Product Info Summary
| SKU: | PA2141-1 |
|---|---|
| Size: | 100 μg/vial |
| Reactive Species: | Mouse, Rat |
| Host: | Rabbit |
| Application: | IF, IHC, WB |
Customers Who Bought This Also Bought
Product info
Product Name
Anti-Collagen II/COL2A1 Antibody Picoband®
SKU/Catalog Number
PA2141-1
BA0533 is an alternative SKU for this antibody, used in previous lots.
Size
100 μg/vial
Form
Lyophilized
Description
Boster Bio Anti-Collagen II/COL2A1 Antibody catalog # PA2141-1. Tested in IHC, WB applications. This antibody reacts with Mouse, Rat. The brand Picoband indicates this is a premium antibody that guarantees superior quality, high affinity, and strong signals with minimal background in Western blot applications. Only our best-performing antibodies are designated as Picoband, ensuring unmatched performance.
Storage & Handling
Store at -20˚C for one year from date of receipt. After reconstitution, at 4˚C for one month. It can also be aliquotted and stored frozen at -20˚C for six months. Avoid repeated freeze-thaw cycles.
Cite This Product
Anti-Collagen II/COL2A1 Antibody Picoband® (Boster Biological Technology, Pleasanton CA, USA, Catalog # PA2141-1)
Host
Rabbit
Contents
Each vial contains antibody formulated with stabilizing components, 0.9mg NaCl, 0.2mg Na2HPO4, 0.05mg Thimerosal, 0.05mg NaN3.
*This antibody is supplied in a stabilized formulation.
Compatibility with conjugation reactions depends on the chemistry of the conjugation method used.
For conjugation methods that are not compatible with the stabilizing components present in this formulation, a carrier-free antibody format is required.
Clonality
Polyclonal
Isotype
Rabbit IgG
Immunogen
A synthetic peptide corresponding to a sequence at the C-terminus of mouse Collagen II1222-1241aa AFAGLGQREKGPDPMQYMRA), different from the related rat sequence by one amino acid.
Cross-reactivity
No cross-reactivity with other proteins
Reactive Species
PA2141-1 is reactive to Col2a1 in Mouse, Rat
Observed Molecular Weight
170 kDa
Calculated molecular weight
142.0 kDa
Background of Col2a1
Collagen, type II, alpha 1, also known as COL2A1, is a human gene which is the major collagen synthesized by chondrocvtes. COL2A1 is mapped to 12q13.11. This gene encodes the alpha-1 chain of type II collagen, a fibrillar collagen found in cartilage and the vitreous humor of the eye. Mutations in this gene are associated with achondrogenesis, chondrodysplasia, early onset familial osteoarthritis, SED congenita, Langer-Saldino achondrogenesis, Kniest dysplasia, Stickler syndrome type I, and spondyloepimetaphyseal dysplasia Strudwick type. In addition, defects in processing chondrocalcin, a calcium binding protein that is the C-propeptide of this collagen molecule, are also associated with chondrodysplasia.
Antibody Validation
Boster validates all antibodies on WB, IHC, ICC, Immunofluorescence, and ELISA with known positive control and negative samples to ensure specificity and high affinity, including thorough antibody incubations.
Application & Images
Applications
PA2141-1 is guaranteed for IF, IHC, WB Boster Guarantee
Recommend Dilution
| Application | Dilution | Species |
|---|---|---|
| Western Blot (WB) | 0.1-0.5μg/ml | Rat, Mouse |
| Immunohistochemistry (IHC) | 0.5-1μg/ml | Rat, Mouse |
Tested application
Suggested blocking solution with 5% non-fat milk or BSA; (*)Recommended protein loading: 20-40 µg per lane
Validation Images & Assay Conditions

Click image to see more details
Anti-Collagen II antibody, PA2141-1, IHC(F)
IHC(F): Rat Trachea Tissue

Click image to see more details
Anti-Collagen II antibody, PA2141-1, Western blotting
Lane 1: Rat Heart Tissue Lysate
Lane 1: Rat Brain Tissue Lysate
Click image to see more details
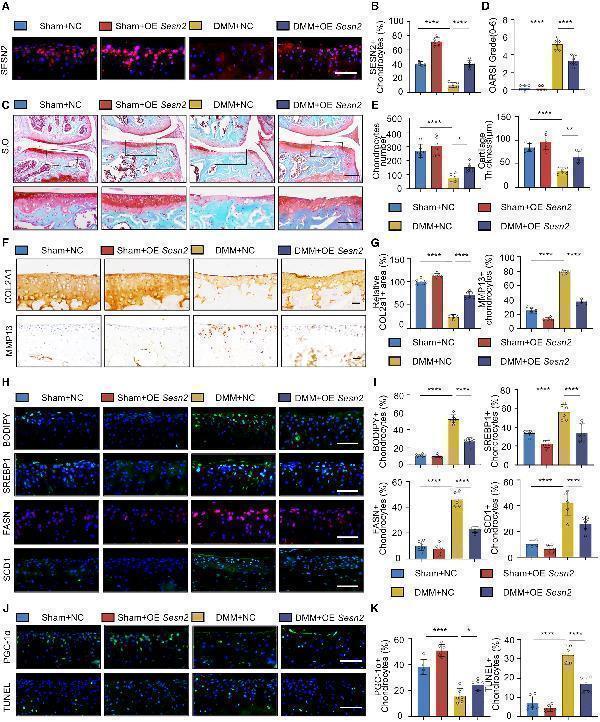
SESN2 overexpression improves fatty acid metabolism disorders and ameliorates cartilage degeneration
(A and B) IF (A) and corresponding quantitative analysis (B) of SESN2 in the articular cartilage of mice induced by sham or destabilization of medial meniscus (DMM) surgery with intraarticular injection of lv-nc and lv-Sesn2 (n = 6).
(C–E) S.O. staining (C) of mice treated as in (A) (n = 6). Quantitation of Osteoarthritis Research Society International (OARSI) scores (D), chondrocyte numbers, and cartilage thickness (E) (n = 6).
(F and G) IHC (F) and corresponding quantitative analysis (G) of COL2A1 and MMP13 in the articular cartilage of mice treated as in (A) (n = 6).
