Product Info Summary
| SKU: | A02059-1 |
|---|---|
| Size: | 100 μg/vial |
| Reactive Species: | Human, Monkey, Mouse, Rat |
| Host: | Rabbit |
| Application: | ELISA, IF, IHC, WB |
Customers Who Bought This Also Bought
Product info
Product Name
Anti-Glutathione Peroxidase 4/GPX4 Antibody Picoband®
SKU/Catalog Number
A02059-1
Size
100 μg/vial
Form
Lyophilized
Description
Boster Bio Anti-Glutathione Peroxidase 4/GPX4 Antibody Picoband® catalog # A02059-1. Tested in ELISA, Flow Cytometry, IHC, WB applications. This antibody reacts with Human, Monkey, Mouse, Rat. The brand Picoband indicates this is a premium antibody that guarantees superior quality, high affinity, and strong signals with minimal background in Western blot applications. Only our best-performing antibodies are designated as Picoband, ensuring unmatched performance.
Storage & Handling
At -20°C for one year from date of receipt. After reconstitution, at 4°C for one month. It can also be aliquotted and stored frozen at -20°C for six months. Avoid repeated freezing and thawing.
Cite This Product
Anti-Glutathione Peroxidase 4/GPX4 Antibody Picoband® (Boster Biological Technology, Pleasanton CA, USA, Catalog # A02059-1)
Host
Rabbit
Contents
Each vial contains 4 mg Trehalose, 0.9 mg NaCl, 0.2 mg Na2HPO4.
Clonality
Polyclonal
Isotype
Rabbit IgG
Immunogen
E.coli-derived human Glutathione Peroxidase 4/GPX4 recombinant protein (Position: A30-F197).
Cross-reactivity
No cross-reactivity with other proteins.
Reactive Species
A02059-1 is reactive to GPX4 in Human, Monkey, Mouse, Rat
Observed Molecular Weight
19 kDa
Calculated molecular weight
22.2 kDa
Background of GPX4
Glutathione peroxidase 4, also known as GPX4, is an enzyme that in humans is encoded by the GPX4 gene. This gene encodes a member of the glutathione peroxidase protein family. Glutathione peroxidase catalyzes the reduction of hydrogen peroxide, organic hydroperoxide, and lipid peroxides by reduced glutathione and functions in the protection of cells against oxidative damage. Human plasma glutathione peroxidase has been shown to be a selenium-containing enzyme and the UGA codon is translated into a selenocysteine. The encoded protein has been identified as a moonlighting protein based on its ability to serve dual functions as a peroxidase as well as a structural protein in mature spermatozoa. Through alternative splicing and transcription initiation, rat produces proteins that localize to the nucleus, mitochondrion, and cytoplasm. In humans, alternative transcription initiation and the cleavage sites of the mitochondrial and nuclear transit peptides need to be experimentally verified. Alternative splicing results in multiple transcript variants.
Antibody Validation
Boster validates all antibodies on WB, IHC, ICC, Immunofluorescence, and ELISA with known positive control and negative samples to ensure specificity and high affinity, including thorough antibody incubations.
Application & Images
Applications
A02059-1 is guaranteed for ELISA, IF, IHC, WB Boster Guarantee
Assay Dilutions Recommendation
The recommendations below provide a starting point for assay optimization. The actual working concentration varies and should be decided by the user.
