Product Info Summary
| SKU: | PB9449 |
|---|---|
| Size: | 100 μg/vial |
| Reactive Species: | Human, Mouse, Rat |
| Host: | Rabbit |
| Application: | IF, IHC, WB |
Customers Who Bought This Also Bought
Product info
Product Name
Anti-Tyrosine Hydroxylase/TH Antibody Picoband®
SKU/Catalog Number
PB9449
Size
100 μg/vial
Form
Lyophilized
Description
Boster Bio Anti-Tyrosine Hydroxylase/TH Antibody Picoband® catalog # PB9449. Tested in IF, IHC, WB applications. This antibody reacts with Human, Mouse, Rat. The brand Picoband indicates this is a premium antibody that guarantees superior quality, high affinity, and strong signals with minimal background in Western blot applications. Only our best-performing antibodies are designated as Picoband, ensuring unmatched performance.
Storage & Handling
Store at -20˚C for one year from date of receipt. After reconstitution, at 4˚C for one month. It can also be aliquotted and stored frozen at -20˚C for six months. Avoid repeated freeze-thaw cycles.
Cite This Product
Anti-Tyrosine Hydroxylase/TH Antibody Picoband® (Boster Biological Technology, Pleasanton CA, USA, Catalog # PB9449)
Host
Rabbit
Contents
Each vial contains antibody formulated with stabilizing components, 0.9 mg NaCl, 0.2 mg Na2HPO4, and 0.05 mg NaN3.
*This antibody is supplied in a stabilized formulation.
Compatibility with conjugation reactions depends on the chemistry of the conjugation method used.
For conjugation methods that are not compatible with the stabilizing components present in this formulation, a carrier-free antibody format is required.
Clonality
Polyclonal
Isotype
Rabbit IgG
Immunogen
A synthetic peptide corresponding to a sequence in the middle region of human Tyrosine Hydroxylase, identical to the related mouse and rat sequences.
Cross-reactivity
No cross-reactivity with other proteins
Reactive Species
PB9449 is reactive to TH in Human, Mouse, Rat
Observed Molecular Weight
59 kDa
Calculated molecular weight
58.6 kDa
Background of TH
TH is equal to tyrosine hydroxylase. The protein encoded by this gene is involved in the conversion of tyrosine to dopamine. It is the rate-limiting enzyme in the synthesis of catecholamines, hence plays a key role in the physiology of adrenergic neurons. Mutations in this gene have been associated with autosomal recessive Segawa syndrome. Alternatively spliced transcript variants encoding different isoforms have been noted for this gene. In humans, tyrosine hydroxylase is encoded by the TH gene, and the enzyme is present in the central nervous system (CNS), peripheral sympathetic neurons and the adrenal medulla. Tyrosine hydroxylase, phenylalanine hydroxylase and tryptophan hydroxylase together make up the family of aromatic amino acid hydroxylases (AAAHs).
Antibody Validation
Boster validates all antibodies on WB, IHC, ICC, Immunofluorescence, and ELISA with known positive control and negative samples to ensure specificity and high affinity, including thorough antibody incubations.
Application & Images
Applications
PB9449 is guaranteed for IF, IHC, WB Boster Guarantee
Recommend Dilution
| Application | Dilution | Species |
|---|---|---|
| Western blot | 0.1-0.5μg/ml | Mouse, Rat |
| Immunohistochemistry (Paraffin-embedded Section) | 2-5μg/ml | Human, Mouse, Rat |
| Immunofluorescence | 5μg/ml | Mouse, Rat |
Tested application
Suggested blocking solution with 5% non-fat milk or BSA; (*)Recommended protein loading: 20-40 µg per lane
Use TE buffer pH 9.0 for antigen retrieval; (*) citrate buffer pH 6.0 is an alternative.
Validation Images & Assay Conditions
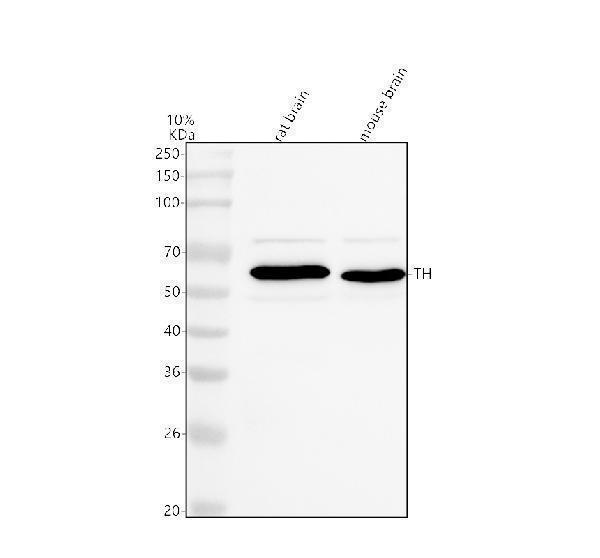
Click image to see more details
Western blot analysis of Tyrosine Hydroxylase/TH using anti-Tyrosine Hydroxylase/TH antibody (PB9449).
Electrophoresis was performed on a 10% SDS-PAGE gel at 80V (Stacking gel) / 120V (Resolving gel) for 2 hours. The sample well of each lane was loaded with 30 ug of sample under reducing conditions.
Lane 1: rat brain tissue lysates,
Lane 2: mouse brain tissue lysates.
After electrophoresis, proteins were transferred to a nitrocellulose membrane at 150 mA for 50-90 minutes. Blocked the membrane with 5% non-fat milk/TBS for 1.5 hour at RT. The membrane was incubated with rabbit anti-Tyrosine Hydroxylase/TH antigen affinity purified polyclonal antibody (Catalog # PB9449) at 0.5 μg/mL overnight at 4°C, then washed with TBS-0.1%Tween 3 times with 5 minutes each and probed with a goat anti-rabbit IgG-HRP secondary antibody at a dilution of 1:5000 for 1.5 hour at RT. The signal is developed using an ECL Plus Western Blotting Substrate (Catalog # AR1196-200) with Tanon 5200 system. A specific band was detected for Tyrosine Hydroxylase/TH at approximately 59 kDa. The expected band size for Tyrosine Hydroxylase/TH is at 59 kDa.
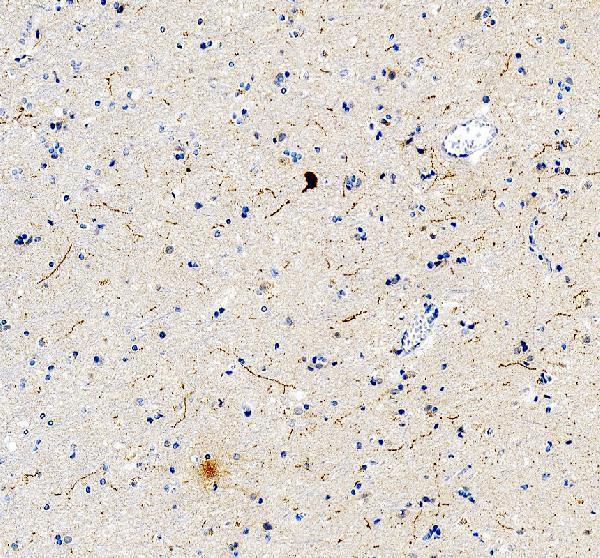
Click image to see more details
IHC analysis of Tyrosine Hydroxylase/TH using anti-Tyrosine Hydroxylase/TH antibody (PB9449).
Tyrosine Hydroxylase/TH was detected in a paraffin-embedded section of human brain tissue. Heat mediated antigen retrieval was performed in EDTA buffer (pH 8.0, epitope retrieval solution). The tissue section was blocked with 10% goat serum. The tissue section was then incubated with 2 μg/ml rabbit anti-Tyrosine Hydroxylase/TH Antibody (PB9449) overnight at 4°C. Peroxidase Conjugated Goat Anti-rabbit IgG was used as secondary antibody and incubated for 30 minutes at 37°C. The tissue section was developed using HRP Conjugated Rabbit IgG Super Vision Assay Kit (Catalog # SV0002) with DAB as the chromogen.
Click image to see more details
IHC analysis of Tyrosine Hydroxylase/TH using anti-Tyrosine Hydroxylase/TH antibody (PB9449).
Tyrosine Hydroxylase/TH was detected in a paraffin-embedded section of normal mouse brain (nomal group) and Alzheimer’s model mouse brain (model group) tissue. Heat mediated antigen retrieval was performed in EDTA buffer (pH 8.0, epitope retrieval solution). The tissue section was blocked with 10% goat serum. The tissue section was then incubated with 1:500 rabbit anti-Tyrosine Hydroxylase/TH Antibody (PB9449) overnight at 4°C. A two-step IHC kit was used for 30 minutes at 37℃,The tissue section was developed using HRP Conjugated Rabbit IgG Super Vision Assay Kit (Catalog # SV0002) with DAB as the chromogen.

Click image to see more details
IHC analysis of Tyrosine Hydroxylase/TH using anti-Tyrosine Hydroxylase/TH antibody (PB9449).