(H and I) BODIPY493/503 staining, IF (H) of SREBP1, FASN, and SCD1, and corresponding quantitative analysis (I) in the articular cartilage of mice treated as in (A) (n = 6).
(J and K) IF of PGC-1α and TUNEL staining (J) and corresponding quantitative analysis (K) in the articular cartilage of mice treated as in (A) (n = 6).
Scale bars: (F, H, and J) 50 μm, (C) 50 and 100 μm. Data (n = 6) are shown as mean ± SD. ∗p < 0.05; ∗∗p < 0.01; ∗∗∗p < 0.001; ∗∗∗∗p < 0.0001. Statistical analysis was performed by one-way ANOVA (B, D, E, G, I, and K).
Index in PubMed under a CC BY license. PMID: 40822351
Click image to see more details
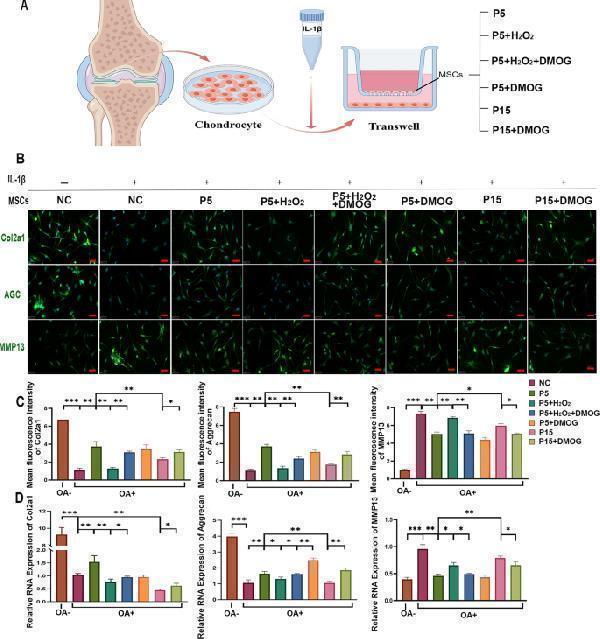
DMOG enhances the therapeutic potential of senescent MSCs to mitigate metabolic dysfunction in OA chondrocytes. ( A ) Schematic diagram of the co-culture system. Human articular chondrocytes were treated with IL-1β to induce OA-like conditions and co-cultured with MSCs from various experimental groups using a Transwell system. ( B ) Representative immunofluorescence staining of ECM-related proteins, including Collagen II (Col2a1), Aggrecan (AGC), and MMP13, in OA chondrocytes co-cultured with MSCs from different experimental groups. Scale bar = 50 μm. ( C ) Quantitative analysis of immunofluorescence intensity for Col2a1, AGC, and MMP13. ( D ) Quantitative real-time PCR (qRT-PCR) analysis of mRNA levels for Col2a1, AGC, and MMP13 in OA chondrocytes co-cultured with MSCs. Data are expressed as mean ± SEM ( n = 3). * p < 0.05, ** p < 0.01, *** p < 0.001 Full size image
Index in PubMed under a CC BY license. PMID: 40457488
Click image to see more details
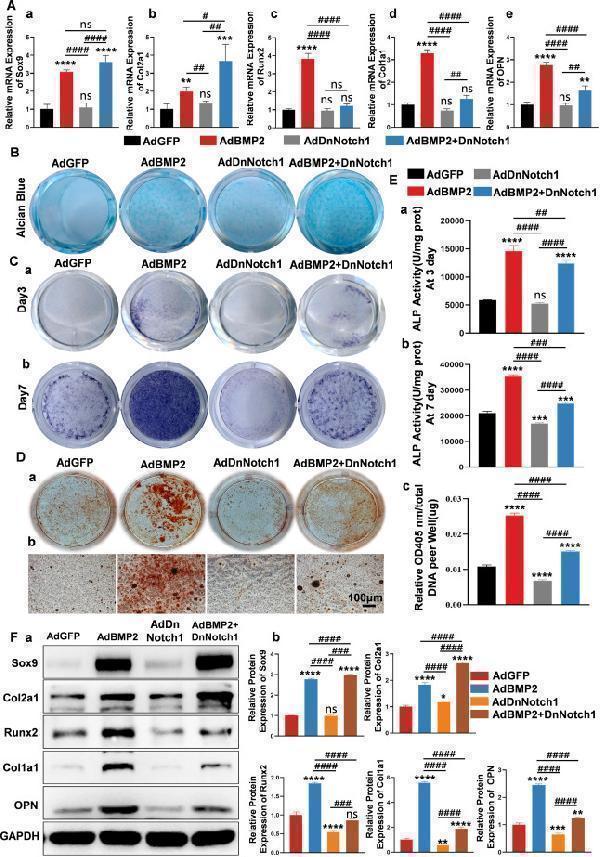
Down-regulation of Notch1 signaling promoted BMP2-induced chondrogenic differentiation and inhibited BMP2-induced osteogenic differentiation of MSCs in vitro. (A) C3H10T1/2 cells were transfected with AdGFP, AdBMP2, AdDnNotch1, and AdBMP2+DnNotch1, respectively. On day 7, quantitative reverse transcription PCR was used to detect the expression of chondrogenic differentiation markers (Sox9 and Col2a1) and osteoblastic differentiation markers (Runx2, Col1a1, and OPN) of MSCs. (B) To detect the expression of sulfated glycosaminoglycan during C3H10T1/2 cell differentiation, alcian blue staining was performed on day 7 after cells were transfected with recombinant adenovirus. (C) Alkaline phosphatase (ALP) staining experiments were used to determine ALP activity on day 3 (a) and day 7 (b) respectively. (D) For matrix mineralization, alizarin red S staining was performed on day 14 (a); microscopic (b) observations showed that down-regulation of Notch1 signaling inhibited BMP2-induced calcium deposition. (E) Quantitative analysis of ALP activities and calcium deposition. The ALP activity was quantified at OD 405 nm and normalized by protein concentration per well (unit/mg protein) on day 3 (a) and day 7 (b). Alizarin red staining was quantified at OD 405 nm and normalized to total DNA per well (OD405 nm/μg DNA) (c). (F) Western blot analysis for the chondrogenic differentiation markers Sox9 and Col2a1 and the osteogenic markers Runx2, Col1a1, and OPN. Protein bands (a) and quantitative analysis (b). The relative expression of Sox9, Col2a1, Runx2, Col1a1, and OPN proteins were analyzed using GAPDH as control (b). One-way analysis of variance; ∗∗∗∗p < 0.0001, ∗∗∗p < 0.001, ∗∗p < 0.01, and ∗p < 0.05 versus the AdGFP group; ####p < 0.0001, ###p < 0.001, ##p < 0.01, and #p < 0.05 versus the indicated group; ns, p > 0.05. BMP2, bone morphogenetic protein 2; Col1a1, collagen type I alpha 1 chain; Col2a1, collagen type II alpha 1 chain; MSC, mesenchymal stem cell; Notch1, Notch receptor 1; OPN, osteopontin; Runx2, RUNX family transcription factor 2; Sox9; SRY-box transcription factor 9.