Western blot, 0.25-0.5 μg/ml, Human, Monkey, Mouse, Rat
Immunohistochemistry(Paraffin-embedded Section), 2-5 μg/ml, Human, Mouse
Flow Cytometry(Fixed), 1-3 μg/1x106 cells, Human
ELISA, 0.1-0.5 μg/ml, -
Positive Control
WB: human HepG2 whole cell, human MCF-7 whole cell, human RT4 whole cell, monkey COS-7 whole cell, rat kidney tissue, rat testis tissue, mouse kidney tissue, human HCT116- WT whole cell, human HCT116-GPX4 KO whole cell
IHC: human testis tissue, mouse testis tissue
FCM: THP-1 cell
Validation Images & Assay Conditions
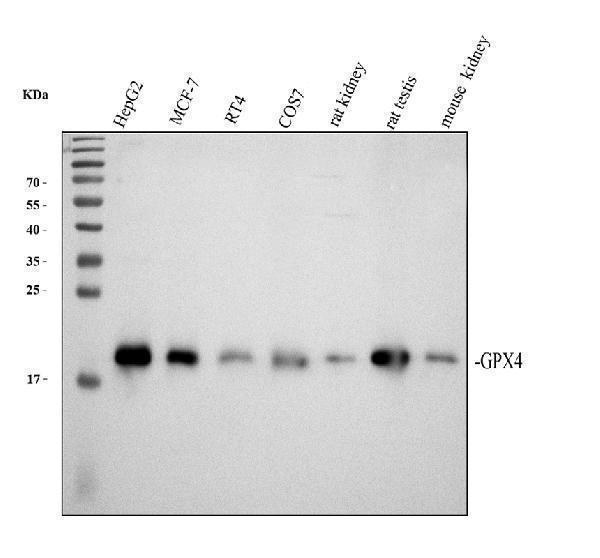
Click image to see more details
Western blot analysis of Glutathione Peroxidase 4/GPX4 using anti-Glutathione Peroxidase 4/GPX4 antibody (A02059-1).
Electrophoresis was performed on a 12% SDS-PAGE gel at 80V (Stacking gel) / 120V (Resolving gel) for 2 hours. The sample well of each lane was loaded with 30 ug of sample under reducing conditions.
Lane 1: human HepG2 whole cell lysates,
Lane 2: human MCF-7 whole cell lysates,
Lane 3: human RT4 whole cell lysates,
Lane 4: monkey COS-7 whole cell lysates,
Lane 5: rat kidney tissue lysates,
Lane 6: rat testis tissue lysates,
Lane 7: mouse kidney tissue lysates.
After electrophoresis, proteins were transferred to a nitrocellulose membrane at 150 mA for 50-90 minutes. Blocked the membrane with 5% non-fat milk/TBS for 1.5 hour at RT. The membrane was incubated with rabbit anti-Glutathione Peroxidase 4/GPX4 antigen affinity purified polyclonal antibody (Catalog # A02059-1) at 0.5 μg/mL overnight at 4°C, then washed with TBS-0.1%Tween 3 times with 5 minutes each and probed with a goat anti-rabbit IgG-HRP secondary antibody at a dilution of 1:5000 for 1.5 hour at RT. The signal is developed using an ECL Plus Western Blotting Substrate (Catalog # AR1196-200) with Tanon 5200 system. A specific band was detected for Glutathione Peroxidase 4/GPX4 at approximately 19 kDa. The expected band size for Glutathione Peroxidase 4/GPX4 is at 22 kDa.