Tyrosine Hydroxylase/TH was detected in a paraffin-embedded section of mouse brain tissue. Heat mediated antigen retrieval was performed in EDTA buffer (pH 8.0, epitope retrieval solution). The tissue section was blocked with 10% goat serum. The tissue section was then incubated with 2 μg/ml rabbit anti-Tyrosine Hydroxylase/TH Antibody (PB9449) overnight at 4°C. Peroxidase Conjugated Goat Anti-rabbit IgG was used as secondary antibody and incubated for 30 minutes at 37°C. The tissue section was developed using HRP Conjugated Rabbit IgG Super Vision Assay Kit (Catalog # SV0002) with DAB as the chromogen.

Click image to see more details
IHC analysis of Tyrosine Hydroxylase/TH using anti-Tyrosine Hydroxylase/TH antibody (PB9449).
Tyrosine Hydroxylase/TH was detected in a paraffin-embedded section of rat brain tissue. Heat mediated antigen retrieval was performed in EDTA buffer (pH 8.0, epitope retrieval solution). The tissue section was blocked with 10% goat serum. The tissue section was then incubated with 2 μg/ml rabbit anti-Tyrosine Hydroxylase/TH Antibody (PB9449) overnight at 4°C. Peroxidase Conjugated Goat Anti-rabbit IgG was used as secondary antibody and incubated for 30 minutes at 37°C. The tissue section was developed using HRP Conjugated Rabbit IgG Super Vision Assay Kit (Catalog # SV0002) with DAB as the chromogen.
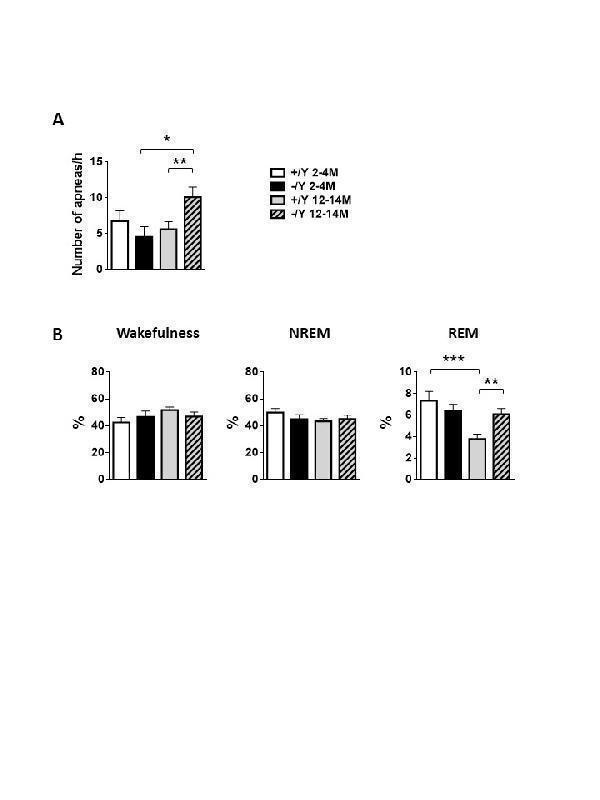
Click image to see more details
Sleep apnea and hypnic occurrence rate in Cdkl5 KO mice. (A) Apnea occurrence rate during non-rapid-eye-movement sleep (NREM) sleep period in young adult Cdkl5 -/Y (n = 8) and Cdkl5 +/Y (n = 9) mice, and middle-aged Cdkl5 -/Y (n = 14) and Cdkl5 +/Y (n = 14) mice. (B) Percentage of time spent in wakefulness, in NREM, and in rapid-eye-movement sleep (REM) during whole-body-plethysmography recordings in the same animals as in A. Values represent mean ± SEM. *p< 0.05, **p< 0.01, ***p< 0.001 (Fisher’s LSD test after two-way ANOVA).
Index in PubMed under a CC BY license. PMID: 34094641

Click image to see more details
IF analysis of Tyrosine Hydroxylase/TH using anti-Tyrosine Hydroxylase/TH antibody (PB9449).
Tyrosine Hydroxylase/TH was detected in a paraffin-embedded section of mouse brian tissue. Heat mediated antigen retrieval was performed in EDTA buffer (pH 8.0, epitope retrieval solution). The tissue section was blocked with 10% goat serum. The tissue section was then incubated with 5 μg/mL rabbit anti-Tyrosine Hydroxylase/TH Antibody (PB9449) overnight at 4°C. Biotin conjugated goat anti-rabbit IgG (BA1003) was used as secondary antibody and incubated for 30 minutes at 37°C. The tissue section was developed using DyLight®488 Conjugated Avidin (BA1128). The section was counterstained with DAPI. Visualize using a fluorescence microscope and filter sets appropriate for the label used.

Click image to see more details
IF analysis of Tyrosine Hydroxylase/TH using antiTyrosine Hydroxylase/TH antibody (PB9449).
Tyrosine Hydroxylase/TH was detected in a paraffin-embedded section of rat brian tissue. Heat mediated antigen retrieval was performed in EDTA buffer (pH 8.0, epitope retrieval solution). The tissue section was blocked with 10% goat serum. The tissue section was then incubated with 5 μg/mL rabbit anti-Tyrosine Hydroxylase/TH Antibody (PB9449) overnight at 4°C. Biotin conjugated goat anti-rabbit IgG (BA1003) was used as secondary antibody and incubated for 30 minutes at 37°C. The tissue section was developed using DyLight®488 Conjugated Avidin (BA1128). The section was counterstained with DAPI. Visualize using a fluorescence microscope and filter sets appropriate for the label used.
Click image to see more details
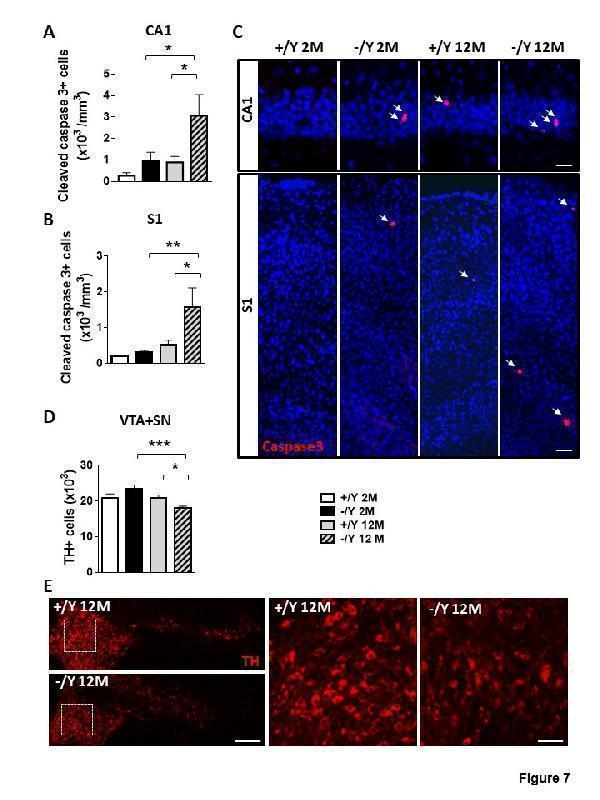
Increased neuronal cell death in Cdkl5 KO mice. (A-B) Quantification of cells positive for cleaved caspase-3 in the CA1 layer of the hippocampus (A) and in layer II/III of the somatosensory cortex (S1, B) from young adult Cdkl5 +/Y (n = 4) and Cdkl5 -/Y (n = 4) mice, and middle-aged Cdkl5 +/Y (n = 4) and Cdkl5 -/Y (n = 4) mice. (C) Representative images, one from each group, of cells, in the hippocampal CA1 region (upper panel) and in layer II/III of the somatosensory cortex (S1 , lower panel), immunopositive for cleaved caspase-3 (red) and stained with Hoechst (blue). Scale bar = 50 μm. (D) Quantification of the total number of TH-positive neurons in the substantia nigra (SN) and ventral tegmental area (VTA) from young adult Cdkl5 +/Y (n = 5) and Cdkl5 -/Y (n = 4) mice, and middle-aged Cdkl5 +/Y (n = 4) and Cdkl5 - /Y (n = 5) mice. (E) Representative images of tyrosine hydroxylase (TH) immunofluorescence staining in the VTA and SN of middle-aged Cdkl5 +/Y and Cdkl5 -/Y mice. Scale bar = 100 μm. The dotted boxes indicate the VTA region shown at a higher magnification in the right panel. Scale bar = 40 μm. Values are represented as means ± SE. *p<0.05, **p<0.01, ***p<0.001 (Fisher’s LSD test after two-way ANOVA).