Index in PubMed under a CC BY license. PMID: 40083323
Click image to see more details
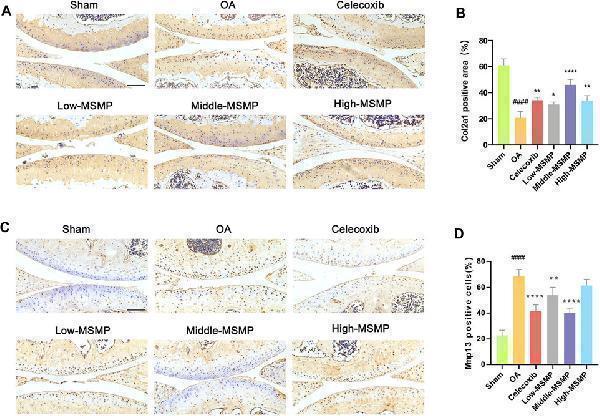
MSMP affected cartilage matrix metabolism in OA mice. (A) Immunohistochemical staining of Col2a1. (B) Statistical analysis of the positive expression for Col2a1. (C) Immunohistochemical staining of Mmp13. (D) Statistical analysis of the positive expression for Mmp13. Scale bar represents 100 μm (Date are mean ± SD, #### p < 0.0001, versus Sham group; * p < 0.05, ** p < 0.01, **** p < 0.0001, versus OA group).
Index in PubMed under a CC BY license. PMID: 38974041

Click image to see more details
Immunohistochemical staining of COL2A1 in vivo at indicated times postoperation. Scale bar: 200 μm
Index in PubMed under a CC BY license. PMID: 30479659
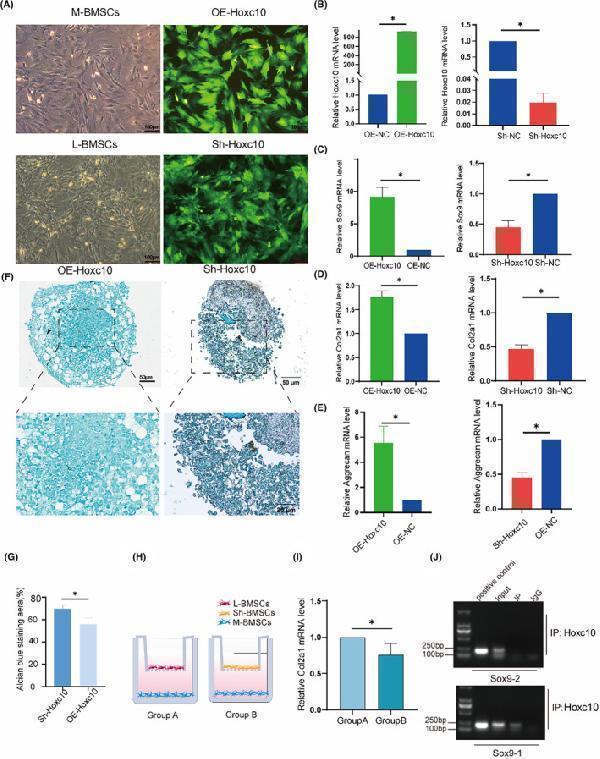
Click image to see more details
Hoxc10 is positively correlated with cartilage. n (C) q‐PCR validated the expression levels of Sox9 gene after overexpression and knockout of Hoxc10. (D) q‐PCR validated the expression levels of the Col2a1 gene after overexpression and knockout of Hoxc10. (E) q‐PCR validated the expression levels of Aggrecan gene after overexpression and knockout of Hoxc10. (F) The proteoglycan of BMSCs after overexpression and knockdown Hoxc10 was observed by Alcian blue staining 21 days after chondrogenic induction. (G) Quantitative analysis of Alcian blue staining. (H) Schematic diagram of co‐culture of L‐BMSCs and M‐BMSCs with and without Hoxc10 knockout. (I) Col2a1 gene expression in L‐BMSCs after Hoxc10 knockout and co‐culture with M‐BMSCs compared to control. (J) ChIP experiment of Sox9 and Hoxc10 protein binding. The data are presented as the mean ± SD ( n = 3).* p < 0.05.
Index in PubMed under a CC BY license. PMID: 39434203
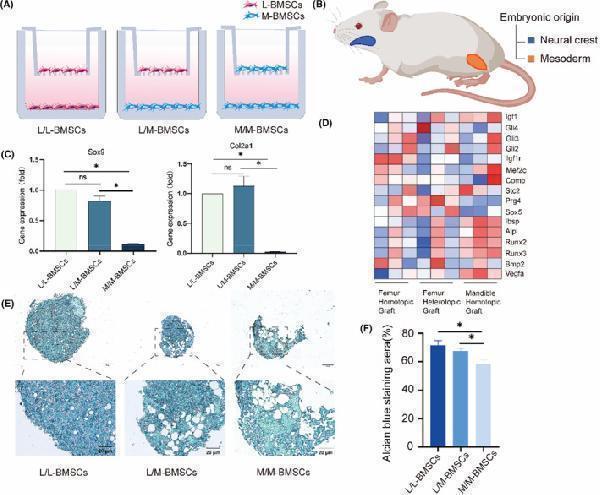
Click image to see more details
Hoxc10 is retained in L/M‐BMSCs in vitro. (A) Schematic of Transwell co‐culture model of L‐BMSCs and M‐BMSCs. (B) Schematic of the limb bones and mandibles from different embryonic origins. The mandible is of neural crest origin (blue) and the limb bone is of mesodermal origin (orange) (C) qPCR verified the gene expression levels of Sox9 and Col2a1 before and after co‐culture of L‐BMSCs and M‐BMSCs. (D) The proliferation, osteogenic and chondrogenic genes of femoral homotopic grafting, femoral heterotopic grafting and mandibles homotopic grafting. (E) After 21 days of chondrogenic induction in the upper layer cells of Transwell model before and after co cultivation with L‐BMSCs and M‐BMSCs, blue stained proteoglycans were observed using Alcian blue. (F) Quantitative analysis of Alizarin blue staining before and after co culture of L‐BMSCs and M‐BMSCs. The data are presented as the mean ± SD ( n = 3). * p < 0.05.
