources
- Data Processing & Analysis Software
- Photo Editing Software
- Scientific Literature Resources
- Research Paper Management Tools
- Molecular Biology Software
- Primer Design Tools
- Bioinformatics Tools
- Phylogenetic Tree Analysis
Customer Reviews
Have you used Anti-Mannose Receptor/MRC1 Picoband® Antibody?
Share your experimental results or join a short interview to earn up to $1,000 in product credits or other rewards.
1 Reviews For Anti-Mannose Receptor/MRC1 Picoband® Antibody
Immunohistochemistry for Anti-Mannose Receptor/MRC1
Excellent
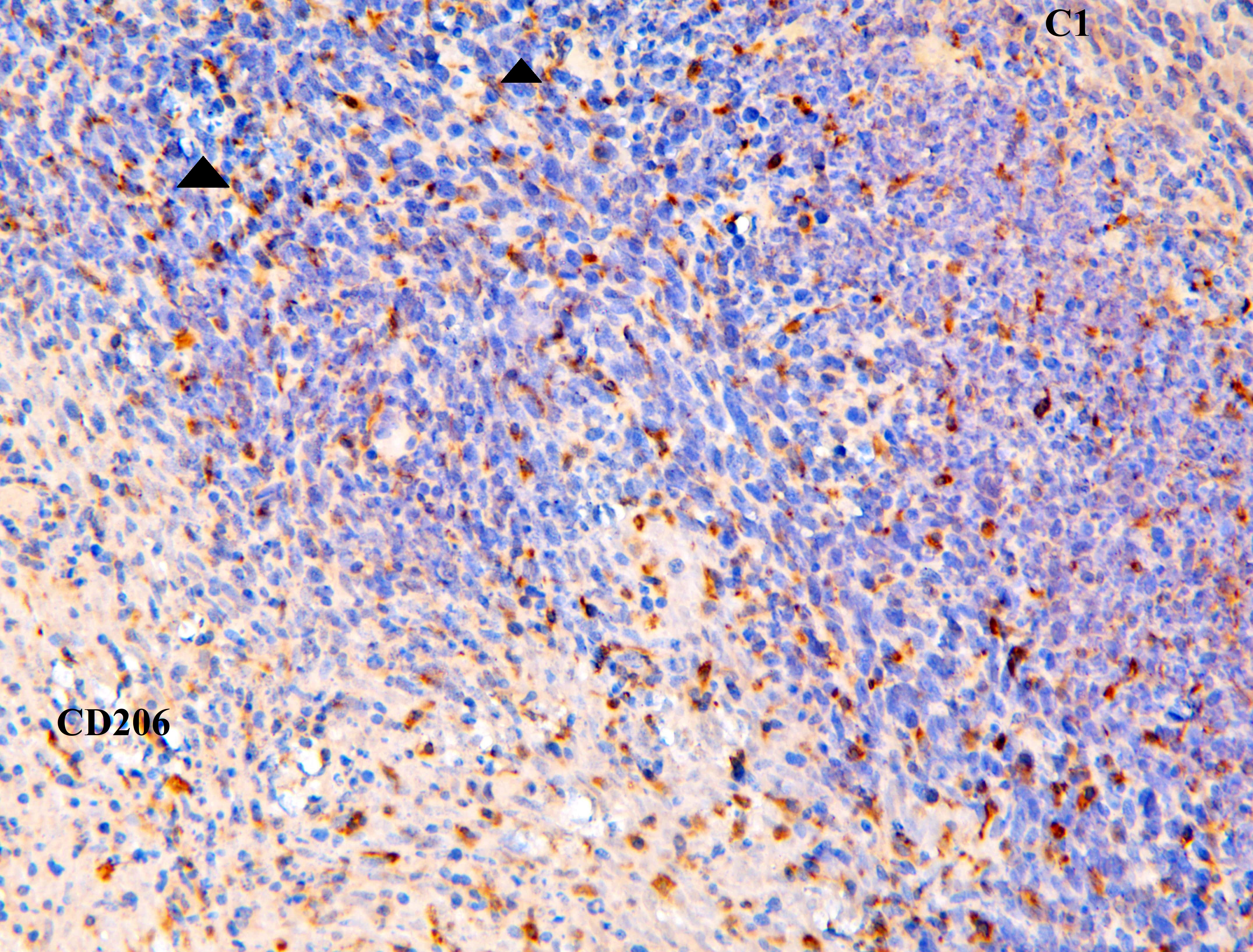
| SKU | A02285-2 |
|---|---|
| Application | Immunohistochemistry (paraffin-embedded) |
| Blocking step | 5% BSA as a blocking agent for 30 min at 37°C |
| Sample | Mouse skin |
| Fixative | Fixed with 4% paraformaldehyde |
| Primary Ab Incubation | 37°C for 30 minutes |
| Primary Ab Incubation diluent | 5% BSA in TBS |
| Primary Ab Concentration | 1ug/ml |
| Secondary Antibody | SABC kit from Boster Bio, (SA1022) |
| Secondary Ab Dilution | The kit was ready to use, no dilution needed |
| Secondary Ab Incubation | at 37°C for 30 min |
Verified Customer
Verified customer
Submitted 2020-02-06
Customer Q&As
Have a question?
Find answers in Q&As, reviews.
Can't find your answer?
Submit your question





