Product Info Summary
| SKU: | PB9253 |
|---|---|
| Size: | 100 μg/vial |
| Reactive Species: | Human, Mouse, Rat |
| Host: | Rabbit |
| Application: | IHC, WB |
Customers Who Bought This Also Bought
Product info
Product Name
Anti-HIF-1-alpha/HIF1A Antibody Picoband®
SKU/Catalog Number
PB9253
Size
100 μg/vial
Form
Lyophilized
Description
Boster Bio Anti-HIF-1-alpha/HIF1A Antibody Picoband® catalog # PB9253. Tested in IHC, WB applications. This antibody reacts with Human, Mouse, Rat. The brand Picoband indicates this is a premium antibody that guarantees superior quality, high affinity, and strong signals with minimal background in Western blot applications. Only our best-performing antibodies are designated as Picoband, ensuring unmatched performance.
Storage & Handling
Store at -20˚C for one year from date of receipt. After reconstitution, at 4˚C for one month. It can also be aliquotted and stored frozen at -20˚C for six months. Avoid repeated freeze-thaw cycles.
Cite This Product
Anti-HIF-1-alpha/HIF1A Antibody Picoband® (Boster Biological Technology, Pleasanton CA, USA, Catalog # PB9253)
Host
Rabbit
Contents
Each vial contains antibody formulated with stabilizing components, 0.9 mg NaCl, 0.2 mg Na2HPO4, and 0.05 mg NaN3.
*This antibody is supplied in a stabilized formulation.
Compatibility with conjugation reactions depends on the chemistry of the conjugation method used.
For conjugation methods that are not compatible with the stabilizing components present in this formulation, a carrier-free antibody format is required.
Clonality
Polyclonal
Isotype
Rabbit IgG
Immunogen
A synthetic peptide corresponding to a sequence at the C-terminal of human HIF-1-alpha, different from the related mouse and rat sequences by three amino acids.
Cross-reactivity
No cross-reactivity with other proteins
Reactive Species
PB9253 is reactive to HIF1A in Human, Mouse, Rat
Observed Molecular Weight
120 kDa
Calculated molecular weight
92.7 kDa
Background of HIF1A
HIF-1α (Hypoxia-inducible factor 1α, HIF1A) is a transcription factor that mediates cellular and systemic homeostatic responses to reduced O2 availability in mammals, including angiogenesis, erythropoiesis and glycolysis. This gene was mapped to 14q21-q24. HIF-1α transactivate genes required for energy metabolism and tissue perfusion and is necessary for embryonic development and tumor explant growth. HIF-1alpha is over expressed during carcinogenesis, myocardial infarction and wound healing. It is crucial for the cellular response to hypoxia and is frequently over expressed in human cancers, resulting in the activation of genes essential for cell survival. HIF-1α regulates the survival and function in the inflammatory microenvironment directly. It is a transcription factor that plays a pivotal role in cellular adaptation to changes in oxygen availability.
Antibody Validation
Boster validates all antibodies on WB, IHC, ICC, Immunofluorescence, and ELISA with known positive control and negative samples to ensure specificity and high affinity, including thorough antibody incubations.
Application & Images
Applications
PB9253 is guaranteed for IHC, WB Boster Guarantee
Recommend Dilution
| Application | Dilution | Species |
|---|---|---|
| Immunohistochemistry (Paraffin-embedded Section) | 0.5-1μg/ml | Human, Mouse, Rat |
| Western blot | 0.1-0.5μg/ml | Human, Mouse |
Tested application
Suggested blocking solution with 5% non-fat milk or BSA; (*)Recommended protein loading: 20-40 µg per lane
Use TE buffer pH 9.0 for antigen retrieval; (*) citrate buffer pH 6.0 is an alternative.