Index in PubMed under a CC BY license. PMID: 39434203
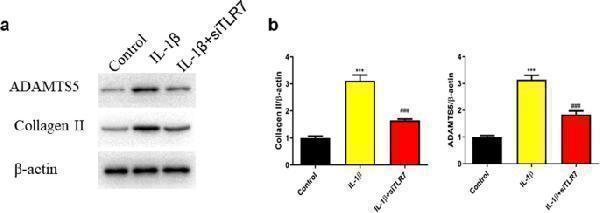
Click image to see more details
SiRNA TLR7 reversed collagen II and ADAMTS-5 degradation in IL-1β-stimulated chondrocytes. Cells were stimulated with IL-1β (10 ng/ml) for 48 h. The expression levels of collagen II and ADAMTS-5 were evaluated by Western blot ( a ) and quantification analysis ( b ). *** P < 0.001 versus the control group, ### P < 0.001 versus the IL-1β group.
Index in PubMed under a CC BY license. PMID: 37803031
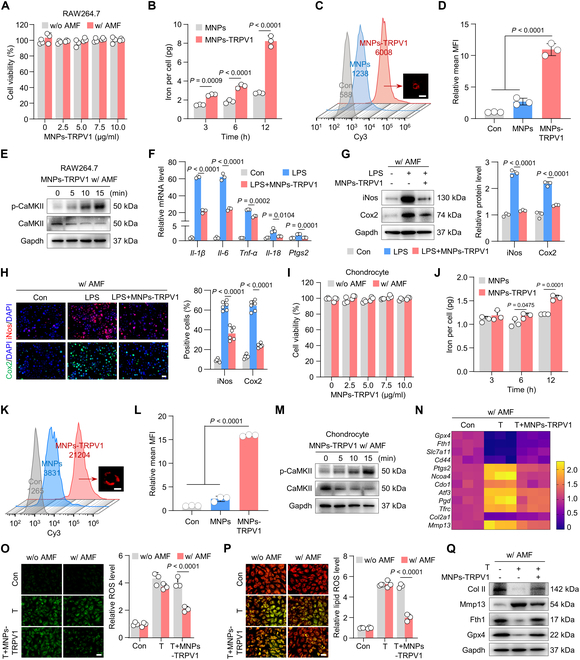
Click image to see more details
Magnetothermal suppression of macrophagic inflammation and chondrocyte ferroptosis by MNPs-TRPV1. (A) Cell viability of RAW264.7 macrophages treated with different concentrations of MNPs-TRPV1 with (w/) or without (w/o) AMF stimulation. (B) The inductively coupled plasma mass spectrometry (ICP-MS) detection of iron content per RAW264.7 cell after incubated with 5 mg/ml MNPs or MNPs-TRPV1 for 3, 6, and 12 h. (C and D) Flow cytometry analysis (C) and quantitative analysis (D) of sulfo-cyanine3 (Cy3) conjugated MNPs or MNPs-TRPV1 incubated RAW264.7 cells for 12 h. The representative image presented in (C) showed the combination of MNPs-TRPV1 to the plasma membrane of RAW264.7 cell. (E) Western blot analysis of the expression levels of CaMKII and p-CaMKII in RAW264.7 cells 0, 5, 10, and 15 min after the treatment of MNPs-TRPV1 with AMF stimulation. (F) qPCR analysis of inflammatory genes, including Il-1β, Il-6, Tnf-α, Il-18, and Ptgs2, in RAW264.7 cells treated with or without 50 ng ml−1 LPS in the presence or absence of MNP-TRPV1 pretreatment under AMF exposure. (G) Western blot analysis (left panel) and corresponding quantitative analysis (right panel) of the expression of inflammatory proteins, iNos and Cox2, expressed in RAW264.7 cells induced as indicated. (H) Immunofluorescence staining (left panel) and quantitative analysis (right panel) of iNos and Cox2 in RAW264.7 cell induced as indicated. (I) Cell viability of mouse primary chondrocytes treated with various concentrations of MNPs-TRPV1 with (w/) or without (w/o) AMF stimulation. (J) The ICP-MS detection of iron content per chondrocyte after incubated with 5 mg/ml MNPs or MNPs-TRPV1 for 3, 6, and 12 h. (K and L) Flow cytometry analysis (C) and quantification (D) of Cy3-conjugated MNPs or MNPs-TRPV1 incubated chondrocytes for 12 h. The representative image presented in (K) showed the combination of MNPs-TRPV1 to the plasma membrane of chondrocyte. (M) The proteins expression levels of CaMKII and p-CaMKII in chondrocytes treated as indicated at the time point of 0, 5, 10, and 15 min. (N) qPCR analysis of gene expression levels of ferroptosis suppressors (Gpx4, Fth1, Slc7a11, and Cd44), ferroptosis drivers (Ptgs2, Ncoa4, Cdo1, Atf3, Pgd, and Tfrc), cartilage anabolic marker (Col2a1), and cartilage catabolic marker (Mmp13) in chondrocytes treated as indicated. (O and P) Representative images (left panel) and quantification (right panel) of ROS (O) and lipid ROS (P) levels in mouse primary chondrocytes treated with TBHP (T) or T + MNPs-TRPV1 under the stimulation of AMF. (Q) Western blot analysis of expression levels of Col II, Mmp13, Fth1, and Gpx4 in chondrocytes treated as indicated. w/o, without; w/, with. Scale bars, 15 μm (C and K), 20 μm (H) and 50 μm (O and P). One-way (D, F, G, H, and L) or 2-way (A, B, I, J, O, and P) ANOVA with Tukey’s post hoc test. Data are shown as mean ± SD.