Index in PubMed under a CC BY license. PMID: 34094641
Click image to see more details
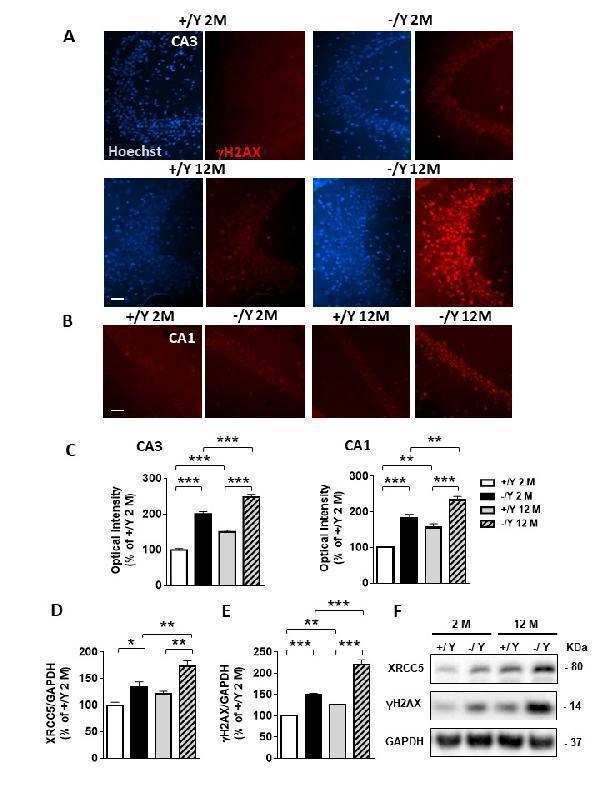
Increased DNA damage in the hippocampus of Cdkl5 KO mice. (A-B) Representative fluorescent images of the CA3 (A) and CA1 (B) hippocampal regions of adult and middle-aged Cdkl5 +/Y and Cdkl5 -/Y mice, immunostained for γH2AX and counterstained with Hoechst. Scale bar = 500 μm (C) Quantification of γH2AX nuclear signal intensity in the CA3 and CA1 pyramidal layer of young adult Cdkl5 +/Y (n = 3) and Cdkl5 -/Y (n = 3) mice, and middle-aged Cdkl5 +/Y (n = 4) and Cdkl5 -/Y (n = 3) mice. (D) Western blot analysis of γH2AX levels normalized to GAPDH levels in the hippocampus of young adult Cdkl5 +/Y (n = 4) and Cdkl5 -/Y (n = 5) mice, and middle-aged Cdkl5 +/Y (n = 3) and Cdkl5 -/Y (n = 3) mice. (E) Western blot analysis of XRCC5 levels normalized to GAPDH levels in the hippocampus of young adult Cdkl5 +/Y (n = 4) and Cdkl5 -/Y (n = 5) mice, and middle-aged Cdkl5 +/Y (n = 4) and Cdkl5 -/Y (n = 3) mice. (F) Immunoblot images of γH2AX, XRCC5, and GAPDH levels in protein extracts from one animal of each experimental group. Data in Western blot analysis are expressed as a percentage of the values compared to young adult Cdkl5 +/Y mice. Values are represented as means ± SE. *p< 0.05 and **p< 0.01, ***p<0.001 (Fisher’s LSD test after two-way ANOVA).
Index in PubMed under a CC BY license. PMID: 34094641
Click image to see more details
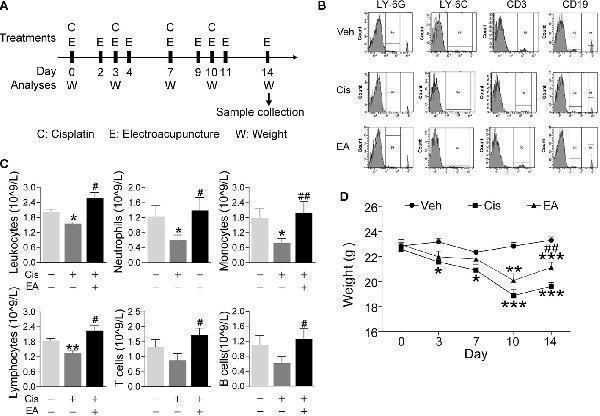
Electroacupuncture prevented cisplatin-induced leukopenia. (A) Experimental flowchart depicting the time of the treatments of Cisplatin (C), electroacupuncture (E), and the analyses of body weight (W) and sample collection. (B) Representative peripheral blood flow cytometry analyses of neutrophils (LY6G + ), monocytes (LY6C + ), T (CD3 + ), and B (CD19 + ) lymphocytes and (C) Blood counts of specific subpopulation of leukocytes of mice with control (Veh), cisplatin alone (Cis; 3 mg/kg), or with electroacupuncture (EA) treatment (leukocytes, lymphocytes: n =6 per group; neutrophils, monocytes, T and B lymphocytes: Veh, n =6; Cis, n =6; EA, n =7). (D) Mice body weight curves treatment at day 0, 3, 7, 10, 14 ( n =6 per group), P values were calculated using two-way repeated-measures ANOVA. Data are mean ± SEM, * P < 0.05, ** P < 0.01, *** P < 0.001 vs Veh; # P < 0.05, ## P < 0.01 vs Cis.
Index in PubMed under a CC BY license. PMID: 34552585
Click image to see more details
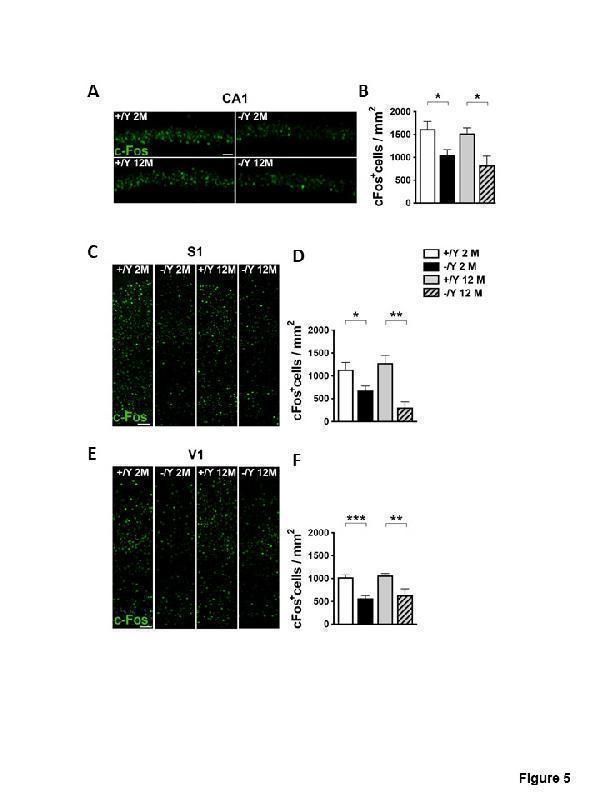
Cdkl5 KO mice exhibit a reduction of the number of c-fos. (A, C, E) Representative examples of c-Fos staining obtained from the pyramidal cell layer of the hippocampal CA1 (A), and from the primary somatosensory S1 (C) and primary visual cortices V1 (E) of young adult and middle-aged Cdkl5 +/Y and Cdkl5 -/Y mice. Scale bar = 50μm. (B) Quantification of the density of c-Fos immunoreactive cells in the CA1 layer from young adult Cdkl5 +/Y (n = 6) and Cdkl5 -/Y (n = 7) mice, and middle-aged Cdkl5 +/Y (n = 3) and Cdkl5 -/Y (n = 4) mice. (D) Quantitative analysis of the density of c-Fos positive cells in the S1 cortex of young adult Cdkl5 +/Y (n = 8) and Cdkl5 -/Y (n = 10) mice, and middle-aged Cdkl5 +/Y (n = 4) and Cdkl5 - /Y (n = 3) mice. (F) Quantification of c-Fos positive cells in the V1 cortex of young adult Cdkl5 +/Y (n = 5) and Cdkl5 -/Y (n = 5) mice, and middle-aged Cdkl5 +/Y (n = 3) and Cdkl5 -/Y (n = 3) mice. Values are represented as means ± SE. *p<0.05, **p<0.01, ***p<0.001 (Fisher’s LSD test after two-way ANOVA).