Validation Images & Assay Conditions
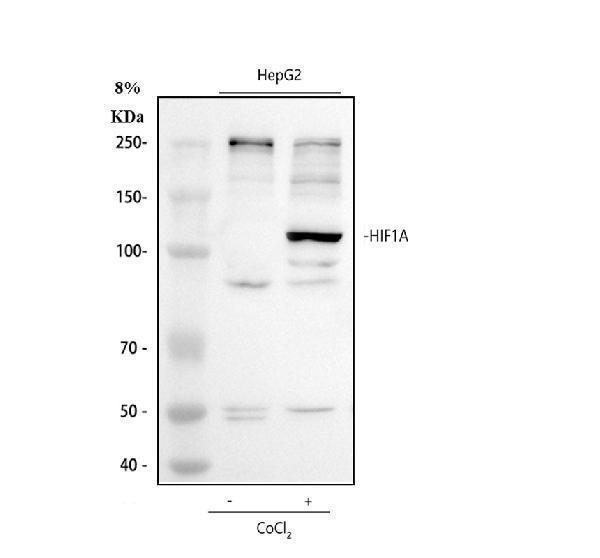
Click image to see more details
Western blot analysis of HIF 1 alpha using anti-HIF 1 alpha antibody (PB9253).
Electrophoresis was performed on a 8% SDS-PAGE gel at 80V (Stacking gel) / 120V (Resolving gel) for 2 hours. The sample well of each lane was loaded with 30 ug of sample under reducing conditions.
Lane 1: Untreated human HepG2 whole cell lysates,
Lane 2: Cobalt Chloride treated human HepG2 whole cell lysates.
After electrophoresis, proteins were transferred to a nitrocellulose membrane at 150 mA for 50-90 minutes. Blocked the membrane with 5% non-fat milk/TBS for 1.5 hour at RT. The membrane was incubated with rabbit anti-HIF 1 alpha antigen affinity purified polyclonal antibody (PB9253) at 0.5 μg/mL overnight at 4°C, then washed with TBS-0.1%Tween 3 times with 5 minutes each and probed with a goat anti-rabbit IgG-HRP secondary antibody (Catalog # BA1054) at a dilution of 1:5000 for 1.5 hour at RT. The signal is developed using an ECL Plus Western Blotting Substrate (Catalog # AR1196-200) with Tanon 5200 system. A specific band was detected for HIF 1 alpha at approximately 120 kDa. The expected band size for HIF 1 alpha is at 93 kDa.
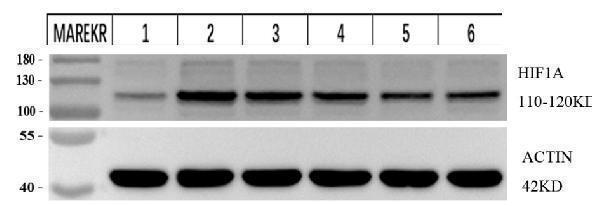
Click image to see more details
Western blot analysis of HIF1A using anti-HIF1A antibody (PB9253).
Electrophoresis was performed on a 5-20% SDS-PAGE gel at 70V (Stacking gel) / 90V (Resolving gel) for 2-3 hours. The sample well of each lane was loaded with 30 ug of sample under reducing conditions.
Lane 1: mouse 4T1 whole cell lysates,
Lane 2: LPS-stimulated mouse 4T1 whole cell lysates,
Lane 3: Low-dose drug mouse 4T1 whole cell lysates,
Lane 4: Medium-dose drug mouse 4T1 whole cell lysates,
Lane 5: High-dose drug mouse 4T1 whole cell lysates,
Lane 6: Positive control drug mouse 4T1 whole cell lysates.
After electrophoresis, proteins were transferred to a nitrocellulose membrane at 150 mA for 50-90 minutes. Blocked the membrane with 5% non-fat milk/TBS for 1 hour at RT. The membrane was incubated with rabbit anti-HIF1A antigen affinity purified monoclonal antibody (Catalog # PB9253) at 1:2500 overnight at 4°C, then washed with TBS-0.1%Tween 3 times with 5 minutes each and probed with a goat anti-rabbit IgG-HRP secondary antibody at a dilution of 1:10000 for 1 hour at RT. The signal is developed using an Enhanced Chemiluminescent detection (ECL) kit with ChemiDoc MP system. A specific band was detected for HIF1A at approximately 110-120 kDa. The expected band size for HIF1A is at 120kDa.