Index in PubMed under a CC BY license. PMID: 38371274
Click image to see more details
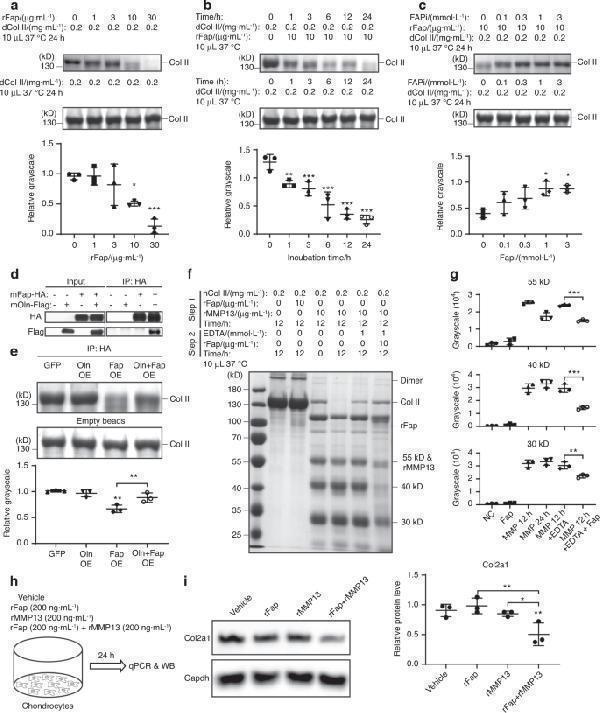
Fap degrades denatured or MMP13-cleaved Col II. a Fap degrades denatured Col II in a dose-dependent manner. Denatured Col II (boiled at 95 °C for 10 min) was incubated with different amounts of rFap at 37 °C for 24 h. Samples were separated by SDS‒PAGE and quantified by colloidal blue staining ( n = 3 independent experiments). b Fap degrades denatured Col II in a time-dependent manner. Denatured Col II was incubated with rFap at 37 °C for 0–24 h ( n = 3 independent experiments). c FAPi inhibits Fap-mediated degradation of denatured Col II. Different doses of FAPi were preincubated with rFap for 30 min before denatured Col II was added and further incubated at 37 °C for 24 h ( n = 3 independent experiments). d Immunoprecipitation of Fap. HEK293T cells were transfected with GFP control, Oln-Flag, Fap-HA, or Oln-Flag + Fap-HA. Two days after transfection, Fap was immunoprecipitated from total cell lysates with 10 μL anti-HA affinity gel. Five percent total cell lysates were loaded as an input control ( n = 3 independent experiments). e Oln inhibits the Fap-mediated degradation of denatured Col II. Col II was coincubated with immunoprecipitated samples (in 10 μL anti-HA affinity gel) at 37 °C for 24 h ( n = 3 independent experiments). f Fap degrades MMP13-cleaved Col II. Native Col II was preincubated with rFap or rMMP13 at 37 °C for 12 h. EDTA was then added to the reaction mixture with or without rFap and incubated at 37 °C for another 12 h ( n = 3 independent experiments). g Grayscale quantification of the 55, 40 and 30 kDa digestion bands in ( f ). h Experimental design. Primary chondrocytes were stimulated with vehicle control (PBS), 200 ng·mL −1 rFap, 200 ng·mL −1 rMMP13, or 200 ng·mL −1 rFap plus 200 ng·mL −1 rMMP13 for 24 h. i Western blot analysis of mouse Col2a1 protein levels in primary chondrocytes stimulated as in ( h ) ( n = 3 independent experiments). The statistical significance was assessed using one-way ANOVAs with Tukey’s multiple comparison tests. Data are presented as the mean ± SD (* P < 0.05, ** P < 0.01, *** P < 0.001)
Index in PubMed under a CC BY license. PMID: 36588124
Click image to see more details
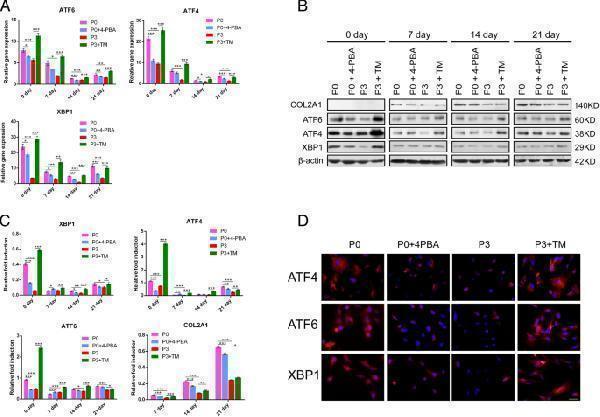
UPR-related gene/protein expression in four groups. a Quantitative RT-PCR for ATF6 , ATF4 , and XBP1 of the four groups in the cultured constructs on 0, 7, 14, and 21 days. These data were normalized to GAPDH . b Western blot results for ATF4, ATF6, XBP1, and COL2A1 at different culture times. COL2A1 was not detectable at 0 day; β-actin was used as a loading control. c Normalized expression of ATF4, ATF6, XBP1, and COL2A1 in response to Western blot analysis. d Immunofluorescence staining of ATF4, ATF6, and XBP1 at 0 days. Scale bar: 50 μm. The values were means ± S.D.; * indicate P < 0.05, ** indicate P < 0.01, *** indicate P < 0.001
Index in PubMed under a CC BY license. PMID: 30479659
Click image to see more details
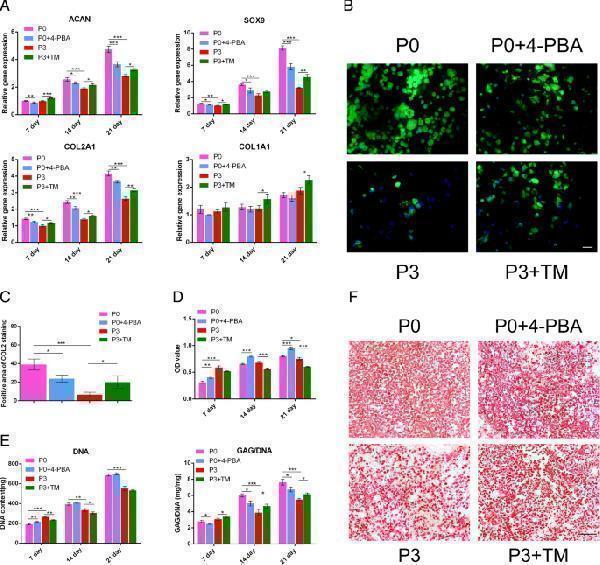