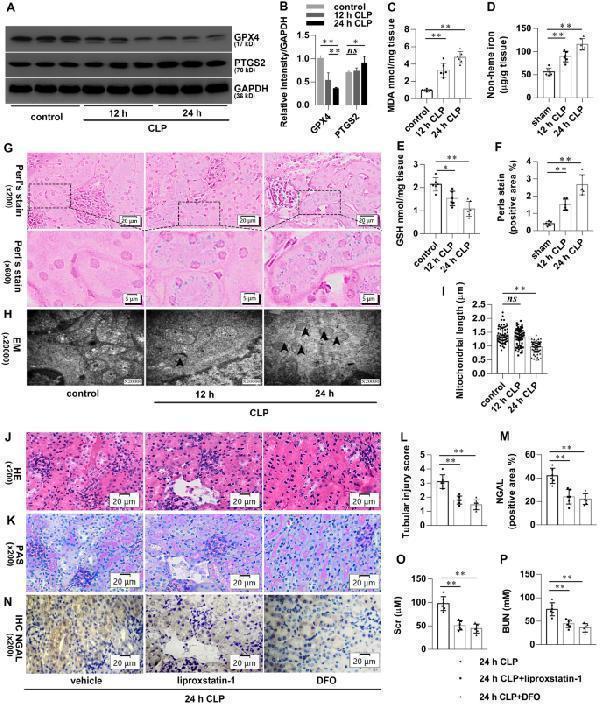
Click image to see more details
Ferroptosis is present in CLP-induced AKI. Mice were subjected to CLP, then the kidneys were collected at the indicated time. A , B Shown are representative western blotting and quantification of GPX4 and PTGS2. C–E Quantitative analyses of MDA, GSH, and non-heme iron. F Quantitative analyses of Perl’s stain. G Representative Perl’s stain images. H Representative transmission electron microscopy (TEM) images. The black arrow indicates ferroptosis-like mitochondria. I Quantitative analyses of mitochondrial length. Mice were pretreated with liproxstatin-1 (10 mg/kg, ip) or DFO (20 mg/kg, ip) 0.5 h before being subjected to CLP. All kidney samples were collected at 24 h after CLP. J–K Shown are representative Hematoxylin-eosin (HE) stains and Periodic Acid-Schiff (PAS) stain. L Histological analyses of renal tubular injury. M Quantitative analyses of IHC stain of NGAL. N Representative immunohistochemistry (IHC) images for NGAL. O , P Quantitative analyses of Scr and BUN. n = 6 mice/group, mean ± SD were presented. *P < 0.05, **P < 0.01 and ns : P > 0.05 . ip: intraperitoneal
Index in PubMed under a CC BY license. PMID: 34961563
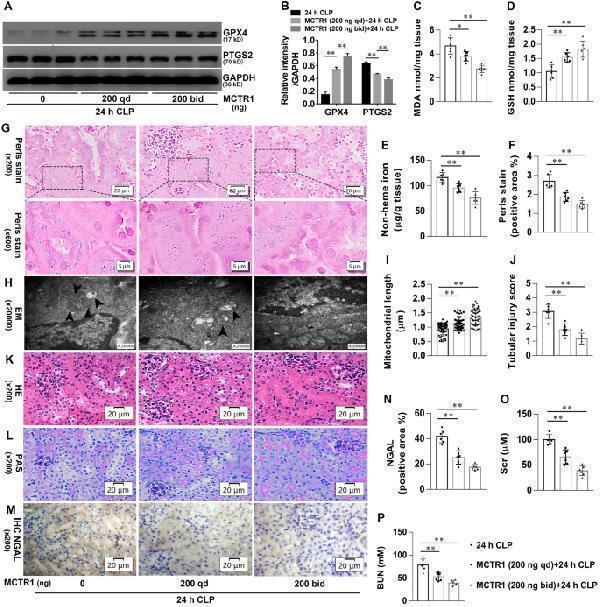
Click image to see more details
Effects of MCTR1 on ferroptosis in CLP-induced AKI. Mice were given MCTR1 once, twice, or not during the 24 h experiment. The once-daily administration mode (qd): Mice were given MCTR1 (200 ng/mice, iv) 0.5 h before being subjected to CLP. The twice-daily administration mode (bid): Mice were given MCTR1 (200 ng/mice, iv) 0.5 h before being subjected to CLP, and then an additional MCTR1 (200 ng/mice, iv) was given 12 h after CLP. All kidney samples were collected at 24 h after CLP. A , B Shown are representative western blotting and quantification of GPX4 and PTGS2. C–E Quantitative analyses of MDA, GSH, and non-heme iron. F Quantitative analyses of Perl’s stain. G Representative Perl’s stain images. H