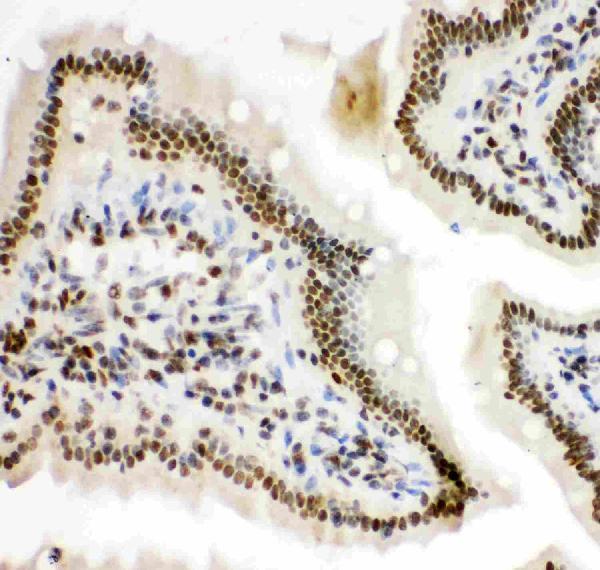
Click image to see more details
IHC analysis of HIF 1 alpha using anti-HIF 1 alpha antibody (PB9253).
HIF 1 alpha was detected in paraffin-embedded section of mouse intestine tissue. Heat mediated antigen retrieval was performed in citrate buffer (pH6, epitope retrieval solution) for 20 mins. The tissue section was blocked with 10% goat serum. The tissue section was then incubated with 1μg/ml rabbit anti-HIF 1 alpha Antibody (PB9253) overnight at 4°C. Biotinylated goat anti-rabbit IgG was used as secondary antibody and incubated for 30 minutes at 37°C. The tissue section was developed using Strepavidin-Biotin-Complex (SABC)(Catalog # SA1022) with DAB as the chromogen.
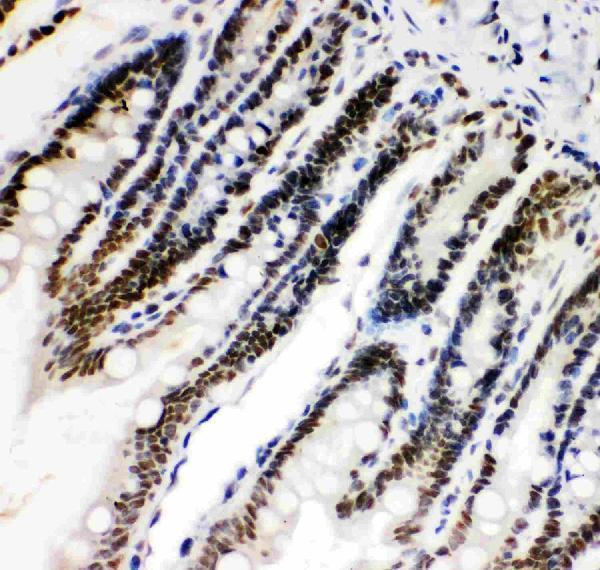
Click image to see more details
IHC analysis of HIF 1 alpha using anti-HIF 1 alpha antibody (PB9253).
HIF 1 alpha was detected in paraffin-embedded section of rat intestine tissue. Heat mediated antigen retrieval was performed in citrate buffer (pH6, epitope retrieval solution) for 20 mins. The tissue section was blocked with 10% goat serum. The tissue section was then incubated with 1μg/ml rabbit anti-HIF 1 alpha Antibody (PB9253) overnight at 4°C. Biotinylated goat anti-rabbit IgG was used as secondary antibody and incubated for 30 minutes at 37°C. The tissue section was developed using Strepavidin-Biotin-Complex (SABC)(Catalog # SA1022) with DAB as the chromogen.
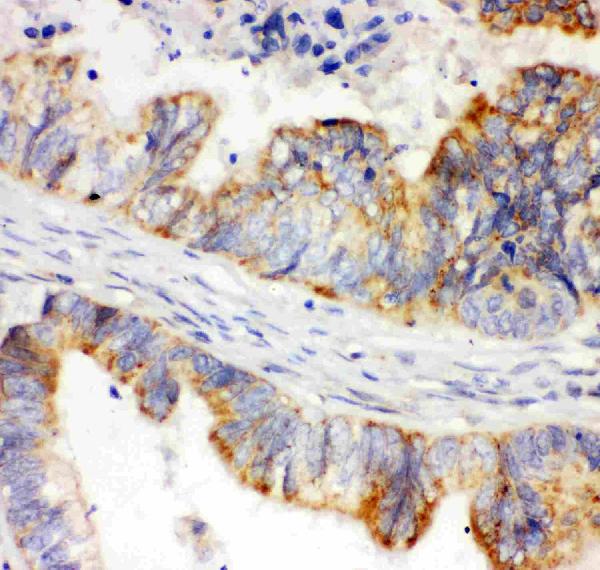
Click image to see more details
IHC analysis of HIF 1 alpha using anti-HIF 1 alpha antibody (PB9253).