Chondrogenic-related gene/protein expression in four groups. a Quantitative RT-PCR for ACAN , SOX9 , COL2A1 , and COL1A1 of the four groups in the cultured constructs on 7, 14, and 21 days. These data were normalized to GAPDH . b Immunofluorescence staining of COL2A1 after 21 days of chondrogenic induction. Scale bar: 25 μm. c The semi-quantitative analysis of positive staining of COL2A1 in response to Fig. b. d MTT assay was used to analyze cell proliferation of the four groups: P0 BMSCs, P0 + 4-PBA, P3 BMSCs and P3 + TM. e The DNA and GAG contents were stained for the Hoechst 33258 and dimethylmethylene blue dye binding assays, respectively, after 7, 14 and 21 days of chondrogenic induction, and the absorbances were measured to quantify the contents of DNA and GAG. The left panel shows the DNA content and the right panel shows the ratio between GAG and DNA content. f GAG production was examined using Safranin-O staining in the four samples: P0 BMSCs, P0 with 4-PBA treatment, P3 BMSCs and P3 with TM treatment at 21 days. Scale bar: 100 μm. The values are means ± S.D.; * indicate P < 0.05, ** indicate P < 0.01, *** indicate P < 0.001
Index in PubMed under a CC BY license. PMID: 30479659
Click image to see more details
IF analysis of COL2A1 using anti-COL2A1 antibody (PA2141-1).
COL2A1 was detected in a paraffin-embedded section of Mouse cartilage tissue tissue. Heat mediated antigen retrieval was performed in EDTA buffer (pH 8.0, epitope retrieval solution). The tissue section was blocked with 10% goat serum. The tissue section was then incubated with 1:100 rabbit anti-COL2A1 Antibody (PA2141-1) overnight at 4°C. Donkey anti-rabbit Alexa Fluor 555was used as secondary antibody at 1:500 dilution and incubated for 1 hour at RT. The section was counterstained with DAPI. Visualize using a fluorescence microscope and filter sets appropriate for the label used.
Specific Publications For Anti-Collagen II/COL2A1 Antibody Picoband® (PA2141-1)
Loading publications
Recommended Resources
Here are featured tools and databases that you might find useful.
- Boster's Pathways Library
- Protein Databases
- Bioscience Research Protocol Resources
- Data Processing & Analysis Software
- Photo Editing Software
- Scientific Literature Resources
- Research Paper Management Tools
- Molecular Biology Software
- Primer Design Tools
- Bioinformatics Tools
- Phylogenetic Tree Analysis
Customer Reviews
Have you used Anti-Collagen II/COL2A1 Antibody Picoband®?
Share your experimental results or join a short interview to earn up to $1,000 in product credits or other rewards.
1 Reviews For Anti-Collagen II/COL2A1 Antibody Picoband®
This antibody exhibits high specificity, an excellent signal-to-noise ratio, and clear, stable staining, and has been successfully applied in immunofluorescent labeling of mouse cartilage tissue.
Excellent
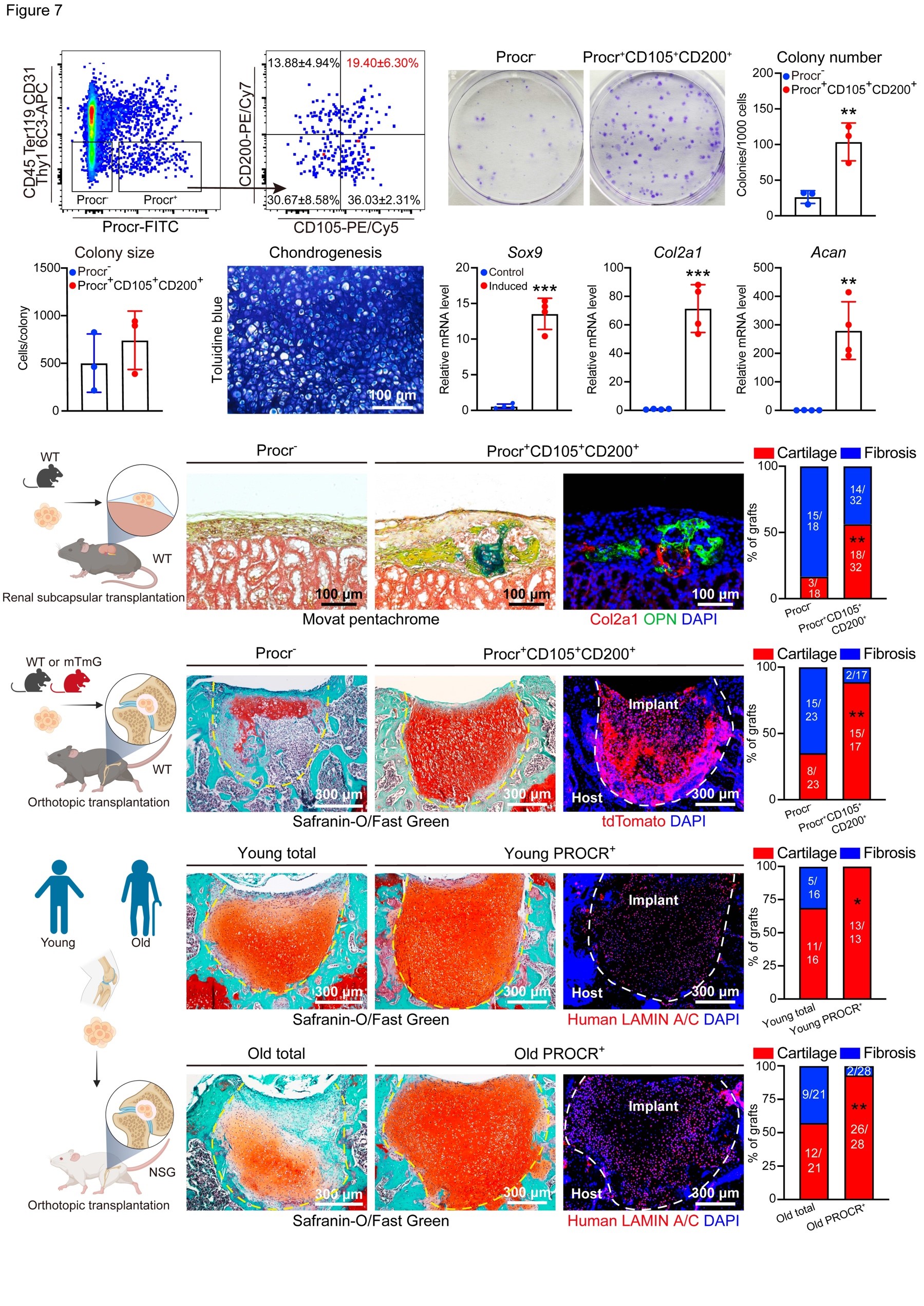
| SKU | PA2141-1 |
|---|---|
| Application | Immunofluorescence |
| Sample | mouse cartilage tissue |
| Sample Processing Description | Freshly dissected bone samples were first fixed overnight in 4% paraformaldehyde (PFA), followed by decalcification for 3 days in 10% ethylenediaminetetraacetic acid (EDTA) solution, and then dehydrated for 1 day in 30% sucrose solution. The bone samples were subsequently embedded in optimal cutting temperature compound (OCT; Sakura, Cat. No. OCT-4583) and cryosectioned at a thickness of 10 μm using a Leica CryoJane cryostat. |
| Primary Antibody | Anti-Collagen II/COL2A1 Antibody Picoband® |
| Primary Incubation | 1:100, overnight at 4 °C. |
| Secondary Antibody | donkey anti-rabbit Alexa Fluor 555 Secondary Antibody |
| Secondary Incubation | 1:500, Incubate at room temperature for 1 hour |
| Other Reagents used | PBS with 10% horse serum, DAPI |
| Detection | fluorescence microscope (Leica) |
| Results Summary | I will purchase BosterBio products again and recommend them to my classmates and colleagues. |
Qiaoling Zhu, Tongji University
Verified customer
Submitted 2025-10-16
Customer Q&As
Have a question?
Find answers in Q&As, reviews.
Can't find your answer?
Submit your question
16 Customer Q&As for Anti-Collagen II/COL2A1 Antibody Picoband®