Index in PubMed under a CC BY license. PMID: 34094641
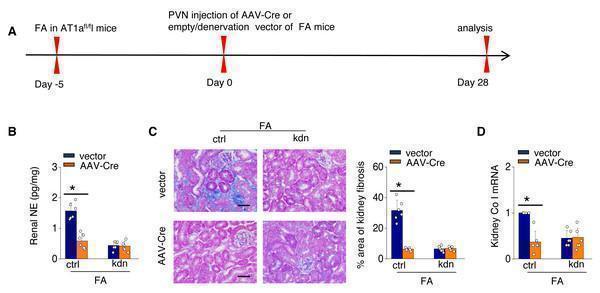
Click image to see more details
Localized Ang II in the PVN triggers fibrosis in the FA-CKD model via SNS activation. Outline of experimental design: PVN-specific deletion of Ang II type 1a receptor (AT1a) was achieved by injecting AAV-Cre into PVN of AT1a fl/fl mice. Blockade of sympathetic outflow was achieved by denervation of the post-obstructed kidney (kdn). (B) Kidney norepinephrine (NE) level in homogenates of the FA-CKD mice. (C) Kidney fibrosis was visualized by Masson trichrome staining in FA-CKD mice: representative images (left) and quantitative data (right). Scale bar, 50 um. (D) Level of collagen I (Co I) mRNA in kidney homogenates of the FA-CKD mice. Error bars, mean ± standard deviation ( n = 6 in each group). *, p < 0.01. One-way analysis of variance or t test with Bonferroni correction. Download full-size image DOI:
Index in PubMed under a CC BY license. PMID: 39346076
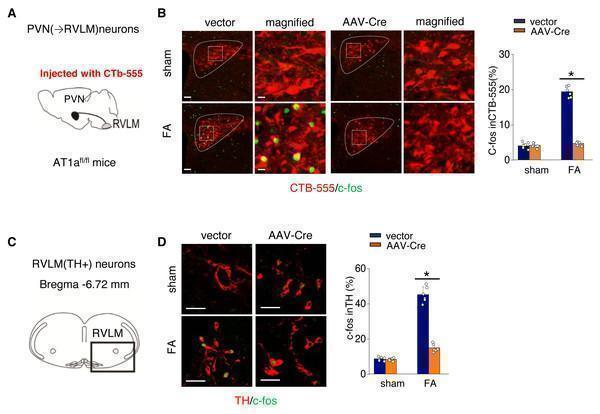
Click image to see more details
Ang II signaling in PVN activates the PVN-RVLM pathway that enhances SNS discharge to the kidney. (A) Schematic showing the injection of a retrograde tracer, CTb-555, into the RVLM and retrograde labeling of PVN (→RVLM) neurons. The source for the component is from . (B) Expression of c-fos was detected in CTb-555 PVN neurons: representative images and summary of percentage of c-fos+ cell in CTb-555+ cells. Scale bar, 50 um. *, p < 0.01. (C) Schematic of the coronal brain section indicating the RVLM. (D) Immunostaining of c-fos and tyrosine hydroxylase (TH) in RVLM: representative images (left) and summary of percentage of c-fos+ cell in TH+ cells(right). Scale bar, 50 um. *, p < 0.01. Download full-size image DOI:
Index in PubMed under a CC BY license. PMID: 39346076
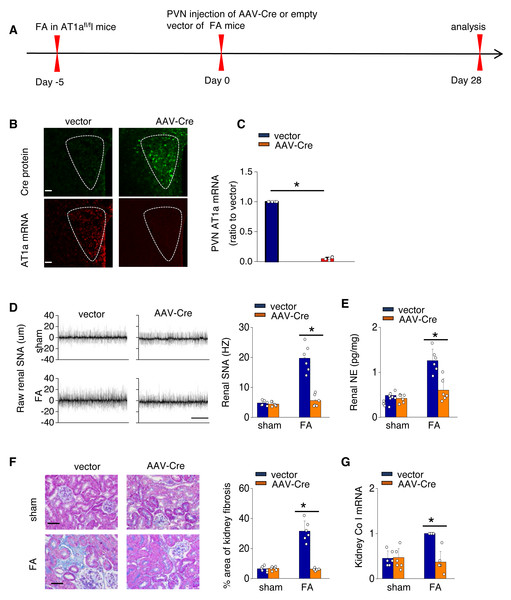
Click image to see more details
Activated Ang II signaling in PVN enhances SNS discharge that promotes fibrosis in the FA-CKD mice. (A) Outline of experimental design: PVN-specific deletion of Ang II type 1a receptor (AT1a) was achieved by injecting AAV-Cre into PVN of AT1a fl/fl mice. (B) Immunostaining of Cre protein and in situ hybridization of AT1a mRNA in PVN. Scale bar, 50 um. (C) Level of AT1a mRNA in PVN homogenates. (D) Sympathetic nerve activity (SNA) in the kidney: representative raw records (left) and quantitative data (right). Scale bar, 1 s. (E) Kidney norepinephrine (NE) level in homogenates of the FA-CKD mice. (F) Kidney fibrosis was visualized by Masson trichrome staining in FA-CKD mice: representative images (left) and quantitative data (right). Scale bar, 50 um. (G) Level of collagen I (Co I) mRNA in kidney homogenates of the FA-CKD mice. Error bars, mean G standard deviation ( n = 6 in each group). *, p < 0.01. One-way analysis of variance or t test with Bonferroni correction. Download full-size image DOI:
Index in PubMed under a CC BY license. PMID: 39346076
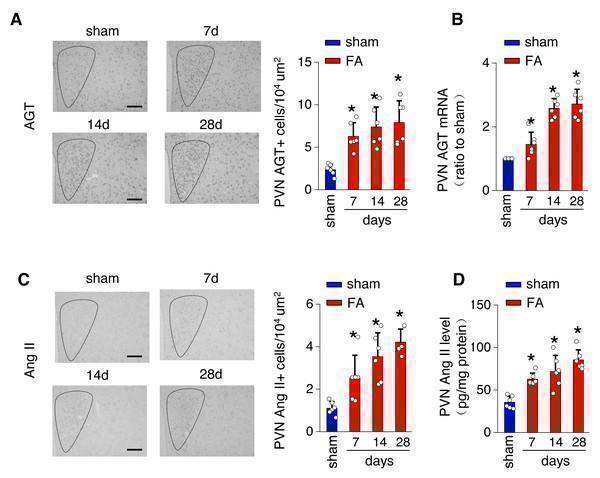
Click image to see more details
Folic acid-induced kidney injury in mice is accompanied by activity of the local renin-angiotensin system in the PVN. (A) Immunostaining for angiotensinogen (AGT) in paraventricular nucleus (PVN, Bregma −0.94 mm): representative images (left) and quantitative data (right). Scale bar, 100 um. (B) Level of AGT mRNA in PVN homogenates. (C) Immunostaining for angiotensin II (Ang II) in PVN (Bregma −0.94 mm): representative images (left) and quantitative data (right). Scale bar, 50 um (D) Concentration of Ang II in PVN homogenates. Download full-size image DOI:
Index in PubMed under a CC BY license. PMID: 39346076
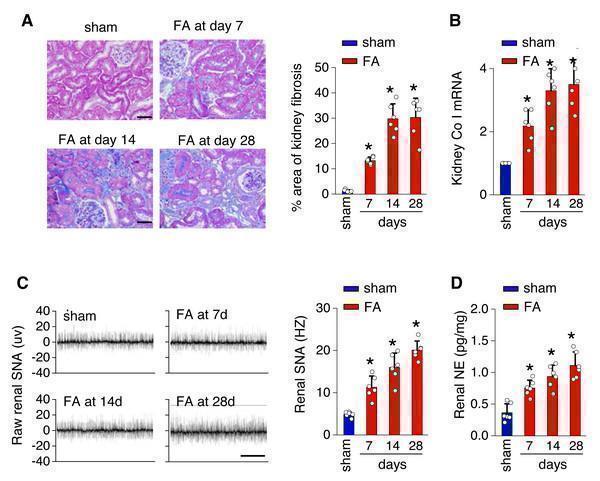
Click image to see more details
Kidney fibrosis in folic acid-induced kidney is accompanied by enhanced sympathetic nervous system (SNS) discharge to the kidney. (A) Kidney fibrosis was visualized by Masson trichrome staining in folic acid-induced kidney injury from mice at various time points: representative images (left) and quantitative data (right). Scale bar, 50 um. (B) Level of collagen I (Co I) mRNA in kidney homogenates of the folic acid-induced kidney injury mice. (C) Sympathetic nerve activity (SNA) in the folic acid-induced kidney: representative raw records (left) and quantitative data (right). Scale bar, 2s. (D) Levels of norepinephrine (NE) in kidney homogenates of the folic acid-induced kidney. *, p < 0.01 versus sham. Error bars, mean ± standard deviation ( n = 6 in each group). One-way analysis of variance or t test with Bonferroni correction. Download full-size image DOI:
Index in PubMed under a CC BY license. PMID: 39346076
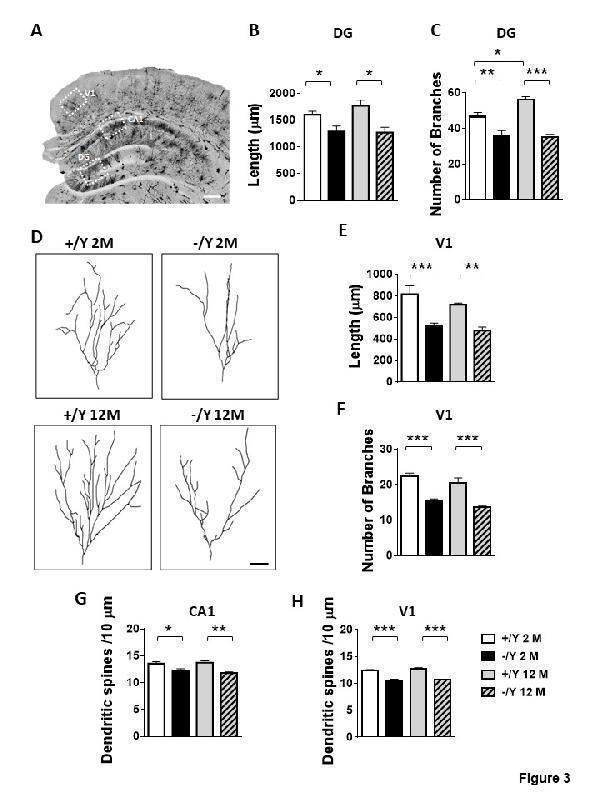
Click image to see more details
Impaired dendritic morphology in Cdkl5 KO mice. (A) Example of a Golgi-stained brain slice. The dotted boxes highlight the brain regions of the hippocampal formation (granular layer (DG) and hippocampal CA1 region (CA1)) and the visual cortex (V1) where dendritic length and spine density and maturation were evaluated. Scale bar = 500 μm. (B-C) Mean total dendritic length (B) and mean number of dendritic segments (C) of Golgi-stained granule cells of the dentate gyrus (DG) in adult Cdkl5 -/Y (n = 3) and Cdkl5 +/Y (n = 3) mice, and middle-aged Cdkl5 -/Y (n = 3) and Cdkl5 +/Y (n = 3) mice. (D) Examples of the reconstructed dendritic tree of Golgi-stained mature granule neurons of one animal from each experimental group. Scale bar = 100 μm. (E-F) Mean total dendritic length (E) and mean number of basal dendritic segments (F) of Golgi-stained pyramidal neurons of the primary visual cortex (V1, layer II/III) in adult Cdkl5 -/Y (n = 4) and Cdkl5 +/Y (n = 4) mice, and middle-aged Cdkl5 -/Y (n = 4) and Cdkl5 +/Y (n = 4) mice. (G-H) Dendritic spine density (number of spines per 10 μm) on apical dendrites of pyramidal neurons of the CA1 layer of the hippocampus (G) and pyramidal neurons of V1 (layer II/III; H) from adult Cdkl5 -/Y (n = 4 or 5, respectively) and Cdkl5 +/Y (n = 4) mice, and middle-aged Cdkl5 -/Y (n = 4) and Cdkl5 +/Y (n = 4) mice. Values are represented as means ± SE. *p< 0.05, **p< 0.01, ***p< 0.001 (Fisher’s LSD after two-way ANOVA).