Representative TEM images. The black arrow indicates ferroptosis-like mitochondria. I Quantitative analyses of mitochondrial length. J Histological analyses of renal tubular injury. K , L Shown are representative HE stains and PAS stain. M Representative IHC images for NGAL. N Quantitative analyses of IHC stain of NGAL. O , P Quantitative analyses of serum Scr and BUN. n = 6 mice/group, mean ± SD were presented. **P < 0.01 . iv: intravenous
Index in PubMed under a CC BY license. PMID: 34961563
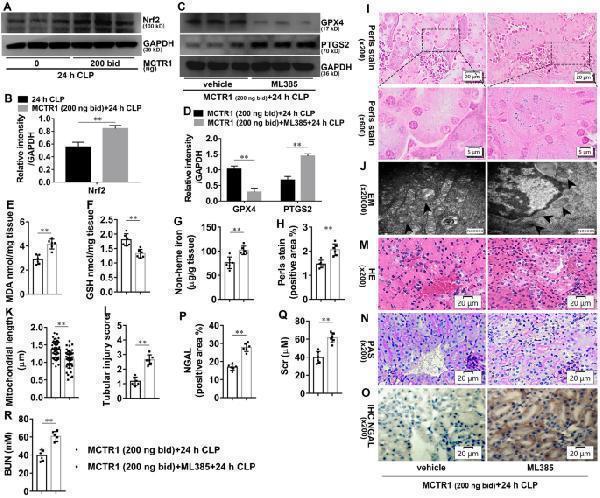
Click image to see more details
Effects of Nrf2 on MCTR1-regulated ferroptosis in CLP-induced AKI. Mice were given ML385 (30 mg/kg, ip, qd) for 7 d. On day 7, ML385 injection with or without a twice-daily administration mode of MCTR1 as described before in the following experiment. All kidney samples were collected at 24 h after CLP. A–D Shown are representative western blotting and quantification of Nrf2, GPX4, and PTGS2. E – G Quantitative analyses of MDA, GSH, and non-heme iron. H Quantitative analyses of Perl’s stain. I Representative Perl’s stain images. J Representative TEM images. The black arrow indicates ferroptosis-like mitochondria. K Quantitative analyses of mitochondrial length. L Histological analyses of renal tubular injury. M , N Representative HE stains and PAS stain. O Representative IHC images for NGAL. P Quantitative analyses of IHC stain of NGAL. Q , R Quantitative analyses of serum Scr and BUN. n = 6 mice/group, mean ± SD were presented. **P < 0.01
Index in PubMed under a CC BY license. PMID: 34961563
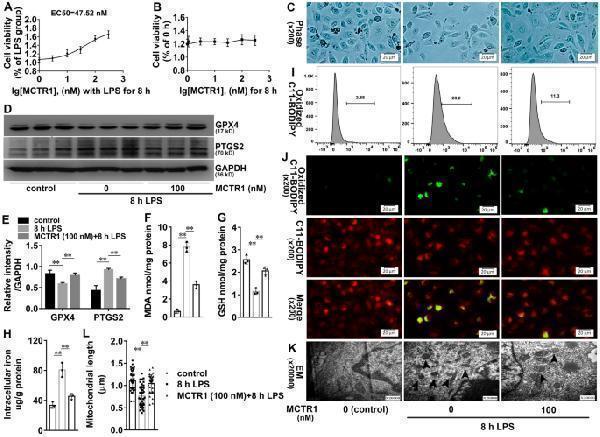
Click image to see more details
Effects of MCTR1 on LPS-induced lipid peroxidation and ferroptosis in vitro. A Viability curves for HK-2 cells treated with different concentrations (0, 1, 3, 10, 30, 100, and 300 nM) of MCTR1 and LPS (1 ug/ml) for 8 h. B Viability curves for HK-2 cells treated with different concentrations (0, 1, 3, 10, 30, 100, and 300 nM) of MCTR1 for 0 and 