Question
I am looking for using your anti-Collagen II/COL2A1 antibody for r-mmu-198933; immunoregulatory interactions between a lymphoid and a non-lymphoid cell studies. Has this antibody been tested with western blotting on brain tissue? We would like to see some validation images before ordering.
Verified Customer
Verified customer
Asked: 2020-04-15
Answer
I appreciate your inquiry. This PA2141-1 anti-Collagen II/COL2A1 antibody is tested on rat trachea tissue, tissue lysate, heart tissue, brain tissue. It is guaranteed to work for IHC, WB in mouse, rat. Our Boster guarantee will cover your intended experiment even if the sample type has not been be directly tested.
Boster Scientific Support
Answered: 2020-04-15
Question
I see that the anti-Collagen II/COL2A1 antibody PA2141-1 works with IHC, what is the protocol used to produce the result images on the product page?
Verified Customer
Verified customer
Asked: 2020-02-18
Answer
You can find protocols for IHC on the "support/technical resources" section of our navigation menu. If you have any further questions, please send an email to support@bosterbio.com
Boster Scientific Support
Answered: 2020-02-18
Question
I am interested in to test anti-Collagen II/COL2A1 antibody PA2141-1 on mouse tibia for research purposes, then I may be interested in using anti-Collagen II/COL2A1 antibody PA2141-1 for diagnostic purposes as well. Is the antibody suitable for diagnostic purposes?
Verified Customer
Verified customer
Asked: 2019-12-27
Answer
The products we sell, including anti-Collagen II/COL2A1 antibody PA2141-1, are only intended for research use. They would not be suitable for use in diagnostic work. If you have the means to develop a product into diagnostic use, and are interested in collaborating with us and develop our product into an IVD product, please contact us for more discussions.
Boster Scientific Support
Answered: 2019-12-27
Question
Our team were satisfied with the WB result of your anti-Collagen II/COL2A1 antibody. However we have been able to see positive staining in fetal sternum extracellular using this antibody. Is that expected? Could you tell me where is COL2A1 supposed to be expressed?
Verified Customer
Verified customer
Asked: 2019-11-01
Answer
Based on literature, fetal sternum does express COL2A1. Generally COL2A1 expresses in secreted, extracellular space, extracellular. Regarding which tissues have COL2A1 expression, here are a few articles citing expression in various tissues:
Blood, Pubmed ID: 8948452
Embryonic stem cell, and Muscle, Pubmed ID: 15489334
Fetal sternum, Pubmed ID: 1999183
Boster Scientific Support
Answered: 2019-11-01
Question
Is this PA2141-1 anti-Collagen II/COL2A1 antibody reactive to the isotypes of COL2A1?
Verified Customer
Verified customer
Asked: 2019-10-08
Answer
The immunogen of PA2141-1 anti-Collagen II/COL2A1 antibody is A synthetic peptide corresponding to a sequence at the C-terminus of mouse Collagen II1222-1241aa AFAGLGQREKGPDPMQYMRA), different from the related rat sequence by one amino acid. Could you tell me which isotype you are interested in so I can help see if the immunogen is part of this isotype?
Boster Scientific Support
Answered: 2019-10-08
Question
I have a question about product PA2141-1, anti-Collagen II/COL2A1 antibody. I was wondering if it would be possible to conjugate this antibody with biotin. I would need it to be without BSA or sodium azide. I am planning on using a buffer exchange of sodium azide with PBS only. Would there be problems for me to conjugate the antibody and store it in -20 degrees in small aliquots?
