Product Info Summary
| SKU: | M00344-3 |
|---|---|
| Size: | 0.1 mg |
| Reactive Species: | Human |
| Host: | Mouse |
| Application: | Flow Cytometry, IP |
Customers Who Bought This Also Bought
Product info
Product Name
Anti-Ms CD4 Purified Azide Free Monoclonal Antibody
SKU/Catalog Number
M00344-3
Size
0.1 mg
Form
Liquid
Description
Boster Bio Anti-Ms CD4 Purified Azide Free Monoclonal Antibody (Catalog# M00344-3). Tested in Flow Cytometry, IP application(s). This antibody reacts with Human.
Storage & Handling
Store at 2-8°C. Do not freeze.
Cite This Product
Anti-Ms CD4 Purified Azide Free Monoclonal Antibody (Boster Biological Technology, Pleasanton CA, USA, Catalog # M00344-3)
Host
Mouse
Contents
Phosphate buffered saline (PBS), pH 7.4, azide-free
Clonality
Monoclonal
Clone Number
BB7.2
Isotype
Mouse IgG2b
Immunogen
HLA-A2 solubilised by papain. The antibody BB7.2 recognizes an extracellular epitope at the C-terminus of alpha-2 helix and a turn on one of the underlying beta strands within the human HLA-A2 histocompatibility antigen.
Reactive Species
M00344-3 is reactive to CD4 in Human
Observed Molecular Weight
42 kDa
Calculated molecular weight
51.3 kDa
Background of CD4
HLA-A2 (44 kDa) is the most frequent HLA-A allele in human ethnic populations. HLA-A, together with HLA-B and HLA-C, represent human HLA class I major histocompatibility (MHC) antigens. These intrinsic membrane glycoproteins are expressed on nucleated cells and noncovalently associate with an invariant beta2 microglobulin. They carry foreign determinants important for immune recognition by cytotoxic T cells, thus important for anti-viral and anti-tumour defence.
Antibody Validation
Boster validates all antibodies on WB, IHC, ICC, Immunofluorescence, and ELISA with known positive control and negative samples to ensure specificity and high affinity, including thorough antibody incubations.
Application & Images
Applications
M00344-3 is guaranteed for Flow Cytometry, IP Boster Guarantee
Assay Dilutions Recommendation
The recommendations below provide a starting point for assay optimization. The actual working concentration varies and should be decided by the user.
Flow cytometry: 1 μg/ml.
Validation Images & Assay Conditions
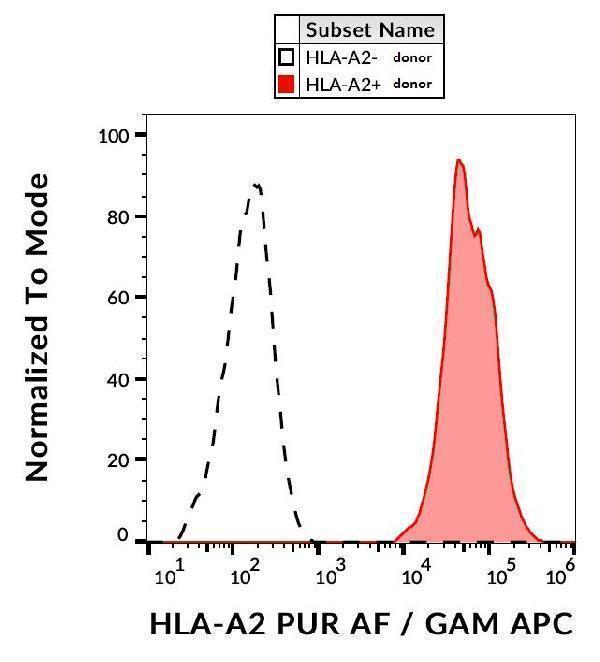
Click image to see more details
Flow cytometry analysis (surface staining) of human peripheral blood cells using anti-HLA-A2 (BB7.2) purified, GAM-APC.
Specific Publications For Anti-Ms CD4 Purified Azide Free Monoclonal Antibody (M00344-3)
Loading publications
Recommended Resources
Here are featured tools and databases that you might find useful.
- Boster's Pathways Library
- Protein Databases
- Bioscience Research Protocol Resources
- Data Processing & Analysis Software
- Photo Editing Software
- Scientific Literature Resources
- Research Paper Management Tools
- Molecular Biology Software
- Primer Design Tools
- Bioinformatics Tools
- Phylogenetic Tree Analysis
Customer Reviews
Have you used Anti-Ms CD4 Purified Azide Free Monoclonal Antibody?
Share your experimental results or join a short interview to earn up to $1,000 in product credits or other rewards.
0 Reviews For Anti-Ms CD4 Purified Azide Free Monoclonal Antibody
Customer Q&As
Have a question?
Find answers in Q&As, reviews.
Can't find your answer?
Submit your question