HIF 1 alpha was detected in paraffin-embedded section of human intestinal cancer tissue. Heat mediated antigen retrieval was performed in citrate buffer (pH6, epitope retrieval solution) for 20 mins. The tissue section was blocked with 10% goat serum. The tissue section was then incubated with 1μg/ml rabbit anti-HIF 1 alpha Antibody (PB9253) overnight at 4°C. Biotinylated goat anti-rabbit IgG was used as secondary antibody and incubated for 30 minutes at 37°C. The tissue section was developed using Strepavidin-Biotin-Complex (SABC)(Catalog # SA1022) with DAB as the chromogen.
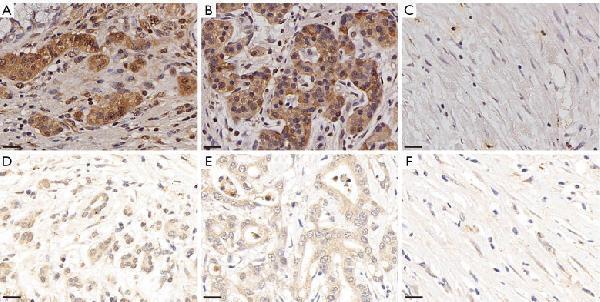
Click image to see more details
HIF-1α expression was analyzed by immunohistochemistry. Results of a 61-year-old male patient who underwent NAC and surgery treatment for PDAC, and had a CAP score of 2, which indicated that he was a responder. HIF-1α was highly expressed in the (A) nucleus (weighted score 12), (B) cytoplasm (weighted score 12), and (C) stroma (weighted score 4); results of a 30-year-old male patient who underwent NAC and surgery treatment for PDAC, and had a CAP score of 3, which indicated that he was a non-responder. HIF-1α was lowly expressed in the (D) nucleus (weighted score 4), (E) cytoplasm (weighted score 4), and (F) stroma (weighted score 4). 40×; scale bar =20 µm. NAC, neoadjuvant chemotherapy; PDAC, pancreatic ductal adenocarcinoma; CAP, the College of American Pathologists; HIF-1α, hypoxia-inducible factor-1-alpha.
Index in PubMed under a CC BY license. PMID: 39839013
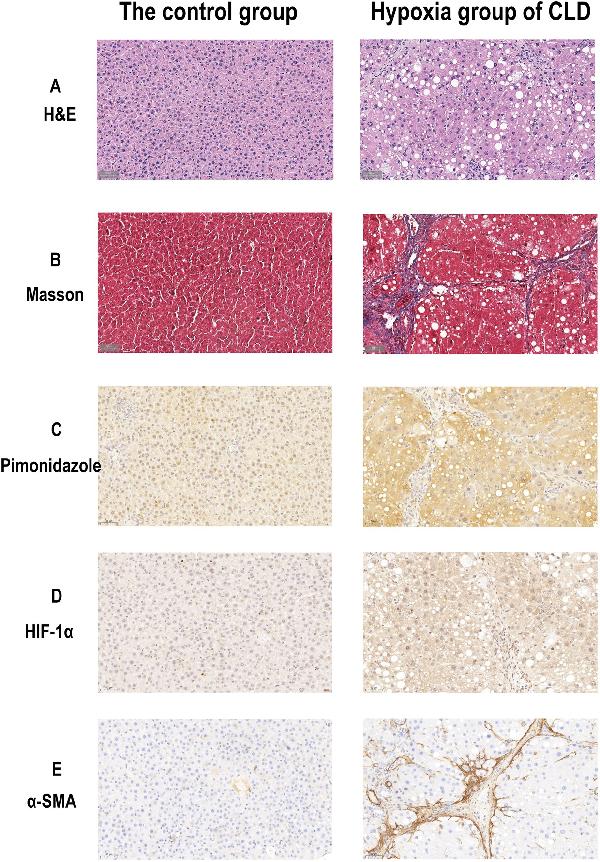
Click image to see more details
Histological and immunohistochemical images of the rat livers of the control group and hypoxia group of CLD. (A) H&E staining images (× 40) of the control and hypoxia group of CLD. (B) Masson staining images (× 40) of the control group and hypoxia group of CLD. (C) Pimonidazole Immunohistochemical staining images (× 40) of control and hypoxia group of CLD. (D) HIF-1α Immunohistochemical staining images (× 40) of control and hypoxia group of CLD. (E) α-SMA Immunohistochemical staining images (× 40) of control and hypoxia group of CLD.