Verified Customer
Verified customer
Asked: 2019-08-29
Answer
It is not recommended storing this antibody with PBS buffer only in -20 degrees. If you want to store it in -20 degrees it is best to add some cryoprotectant like glycerol. If you want carrier free PA2141-1 anti-Collagen II/COL2A1 antibody, we can provide it to you in a special formula with trehalose and/or glycerol. These molecules will not interfere with conjugation chemistry and provide a good level of protection for the antibody from degradation. Please be sure to specify this in your purchase order.
Boster Scientific Support
Answered: 2019-08-29
Question
Is there a BSA free version of anti-Collagen II/COL2A1 antibody PA2141-1 available?
Verified Customer
Verified customer
Asked: 2019-07-08
Answer
We appreciate your recent telephone inquiry. I can confirm that some lots of this anti-Collagen II/COL2A1 antibody PA2141-1 are BSA free. For now, these lots are available and we can make a BSA free formula for you free of charge. It will take 3 extra days to prepare. If you require this antibody BSA free again in future, please do not hesitate to contact me and I will be pleased to check which lots we have in stock that are BSA free.
Boster Scientific Support
Answered: 2019-07-08
Question
We have tried in the past anti-Collagen II/COL2A1 antibody for WB on blood last year. I am using rat, and We intend to use the antibody for IHC next. I am interested in examining blood as well as tibia in our next experiment. Do you have any suggestion on which antibody would work the best for IHC?
Verified Customer
Verified customer
Asked: 2018-07-16
Answer
I took a look at the website and datasheets of our anti-Collagen II/COL2A1 antibody and it seems that PA2141-1 has been tested on rat in both WB and IHC. Thus PA2141-1 should work for your application. Our Boster satisfaction guarantee will cover this product for IHC in rat even if the specific tissue type has not been validated. We do have a comprehensive range of products for IHC detection and you can check out our website bosterbio.com to find out more information about them.
Boster Scientific Support
Answered: 2018-07-16
Question
Thank you for helping with my inquiry over the phone. Here are the WB image, lot number and protocol we used for tibia using anti-Collagen II/COL2A1 antibody PA2141-1. Let me know if you need anything else.
Verified Customer
Verified customer
Asked: 2018-07-02
Answer
We appreciate the data. You have provided everything we needed. Our lab team are working to resolve your inquiry as quickly as possible, and we appreciate your patience and understanding! Please let me know if there is anything you need in the meantime.
Boster Scientific Support
Answered: 2018-07-02
Question
Would anti-Collagen II/COL2A1 antibody PA2141-1 work for IHC with tibia?
Verified Customer
Verified customer
Asked: 2018-05-24
Answer
According to the expression profile of tibia, COL2A1 is highly expressed in tibia. So, it is likely that anti-Collagen II/COL2A1 antibody PA2141-1 will work for IHC with tibia.
Boster Scientific Support
Answered: 2018-05-24
Question
Will PA2141-1 anti-Collagen II/COL2A1 antibody work on parafin embedded sections? If so, which fixation method do you recommend we use (PFA, paraformaldehyde, other)?
Verified Customer
Verified customer
Asked: 2018-01-29
Answer
It shows on the product datasheet, PA2141-1 anti-Collagen II/COL2A1 antibody as been validated on IHC. It is best to use PFA for fixation because it has better tissue penetration ability. PFA needs to be prepared fresh before use. Long term stored PFA turns into formalin, as the PFA molecules congregate and become formalin.
Boster Scientific Support
Answered: 2018-01-29
Question
I have attached the WB image, lot number and protocol we used for tibia using anti-Collagen II/COL2A1 antibody PA2141-1. Please let me know if you require anything else.
Verified Customer
Verified customer
Asked: 2017-12-29
Answer
Thank you very much for the data. Our lab team are working to resolve this as quickly as possible, and we appreciate your patience and understanding! You have provided everything we needed. Please let me know if there is anything you need in the meantime.
Boster Scientific Support
Answered: 2017-12-29
Question
I was wanting to use your anti-Collagen II/COL2A1 antibody for IHC for mouse tibia on frozen tissues, but I want to know if it has been tested for this particular application. Has this antibody been tested and is this antibody a good choice for mouse tibia identification?
B. Jones
Verified customer
Asked: 2017-12-26
Answer
As indicated on the product datasheet, PA2141-1 anti-Collagen II/COL2A1 antibody has been tested for IHC, WB on mouse, rat tissues. We have an innovator award program that if you test this antibody and show it works in mouse tibia in IHC-frozen, you can get your next antibody for free.
Boster Scientific Support
Answered: 2017-12-26
Question
Is a blocking peptide available for product anti-Collagen II/COL2A1 antibody (PA2141-1)?
Verified Customer
Verified customer
Asked: 2017-07-14
Answer
We do provide the blocking peptide for product anti-Collagen II/COL2A1 antibody (PA2141-1). If you would like to place an order for it please contact support@bosterbio.com and make a special request.
Boster Scientific Support
Answered: 2017-07-14
Question
We are currently using anti-Collagen II/COL2A1 antibody PA2141-1 for mouse tissue, and we are well pleased with the IHC results. The species of reactivity given in the datasheet says mouse, rat. Is it possible that the antibody can work on zebrafish tissues as well?
Verified Customer
Verified customer
Asked: 2017-06-06
Answer
The anti-Collagen II/COL2A1 antibody (PA2141-1) has not been validated for cross reactivity specifically with zebrafish tissues, though there is a good chance of cross reactivity. We have an innovator award program that if you test this antibody and show it works in zebrafish you can get your next antibody for free. Please contact me if I can help you with anything.
Boster Scientific Support
Answered: 2017-06-06
Question
We have seen staining in mouse blood. Any tips? Is anti-Collagen II/COL2A1 antibody supposed to stain blood positively?
D. Moore
Verified customer
Asked: 2016-08-19
Answer
From what I have seen in literature blood does express COL2A1. From what I have seen in Uniprot.org, COL2A1 is expressed in tibia, blood, embryonic stem cell muscle, fetal sternum, among other tissues. Regarding which tissues have COL2A1 expression, here are a few articles citing expression in various tissues:
Blood, Pubmed ID: 8948452
Embryonic stem cell, and Muscle, Pubmed ID: 15489334
Fetal sternum, Pubmed ID: 1999183
Boster Scientific Support
Answered: 2016-08-19