8 h. C HK-2 cells treated with MCTR1 (100 nM) and LPS (1 ug/ml) for 8 h, and the visualization of cell viability were evaluated by phase-contrast microscopy. D , E Representative western blotting and quantification of GPX4 and PTGS2 protein. F – H Quantitative analyses of MDA, GSH and, non-heme iron. I Quantitative analyses of oxidized C11-BODIPY 581/591 probe by flow cytometry. J Representative images of C11-BODIPY 581/591 fluorescent ratio-probe. K Representative TEM images. The black arrow indicates ferroptosis-like mitochondria. L Quantitative analyses of mitochondrial length. n = 3, mean ± SD were presented. **P < 0.01
Index in PubMed under a CC BY license. PMID: 34961563
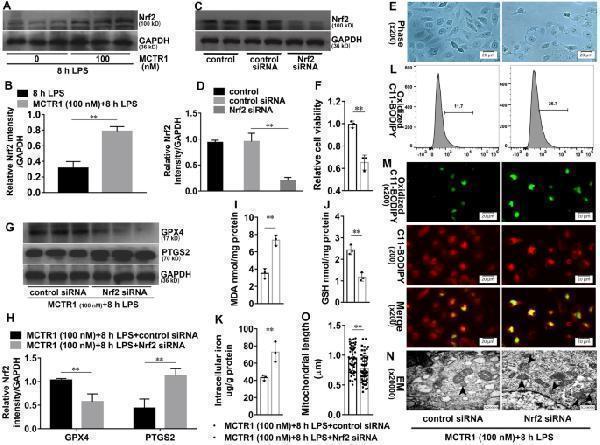
Click image to see more details
Nrf2 is involved in the inhibitory effects of MCTR1 on LPS-induced ferroptosis. A , B HK-2 cells were treated with MCTR1 (100 nM) and LPS (1 ug/ml) for 8 h. Shown are representative western blotting and quantification of Nrf2. HK-2 cells were transfected with 30 nM Nrf2 siRNA for 48 h and then treated with MCTR1 (100 nM) and LPS (1 ug/ml) for 8 h. C , D Representative western blotting and quantification of Nrf2. E Visualization of cell viability were evaluated by phase-contrast microscopy. F Fold change of cell viability. G , H Representative western blotting and quantification of GPX4 and PTGS2. I – K Quantitative analyses of MDA, GSH, and non-heme iron. L Quantitative analyses of oxidized C11-BODIPY 581/591 probe by flow cytometry. M Representative images of C11-BODIPY 581/591 fluorescent ratio-probe. N Representative TEM images. The black arrow indicates ferroptosis-like mitochondria. O Quantitative analyses of mitochondrial length. n = 3, mean ± SD were presented. **P < 0.01
Index in PubMed under a CC BY license. PMID: 34961563
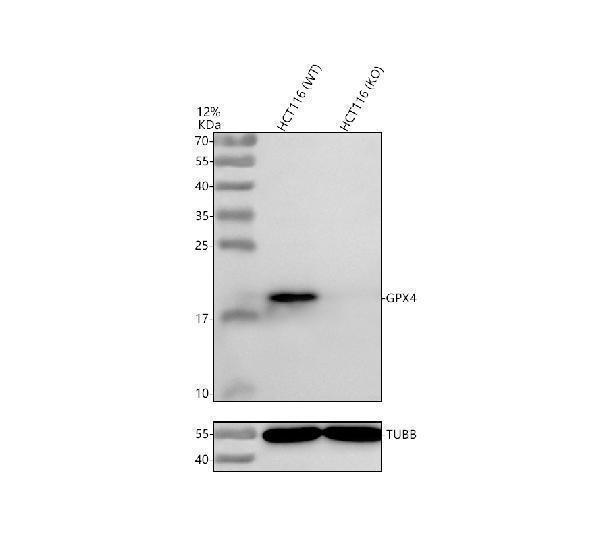
Click image to see more details
Western blot analysis of Glutathione Peroxidase 4/GPX4 using anti-Glutathione Peroxidase 4/GPX4 antibody (A02059-1).
