This website uses cookies to ensure you get the best experience on our website.
- Table of Contents
7 Citations
1 Citations
Facts about Cytochrome P450 7A1.
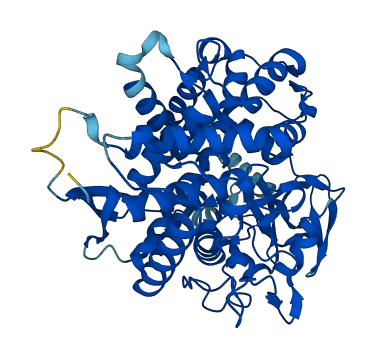
.
| Human | |
|---|---|
| Gene Name: | CYP7A1 |
| Uniprot: | P22680 |
| Entrez: | 1581 |

| Belongs to: |
|---|
| cytochrome P450 family |

Cholesterol 7-alpha-hydroxylase; cholesterol 7-alpha-monooxygenase; CP7A; CYP7; CYP7A1; CYP7MGC126826; CYPVII; Cytochrome P450 7A1; cytochrome P450, family 7, subfamily A, polypeptide 1; cytochrome P450, subfamily VIIA (cholesterol 7 alpha-monooxygenase); cytochrome P450, subfamily VIIA polypeptide 1; EC 1.14.13; EC 1.14.13.17; MGC138389; polypeptide 1
Mass (kDA):
57.661 kDA

| Human | |
|---|---|
| Location: | 8q12.1 |
| Sequence: | 8; NC_000008.11 (58490178..58500163, complement) |
Detected in liver.
Endoplasmic reticulum membrane; Single-pass membrane protein. Microsome membrane; Single-pass membrane protein.




PMID: 8439551 by Nishimoto M., et al. Structure of the gene encoding human liver cholesterol 7 alpha- hydroxylase.
PMID: 2384150 by Noshiro M., et al. Molecular cloning and sequence analysis of cDNA encoding human cholesterol 7 alpha-hydroxylase.
*More publications can be found for each product on its corresponding product page