Index in PubMed under a CC BY license. PMID: 34094641
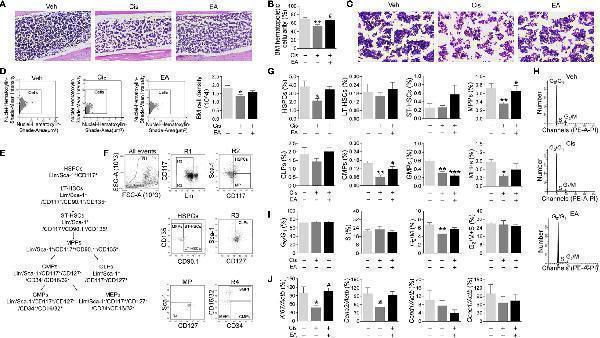
Click image to see more details
Electroacupuncture preserved hematopoiesis in mice with cisplatin chemotherapy. (A) Representative H&E staining of tibia BM from mice with control (Veh), cisplatin alone (Cis; 3 mg/kg), or with electroacupuncture (EA) treatment (scale bar=20.0 μm) and (B) Histogram representation of BM hematopoietic cellularity of H&E staining analyzed by Image-Pro Plus 6.0 software ( n= 4 per group). (C) Representative H&E staining of tibia BM from mice with Veh, cisplatin alone or with EA treatment at high configuration (scale bar=10.0 μm). (D) Representative HistoFAXS Tissue Analysis of BM cell nuclei hematoxylin-shade-mean intensity, and quantitative analysis of BM cell density ( n =4 per group). (E) Flowchart of hematopoiesis and hematopoietic cells markers. (F) Representative flow cytometry analyses and (G) quantification of hematopoietic BM cell subpopulations (Positive cells events (%) = (the events in target gate/the total cell) × 100) ( n =6 per group). (H) Representative PI nuclear staining flow cytometry analyses in BM cell cycle (G 0 /G 1 , S, G 2 /M phases) and (I) Quantification of PI nuclear staining of BM cells in G 0 /G 1 , S, G 2 /M phases by ModFit 3.1 software ( n =6 per group). (J) Expression of cell cycle related genes in BM cells ( Ki67 : Veh, n =7; Cis, n =5; EA, n =7. Ccna2 : n =7 per group. Ccnd1 : Veh, n =5; Cis, n =4; EA, n =6. Ccne1 : Veh, n =6; Cis, n =4; EA, n =5). Data are mean ± SEM, * P < 0.05, ** P < 0.01, *** P < 0.001 vs Veh; # P < 0.05 vs Cis.
Index in PubMed under a CC BY license. PMID: 34552585
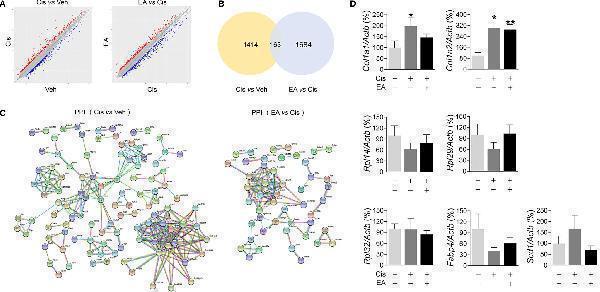
Click image to see more details
Analyses of expression and enrichment of electroacupuncture in the bone marrow of mice with cisplatin. (A) Scatter plots of differentially expressed genes (DEG)s in Cis vs Veh and EA vs Cis group Each probe is represented by a point with red and blue points showing up- and down-regulated genes defined above Log2 FC > 2. (B) Venn diagram and (C) PPI network analyses of DEGs results. (D) RT-qPCR analyses of factors related to extracellular matrix ( Col1a1 , Col1a2 ), ribosome ( Rpl14 , Rpl29 , Rpl32) , and PPAR signaling ( Fabp4 , Scd1) ( Col1a1 , Col1a2 : n =7 per group. Rpl14 , Rpl29 , Scd1 : n =6 per group. Rpl32 , Fabp4 : Veh, n =6; Cis, n =5; EA, n =6). Data are mean ± SEM, * P < 0.05, ** P < 0.01 vs Veh.
Index in PubMed under a CC BY license. PMID: 34552585
Click image to see more details
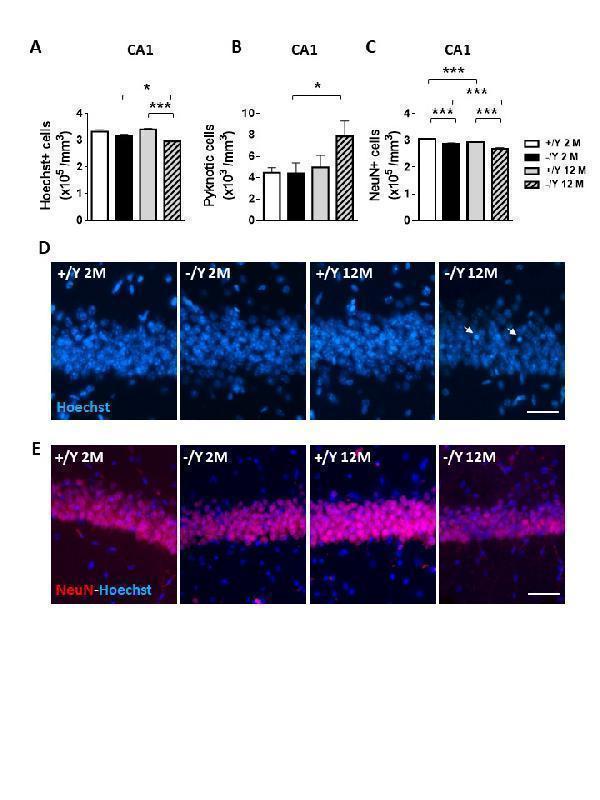
Reduced neuronal survival in Cdkl5 KO mice. (A) Quantification of Hoechst-positive cells in CA1 of hippocampal sections from young adult Cdkl5 +/Y (n = 4) and Cdkl5 -/Y (n = 4) mice, and middleaged Cdkl5 +/Y (n = 4) and Cdkl5 -/Y (n = 4) mice. (B) Quantification of pyknotic cells in the CA1 layer of the hippocampus from mice as in B. (C) Quantification of NeuN-positive cells in CA1 of hippocampal sections from young adult Cdkl5 +/Y (n = 4) and Cdkl5 -/Y (n = 4) mice, and middle-aged Cdkl5 +/Y (n = 5) and Cdkl5 -/Y (n = 4) mice. (D) Representative fluorescence images of cell layers in the hippocampal CA1 region from young adult and middle-aged Cdkl5 +/Y and Cdkl5 -/Y mice. Arrows indicate pyknotic nuclei. Scale bar = 100 μm. E) Representative fluorescence images of sections immunopositive for NeuN (red) and counterstained with Hoechst (blue) in the hippocampal CA1 region of one animal from each group. Scale bar = 100 μm. Values are represented as means ± SE. *p<0.05, **p<0.01, ***p<0.001 (Fisher’s LSD test after two-way ANOVA).