Electrophoresis was performed on a 12% SDS-PAGE gel at 80V (Stacking gel) / 120V (Resolving gel) for 2 hours. The sample well of each lane was loaded with 30 ug of sample under reducing conditions.
Lane 1: human HCT116- WT whole cell lysates,
Lane 2: human HCT116-GPX4 KO whole cell lysates.
After electrophoresis, proteins were transferred to a nitrocellulose membrane at 150 mA for 50-90 minutes. Blocked the membrane with 5% non-fat milk/TBS for 1.5 hour at RT. Then the membrane was incubated with rabbit anti-Glutathione Peroxidase 4/GPX4 antigen affinity purified polyclonal antibody (A02059-1) at 0.5 μg/mL overnight at 4°C, then washed with TBS-0.1%Tween 3 times with 5 minutes each and probed with a goat anti-rabbit IgG-HRP secondary antibody (Catalog # BA1054) at a dilution of 1:5000 for 1.5 hour at RT. The signal is developed using an ECL Plus Western Blotting Substrate (Catalog # AR1196-200) with Tanon 5200 system. A specific band was detected for Glutathione Peroxidase 4/GPX4 at approximately 19 kDa. The expected band size for Glutathione Peroxidase 4/GPX4 is at 22 kDa.
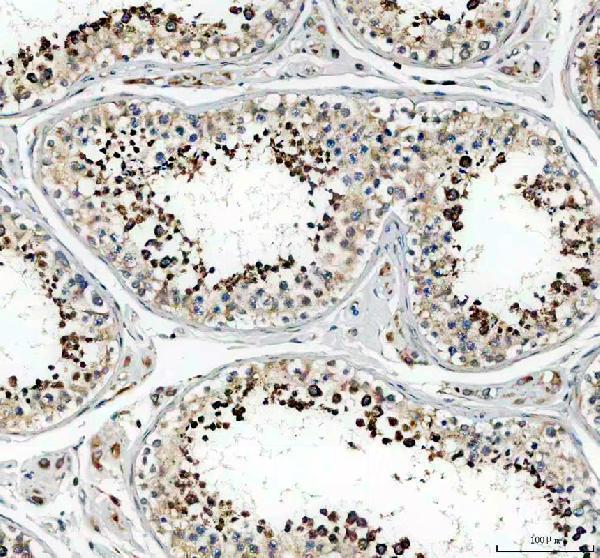
Click image to see more details
IHC analysis of Glutathione Peroxidase 4/GPX4 using anti-Glutathione Peroxidase 4/GPX4 antibody (A02059-1).
Glutathione Peroxidase 4/GPX4 was detected in a paraffin-embedded section of human testis tissue. Heat mediated antigen retrieval was performed in EDTA buffer (pH 8.0, epitope retrieval solution). The tissue section was blocked with 10% goat serum. The tissue section was then incubated with 2 μg/ml rabbit anti-Glutathione Peroxidase 4/GPX4 Antibody (A02059-1) overnight at 4°C. Peroxidase Conjugated Goat Anti-rabbit IgG was used as secondary antibody and incubated for 30 minutes at 37°C. The tissue section was developed using HRP Conjugated Rabbit IgG Super Vision Assay Kit (Catalog # SV0002) with DAB as the chromogen.
Click image to see more details
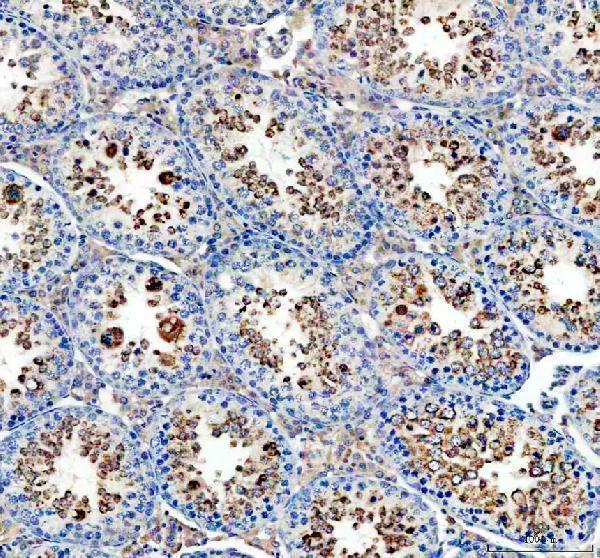
IHC analysis of Glutathione Peroxidase 4/GPX4 using anti-Glutathione Peroxidase 4/GPX4 antibody (A02059-1).
