This website uses cookies to ensure you get the best experience on our website.
- Table of Contents
21 Citations 10 Q&As
12 Citations 7 Q&As
11 Citations 10 Q&As
14 Citations 6 Q&As
3 Citations
Facts about Metalloproteinase inhibitor 1.
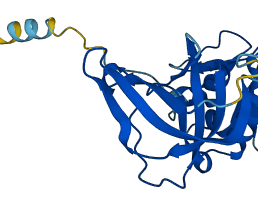
Does not act on MMP14. Also functions as a growth factor that regulates cell differentiation, migration and cell death and activates cellular signaling cascades through CD63 and ITGB1.
| Human | |
|---|---|
| Gene Name: | TIMP1 |
| Uniprot: | P01033 |
| Entrez: | 7076 |

| Belongs to: |
|---|
| protease inhibitor I35 (TIMP) family |

CLGI; Collagenase inhibitor; collagenase inhibitor); EPATIMP-1; EPO; erythroid potentiating activity; Erythroid-potentiating activity; Fibroblast collagenase inhibitor; FLJ90373; HCI; metalloproteinase inhibitor 1; TIMP metallopeptidase inhibitor 1; TIMP1; TIMP-1; TIMPtissue inhibitor of metalloproteinase 1 (erythroid potentiating activity; Tissue inhibitor of metalloproteinases 1
Mass (kDA):
23.171 kDA

| Human | |
|---|---|
| Location: | Xp11.3 |
| Sequence: | X; NC_000023.11 (47582408..47586789) |
Detected in rheumatoid synovial fluid (at protein level).
Secreted.





PMID: 3903517 by Docherty A.J.P., et al. Sequence of human tissue inhibitor of metalloproteinases and its identity to erythroid-potentiating activity.
PMID: 3839290 by Gasson J.C., et al. Molecular characterization and expression of the gene encoding human erythroid-potentiating activity.
*Showing only the more recent 20. More publications can be found for each product on its corresponding product page