Index in PubMed under a CC BY license. PMID: 34094641
Click image to see more details
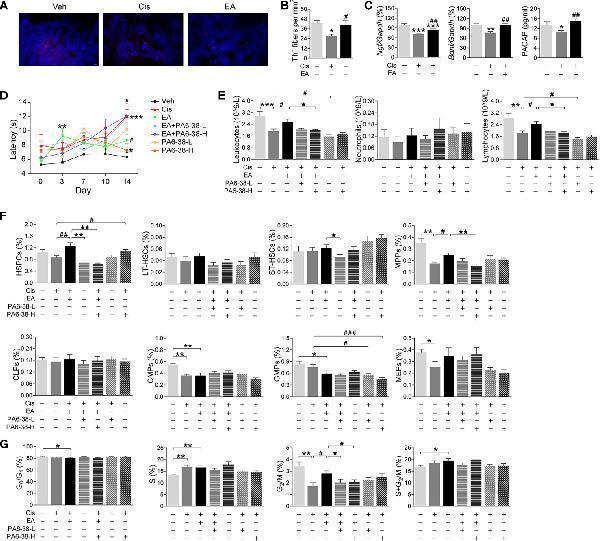
Neurogenic PACAP mediated electroacupuncture-induced protection to cisplatin. (A) Representative immunofluorescence images (Scale bar=20.0 μm) and (B) Quantification of sympathetic Th + fibers (red) and nuclear (blue) in the BM of the experimental mice ( n= 4 per group). (C) Expression analyses of neurotrophic factors ( Ngf , Bndf : Veh, n =7; Cis, n =6; EA, n =7. PACAP: Veh, n =5; Cis, n =6; EA, n =6). (D) Representation of the latency time (seconds) in hot-plate tests of mice treated with control (Veh), cisplatin (Cis; 3mg/kg), and cisplatin + electroacupuncture (EA) without or with PACAP6-38 (a blocker for PACAP receptor, PAC1) at low (10 μg/kg) or high (100 μg/kg) concentrations (Cis, n =7; other groups, n =8), P values were calculated using two-way repeated-measures ANOVA. (E) Peripheral blood counts of specific subpopulation of leukocytes (Veh, Cis, EA: n =6; other groups, n =7). (F) Analyses of hematopoietic BM subpopulation cells (Veh, Cis, EA, EA+PA6-38-L, EA+PA6-38-H: n =7; PA6-38-L, n =8, PA6-38-H, n =6). (G) Quantification of PI nuclear staining of BM cells (Veh, n =8; Cis, n =7; EA, n =8; EA+PA6-38-L, n =8; EA+PA6-38-H, n =7; PA6-38-L, n =8; PA6-38-H, n =7). Data are mean ± SEM * P < 0.05, ** P < 0.01, *** P < 0.001 vs Veh; # P < 0.05, ## P < 0.01, ### P < 0.001 vs Cis; ⋆ P < 0.05, ⋆⋆ P < 0.01 vs EA.
Index in PubMed under a CC BY license. PMID: 34552585
Click image to see more details
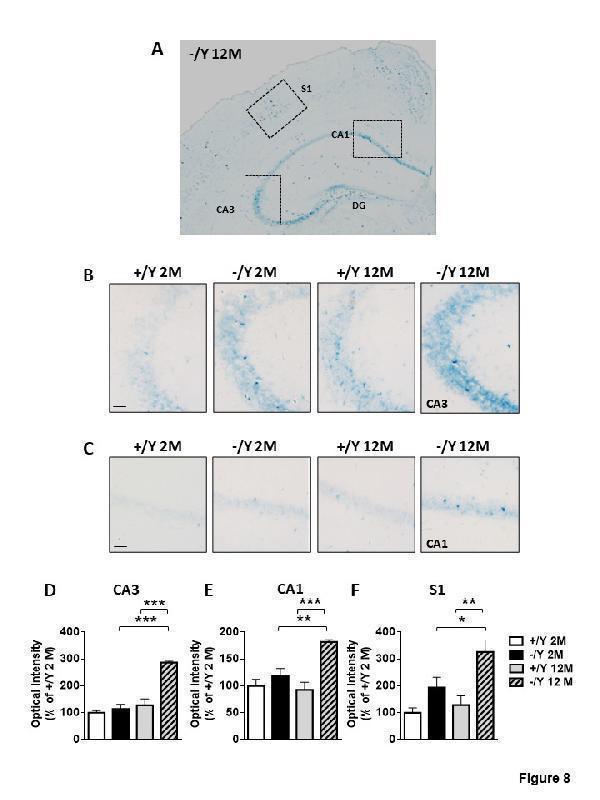
Senescence-associated-β-galactosidase (SA-β-gal) activity in Cdkl5 KO mice. (A) Representative image at low magnification of a hippocampus of a middle-aged Cdkl5 -/Y mouse. Scale bar = 500 μm. The dotted boxes indicate the regions used for quantification in (D-F). (B-C) Higher magnification shows representative images of SA-β-gal staining in the hippocampal CA3 (B) and CA1 (C) region from adult and middle-aged Cdkl5 +/Y and Cdkl5 -/Y mice. Scale bar = 50 μm. (D-F) Quantification of SA-β-gal intensity in CA3 (D) and CA1 (E) hippocampal layers, and in layer II/III of the S1 cortex (F) from young adult Cdkl5 +/Y (n = 3) and Cdkl5 -/Y (n = 4) mice, and middle-aged Cdkl5 +/Y (n = 4) and Cdkl5 -/Y (n = 3) mice. Values are represented as means ± SE. *p< 0.05, **p< 0.01, ***p< 0.001 (Fisher’s LSD test after two-way ANOVA).
Index in PubMed under a CC BY license. PMID: 34094641
Click image to see more details
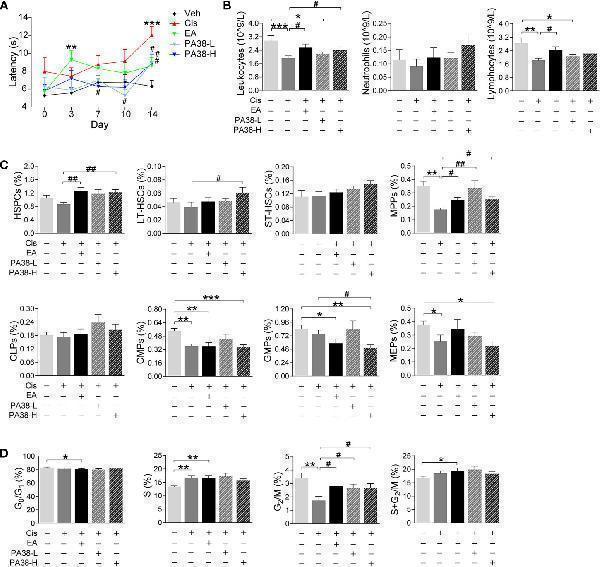
PAC1-agonist mimics electroacupuncture-induced protection to cisplatin. (A) Representation of the latency time (seconds) in hot-plate tests of mice with control (Veh), cisplatin (Cis; 3 mg/kg), EA (cisplatin + electroacupuncture), cisplatin mice were treated with low (10 μg/kg) or high (50 μg/kg) concentrations PAC1-agonist, PACAP1-38 (Veh, n =8; Cis, n =7; EA, n =8; PA38-L, n =8; PA38-H, n =7), P values were calculated using two-way repeated-measures ANOVA. (B) Peripheral blood counts of specific subpopulation of leukocytes ((Veh, Cis, EA: n =6; other groups, n =7). (C) Analyses of hematopoietic BM cell subpopulation (Veh, Cis, EA: n =7; PA38-L, PA38-H: n =8). (D) Quantification of PI nuclear staining of BM cells (Veh, n =8; Cis, n =7; EA, n =8; PA38-L, n =8; PA38-H, n =7). Data are mean ± SEM, * P < 0.05, ** P < 0.01, *** P < 0.001 vs Veh; # P < 0.05, ## P < 0.01 vs Cis.
Index in PubMed under a CC BY license. PMID: 34552585
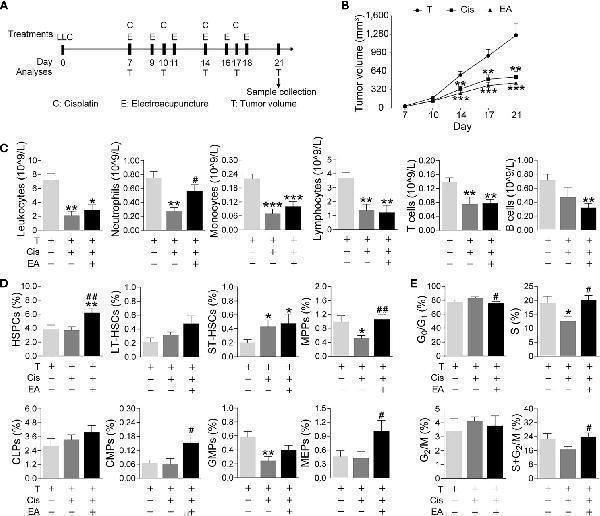
Click image to see more details
Electroacupuncture restores hematopoiesis in cancer mice during cisplatin chemotherapy. (A) Experimental flowchart depicting the time of treatments of tumor (LLC) cells at day 0, cisplatin (C), electroacupuncture (E), and analyses of tumor volume (T) and sample collection. (B) Tumor growth curve ( n =9 per group), P values were calculated using two-way repeated-measures ANOVA. (C) Peripheral blood counts of specific subpopulation of leukocytes (leukocytes, lymphocytes: T, n =8; Cis, n =6; EA, n =8. neutrophils, monocytes, T and B lymphocytes: T, n =8; Cis, n =6; EA, n =7). (D) Analyses of hematopoietic BM cell subpopulation ( n =9 per group). (E) Quantification of PI nuclear staining of BM cells ( n =9 per group). Data are mean ± SEM, * P < 0.05, ** P < 0.01, *** P < 0.001 vs Veh; # P < 0.05, ## P < 0.01 vs Cis.