Index in PubMed under a CC BY license. PMID: 39906347
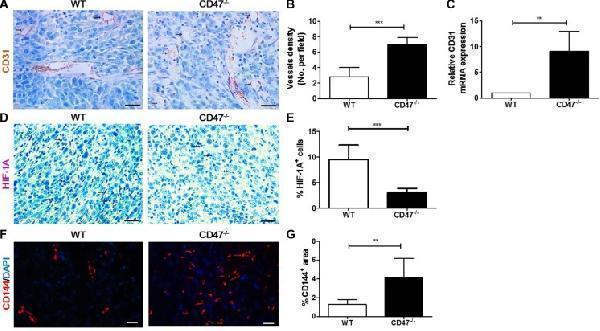
Click image to see more details
Tumors from CD47-deficient mice exhibited improved tumor angiogenesis and vascular integrity compared to those from WT mice. Tumors were surgically removed from WT or CD47 −/− mice 11 days after tumor cell injection for the following analyses ( n = 4 per group). ( A ) Representative images of CD31 staining (black arrow) of tumor sections (Scale bar, 20 μm). ( B ) The microvessel density (MVD) quantified by counting positive cells in six randomly selected fields (400×) using Image Pro Plus 6.0 software. ( C ) CD31 mRNA levels in tumors quantified by real-time qPCR. ( D ) Representative images of HIF-1A staining (pink) of tumor sections (Scale bar, 20 μm). ( E ) Percentages of HIF-1A + cells in tumors quantified by counting positive cells in six randomly selected fields (400×) using Image Pro Plus 6.0 software. ( F ) Representative images of CD144 staining (red) of tumor sections (Scale bar, 20 μm). ( G ) Percentages of CD144 + area in tumors quantified using Image Pro Plus 6.0 software. Data are mean ± SDs. ** P < 0.01, *** P < 0.001.
Index in PubMed under a CC BY license. PMID: 27283989
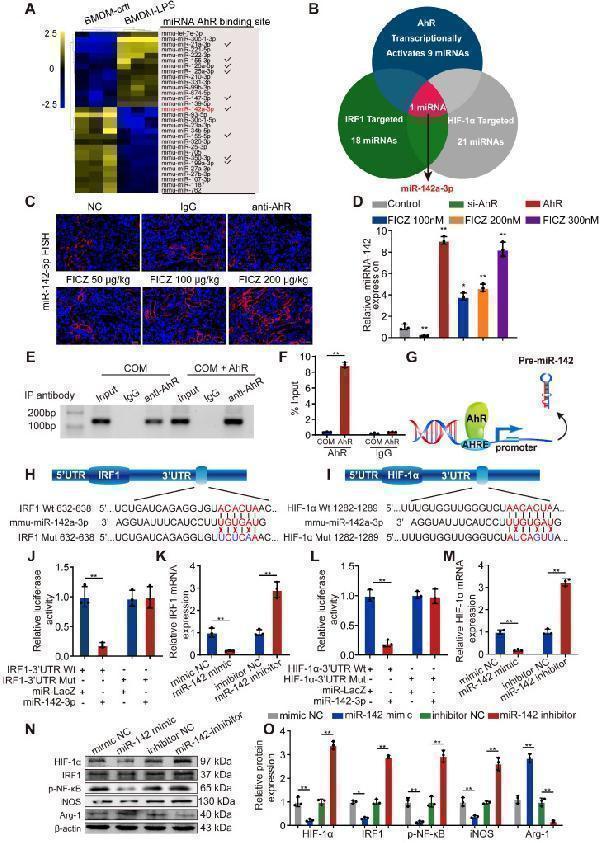
Click image to see more details
AhR transcriptionally activates miR-142a to inhibit IRF1 and HIF-1α expression. (A) The top 30 miRNAs in BMDMs that are regulated by LPS are arranged in a miRNA array heatmap. In addition, miRNAs predicted to be under the transcriptional control of AhR (according to analysis with the JASPAR database) are noted. (B) Venn diagram analyses were performed to identify miRNAs that can both target IRF1 and HIF-1α and that are under the transcriptional control of AhR. (C) Renal expression of mmu-miR-142a-3p in mice (n = 6) with CaOx nephrocalcinosis following treatment with an AhR neutralizing antibody or FICZ treatment was assessed via FISH (200×; scale bar: 20 µm). (D) qRT-PCR was performed