Glutathione Peroxidase 4/GPX4 was detected in a paraffin-embedded section of mouse testis tissue. Heat mediated antigen retrieval was performed in EDTA buffer (pH 8.0, epitope retrieval solution). The tissue section was blocked with 10% goat serum. The tissue section was then incubated with 2 μg/ml rabbit anti-Glutathione Peroxidase 4/GPX4 Antibody (A02059-1) overnight at 4°C. Peroxidase Conjugated Goat Anti-rabbit IgG was used as secondary antibody and incubated for 30 minutes at 37°C. The tissue section was developed using HRP Conjugated Rabbit IgG Super Vision Assay Kit (Catalog # SV0002) with DAB as the chromogen.
Click image to see more details
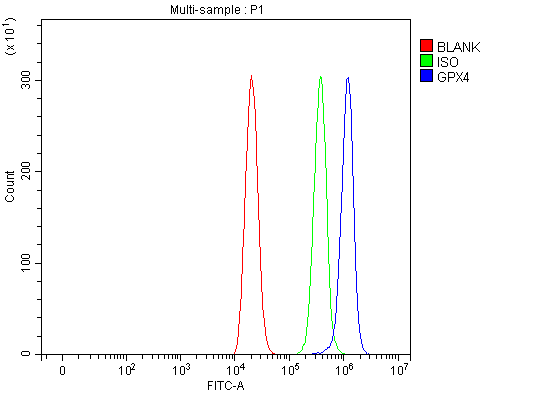
Flow Cytometry analysis of THP-1 cells using anti-Glutathione Peroxidase 4/GPX4 antibody (A02059-1).
Overlay histogram showing THP-1 cells stained with A02059-1 (Blue line). To facilitate intracellular staining, cells were fixed with 4% paraformaldehyde and permeabilized with permeabilization buffer. The cells were blocked with 10% normal goat serum. And then incubated with rabbit anti-Glutathione Peroxidase 4/GPX4 Antibody (A02059-1, 1 μg/1x106 cells) for 30 min at 20°C. DyLight®488 conjugated goat anti-rabbit IgG (BA1127, 5-10 μg/1x106 cells) was used as secondary antibody for 30 minutes at 20°C. Isotype control antibody (Green line) was rabbit IgG (1 μg/1x106) used under the same conditions. Unlabelled sample without incubation with primary antibody and secondary antibody (Red line) was used as a blank control.
Specific Publications For Anti-Glutathione Peroxidase 4/GPX4 Antibody Picoband® (A02059-1)
Loading publications
Recommended Resources
Here are featured tools and databases that you might find useful.
- Boster's Pathways Library
- Protein Databases
- Bioscience Research Protocol Resources
- Data Processing & Analysis Software
- Photo Editing Software
- Scientific Literature Resources
- Research Paper Management Tools
- Molecular Biology Software
- Primer Design Tools
- Bioinformatics Tools
- Phylogenetic Tree Analysis
Customer Reviews
Have you used Anti-Glutathione Peroxidase 4/GPX4 Antibody Picoband®?
Share your experimental results or join a short interview to earn up to $1,000 in product credits or other rewards.
0 Reviews For Anti-Glutathione Peroxidase 4/GPX4 Antibody Picoband®
Customer Q&As
Have a question?
Find answers in Q&As, reviews.
Can't find your answer?
Submit your question