Index in PubMed under a CC BY license. PMID: 34552585
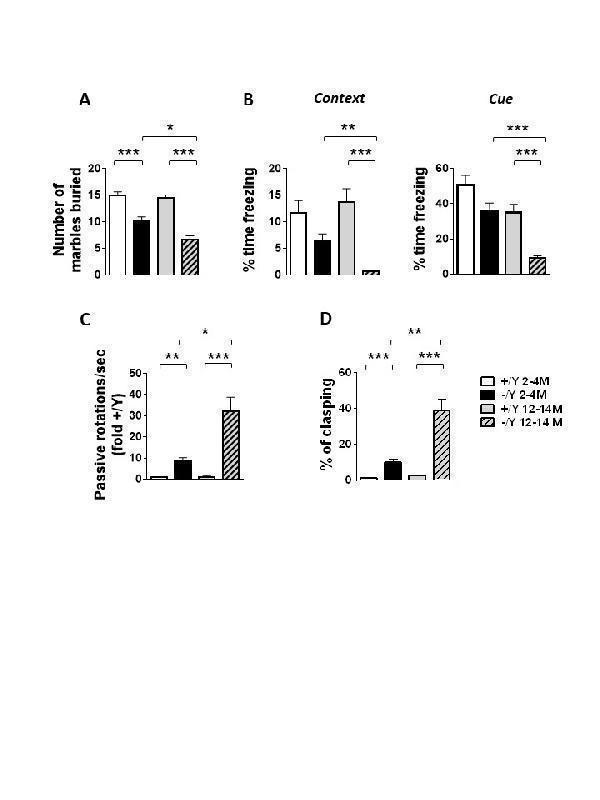
Click image to see more details
Age-dependent deterioration of behavior in Cdkl5 KO mice. (A) Marble burying test in young adult Cdkl5 -/Y (n = 38) and Cdkl5 +/Y (n = 31) mice, and middle-aged Cdkl5 -/Y (n = 29) and Cdkl5 +/Y (n = 22) mice. (B) Fear conditioning paradigm, measuring time spent freezing in response to a mild footshock, upon the return to the testing chamber (context) and upon hearing the testing tone (cue) in adult Cdkl5 -/Y (n = 29) and Cdkl5 +/Y (n = 28) mice, and middle-aged Cdkl5 -/Y (n = 22) and Cdkl5 +/Y (n = 20) mice. (C) Frequency of passive rotations (rotations in which the does not perform any coordinated movement but is passively transported on the rotating apparatus) in adult Cdkl5 -/Y (n = 30) and Cdkl5 +/Y (n = 19) mice, and middle-aged Cdkl5 -/Y (n = 13) and Cdkl5 +/Y (n = 10) mice. The mean frequency of passive rotations was expressed as fold of the wild-type counterparts of the same age. (D) Percentage of time spent hind-limb clasping during a 2-min interval in adult Cdkl5 -/Y (n = 29) and Cdkl5 +/Y (n =19) mice, and middle-aged Cdkl5 -/Y (n = 27) and Cdkl5 +/Y (n = 19) mice. Values represent mean ± SEM. *p< 0.05, **p< 0.01, ***p< 0.001 (Dunn’s test after Kruskal-Wallis).
Index in PubMed under a CC BY license. PMID: 34094641
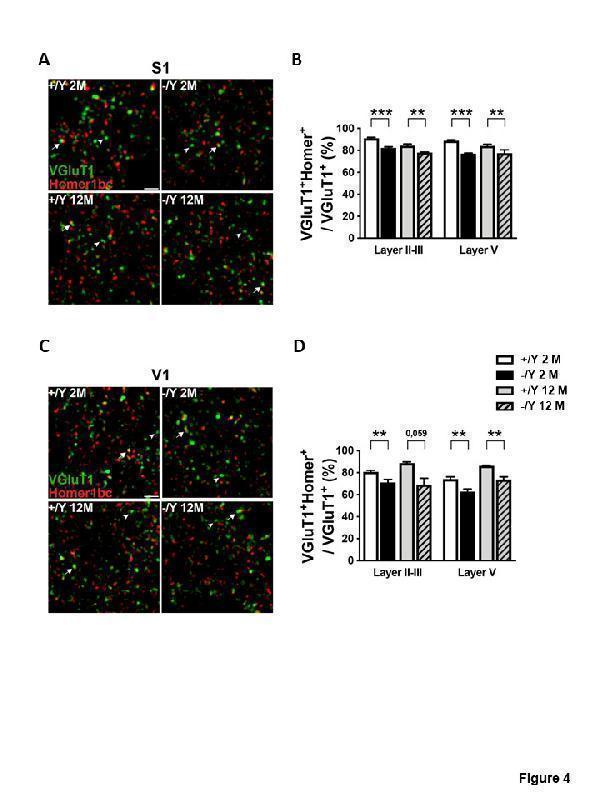
Click image to see more details
Cdkl5 KO mice exhibit a decrease in the number of excitatory synaptic contacts. (A, C) Representative confocal micrographs of the neuropil in layer II-III of both S1 (A) and V1 (C) cortices showing immunofluorescence staining for VGluT1 (green) and Homer1bc (red) in young adult and middleaged Cdkl5 +/Y and Cdkl5 -/Y mice. Scale bar 5 μm. Arrows point to examples of VGluT1-Homer1bc coappositions. Arrowheads point to examples of solitary VGluT1+ terminals. (B, D) Quantitative analysis of the percentage of VGluT1+ terminals juxtaposed to Homer1bc+ in cerebral cortex from young adult Cdkl5 -/Y (n = 4) and Cdkl5 +/Y (n = 5) mice, and middle-aged Cdkl5 -/Y (n = 3) and Cdkl5 +/Y (n = 4) mice. Values are represented as means ± SE. **p<0.01, ***p<0.001 (Fisher’s LSD test after two-way ANOVA
Index in PubMed under a CC BY license. PMID: 34094641
Specific Publications For Anti-Tyrosine Hydroxylase/TH Antibody Picoband® (PB9449)
Loading publications
Recommended Resources
Here are featured tools and databases that you might find useful.
- Boster's Pathways Library
- Protein Databases
- Bioscience Research Protocol Resources
- Data Processing & Analysis Software
- Photo Editing Software
- Scientific Literature Resources
- Research Paper Management Tools
- Molecular Biology Software
- Primer Design Tools
- Bioinformatics Tools
- Phylogenetic Tree Analysis
Customer Reviews
Have you used Anti-Tyrosine Hydroxylase/TH Antibody Picoband®?
Share your experimental results or join a short interview to earn up to $1,000 in product credits or other rewards.
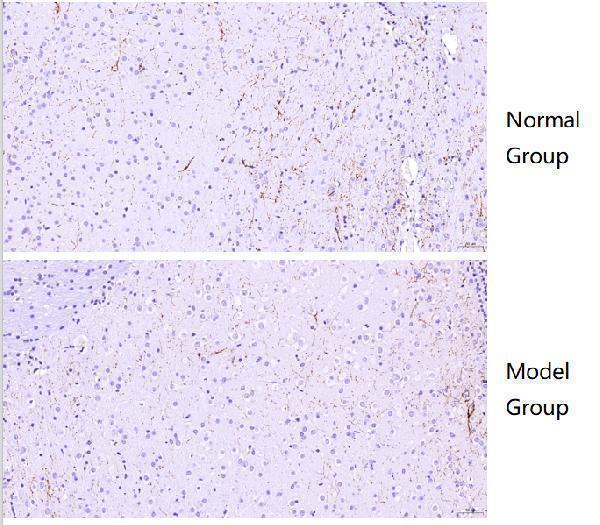
1 Reviews For Anti-Tyrosine Hydroxylase/TH Antibody Picoband®
IHC using Tyrosine Hydroxylase/TH Antibody Picoband® (P00683) showed clear staining with no background, revealing markedly reduced TH expression in the medulla of Alzheimer’s mouse brain compared to normal mouse brain.
Excellent
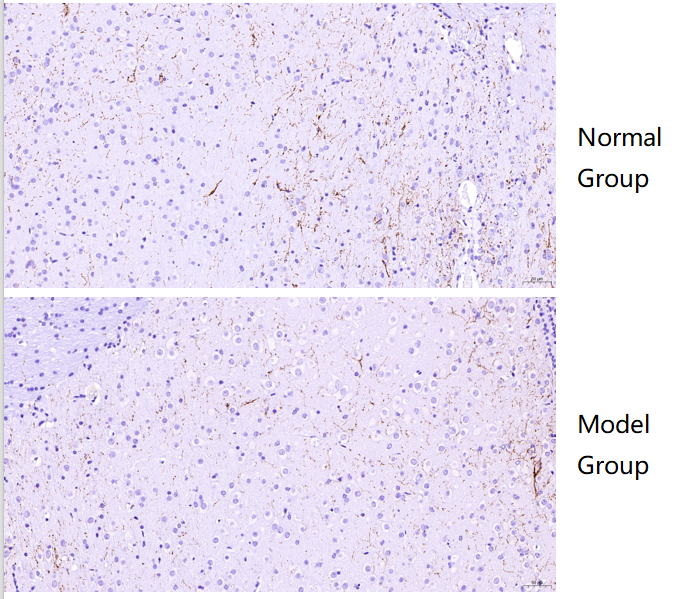
| SKU | PB9449 |
|---|---|
| Application | Immunohistochemistry |
| Sample | Normal mouse brain and Alzheimer’s model mouse brain tissue |
| Sample Processing Description | Paraffin-embedded normal mouse brain and Alzheimer’s model mouse brain. |
| Other Reagents | Goat serum, DAB |
| Primary Antibody | Tyrosine Hydroxylase/TH Antibody Picoband® |
| Primary Incubation | 1:500, overnight at 4 ℃ |
| Secondary Antibody | Two-step IHC kit |
| Secondary Incubation | 37 minutes in 37 ℃ |
| Detection | Imaging system:Leica DM2500 |
| Results Summary | TH is a marker of catecholaminergic neurons, specifically labeling dopaminergic, noradrenergic, and adrenergic neurons. IHC results showed that TH expression in the medulla was markedly lower in Alzheimer’s mouse compared to normal mouse. |
Song Qian, Zhejiang Police College
Verified customer
Submitted 2026-01-26
Customer Q&As
Have a question?
Find answers in Q&As, reviews.
Can't find your answer?
Submit your question
14 Customer Q&As for Anti-Tyrosine Hydroxylase/TH Antibody Picoband®
Question
Is there a BSA free version of anti-Tyrosine Hydroxylase/TH antibody PB9449 available?
Verified Customer
Verified customer
Asked: 2020-04-03
Answer
I appreciate your recent telephone inquiry. I can confirm that some lots of this anti-Tyrosine Hydroxylase/TH antibody PB9449 are BSA free. For now, these lots are available and we can make a BSA free formula for you free of charge. It will take 3 extra days to prepare. If you require this antibody BSA free again in future, please do not hesitate to contact me and I will be pleased to check which lots we have in stock that are BSA free.
Boster Scientific Support
Answered: 2020-04-03
Question
We are currently using anti-Tyrosine Hydroxylase/TH antibody PB9449 for rat tissue, and we are content with the WB results. The species of reactivity given in the datasheet says mouse, rat. Is it possible that the antibody can work on goat tissues as well?
Verified Customer
Verified customer
Asked: 2020-02-28
Answer
The anti-Tyrosine Hydroxylase/TH antibody (PB9449) has not been validated for cross reactivity specifically with goat tissues, though there is a good chance of cross reactivity. We have an innovator award program that if you test this antibody and show it works in goat you can get your next antibody for free. Please contact me if I can help you with anything.
Boster Scientific Support
Answered: 2020-02-28
Question
We want using your anti-Tyrosine Hydroxylase/TH antibody for heart development studies. Has this antibody been tested with western blotting on tissue lysate? We would like to see some validation images before ordering.
Verified Customer
Verified customer
Asked: 2019-11-11
Answer
I appreciate your inquiry. This PB9449 anti-Tyrosine Hydroxylase/TH antibody is validated on rat brain tissue, tissue lysate, mouse brain. It is guaranteed to work for IHC, WB in mouse, rat. Our Boster guarantee will cover your intended experiment even if the sample type has not been be directly tested.
Boster Scientific Support
Answered: 2019-11-11
Question
Is PB9449 suitable for immunostaining of mouse long bone nerve fibers?
Verified customer
Asked: 2019-10-06
Answer
Yes, Anti-Tyrosine Hydroxylase/TH Antibody Picoband™ (PB9449) is suitable for the immunostaining of mouse long bone nerve fibers.
Boster Scientific Support
Answered: 2019-10-09
Question
I was wanting to use your anti-Tyrosine Hydroxylase/TH antibody for WB for rat substantia nigra on frozen tissues, but I want to know if it has been tested for this particular application. Has this antibody been tested and is this antibody a good choice for rat substantia nigra identification?
Verified Customer
Verified customer
Asked: 2019-09-05
Answer
It shows on the product datasheet, PB9449 anti-Tyrosine Hydroxylase/TH antibody has been tested for IHC, WB on mouse, rat tissues. We have an innovator award program that if you test this antibody and show it works in rat substantia nigra in IHC-frozen, you can get your next antibody for free.
Boster Scientific Support
Answered: 2019-09-05
Question
Thank you for helping with my inquiry over the phone. Here are the WB image, lot number and protocol we used for substantia nigra using anti-Tyrosine Hydroxylase/TH antibody PB9449. Let me know if you need anything else.
Verified Customer
Verified customer
Asked: 2019-07-25
Answer
We appreciate the data. You have provided everything we needed. Our lab team are working to resolve your inquiry as quickly as possible, and we appreciate your patience and understanding! Please let me know if there is anything you need in the meantime.
Boster Scientific Support
Answered: 2019-07-25
Question
Is a blocking peptide available for product anti-Tyrosine Hydroxylase/TH antibody (PB9449)?
Verified Customer
Verified customer
Asked: 2019-06-24
Answer
We do provide the blocking peptide for product anti-Tyrosine Hydroxylase/TH antibody (PB9449). If you would like to place an order for it please contact support@bosterbio.com and make a special request.
Boster Scientific Support
Answered: 2019-06-24
Question
Does PB9449 anti-Tyrosine Hydroxylase/TH antibody work on parafin embedded sections? If so, which fixation method do you recommend we use (PFA, paraformaldehyde, other)?
S. Moore
Verified customer
Asked: 2019-03-26
Answer
It shows on the product datasheet, PB9449 anti-Tyrosine Hydroxylase/TH antibody as been validated on WB. It is best to use PFA for fixation because it has better tissue penetration ability. PFA needs to be prepared fresh before use. Long term stored PFA turns into formalin, as the PFA molecules congregate and become formalin.
Boster Scientific Support
Answered: 2019-03-26
Question
Does anti-Tyrosine Hydroxylase/TH antibody PB9449 work for WB with substantia nigra?
Verified Customer
Verified customer
Asked: 2019-02-14
Answer
According to the expression profile of substantia nigra, TH is highly expressed in substantia nigra. So, it is likely that anti-Tyrosine Hydroxylase/TH antibody PB9449 will work for WB with substantia nigra.
Boster Scientific Support
Answered: 2019-02-14
Question
I have a question about product PB9449, anti-Tyrosine Hydroxylase/TH antibody. I was wondering if it would be possible to conjugate this antibody with biotin. I would need it to be without BSA or sodium azide. I am planning on using a buffer exchange of sodium azide with PBS only. Would there be problems for me to conjugate the antibody and store it in -20 degrees in small aliquots?
H. Baker
Verified customer
Asked: 2018-09-26
Answer
We do not advise storing this antibody with PBS buffer only in -20 degrees. If you want to store it in -20 degrees it is best to add some cryoprotectant like glycerol. If you want carrier free PB9449 anti-Tyrosine Hydroxylase/TH antibody, we can provide it to you in a special formula with trehalose and/or glycerol. These molecules will not interfere with conjugation chemistry and provide a good level of protection for the antibody from degradation. Please be sure to specify this in your purchase order.
Boster Scientific Support
Answered: 2018-09-26
Question
I see that the anti-Tyrosine Hydroxylase/TH antibody PB9449 works with WB, what is the protocol used to produce the result images on the product page?
Verified Customer
Verified customer
Asked: 2018-09-07
Answer
You can find protocols for WB on the "support/technical resources" section of our navigation menu. If you have any further questions, please send an email to support@bosterbio.com
Boster Scientific Support
Answered: 2018-09-07
Question
See below the WB image, lot number and protocol we used for substantia nigra using anti-Tyrosine Hydroxylase/TH antibody PB9449. Please let me know if you require anything else.
Verified Customer
Verified customer
Asked: 2018-06-28
Answer
Thank you very much for the data. Our lab team are working to resolve this as quickly as possible, and we appreciate your patience and understanding! You have provided everything we needed. Please let me know if there is anything you need in the meantime.
Boster Scientific Support
Answered: 2018-06-28
Question
Is this PB9449 anti-Tyrosine Hydroxylase/TH antibody reactive to the isotypes of TH?
Verified Customer
Verified customer
Asked: 2018-05-16
Answer
The immunogen of PB9449 anti-Tyrosine Hydroxylase/TH antibody is A synthetic peptide corresponding to a sequence in the middle region of human Tyrosine Hydroxylase (193-222aa KVPWFPRKVSELDKCHHLVTKFDPDLDLDH), identical to the related mouse and rat sequences. Could you tell me which isotype you are interested in so I can help see if the immunogen is part of this isotype?
Boster Scientific Support
Answered: 2018-05-16
Question
you antibody to test anti-Tyrosine Hydroxylase/TH antibody PB9449 on rat substantia nigra for research purposes, then I may be interested in using anti-Tyrosine Hydroxylase/TH antibody PB9449 for diagnostic purposes as well. Is the antibody suitable for diagnostic purposes?
T. Baker
Verified customer
Asked: 2015-10-27
Answer
The products we sell, including anti-Tyrosine Hydroxylase/TH antibody PB9449, are only intended for research use. They would not be suitable for use in diagnostic work. If you have the means to develop a product into diagnostic use, and are interested in collaborating with us and develop our product into an IVD product, please contact us for more discussions.
Boster Scientific Support
Answered: 2015-10-27