to measure mmu-miR-142a-3p expression in BMDMs using U6 RNA as a normalization control. (E, F) ChIP assays and ChIP qPCR analysis showed that AhR bound to the miR-142a promoter in BMDMs treated with the AhR overexpression plasmid. (G) A schematic model showed that AhR directly binds to the miR-142a promoter and activates its transcription. (H, I) WT and mutated miR-142a targeting sequences in the IRF1 and HIF-1α 3'-UTR regions that were used to construct luciferase reporters, with reporters bearing these IRF1 (J) or HIF-1α (L) 3'-UTR sequences co-transfected along with miR-142a mimic (100 nM). IRF1 (K) and HIF-1α (M) mRNA levels were detected via qRT-PCR in BMDMs following miR-142a mimic or inhibitor transfection. Western blotting (N, O) analysis enabled the detection of IRF1 and HIF-1α expression while also assessing the levels of iNOS and Arg-1 to monitor the polarization state of BMDMs following miR-142a mimic or inhibitor transfection. β-actin was employed as a normalization control. The data are shown as the means ± SD of triplicate experiments. *P < 0.05; **P < 0.01, as assessed via Student's t test (D, F) or one-way ANOVA (J-M, O).
Index in PubMed under a CC BY license. PMID: 33204326
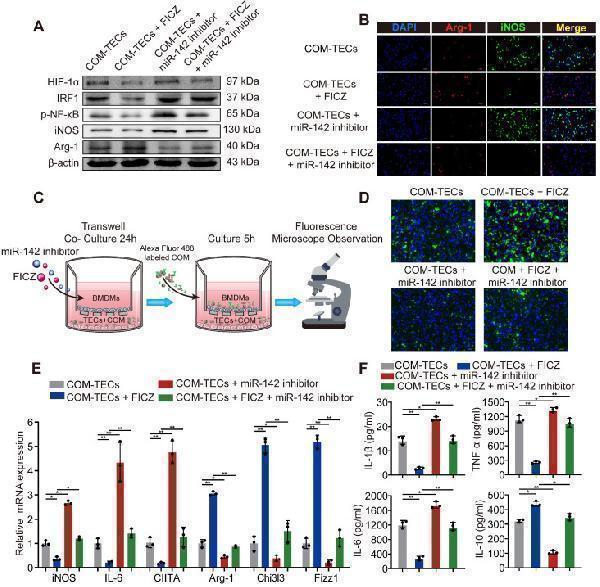
Click image to see more details
AhR activation in vitro decrease M1 macrophage polarization to inhibit kidney inflammation and injury through the AhR-miR-142a-IRF1/HIF-1α axis in vitro . (A) Western blotting analysis enabled the detection of AhR, HIF-1α, IRF1, NF-κB p65, iNOS, and Arg-1 expression in BMDMs. β-actin was detected as an internal control. (B) iNOS (M1 macrophage marker, green) and Arg-1 (M2 macrophage marker, red) distributions in BMDMs were detected by immunofluorescence (200×; scale bar: 20 µm). (C) Schematic diagram of BMDMs phagocytic capacity testing. (D) Fluorescence microscopy was performed to analyse the phagocytic ability of BMDMs (200×; scale bar: 20 µm). (E) qRT-PCR analysis of iNOS, IL-6, CIITA, Arg-1, Chi3l3 and Fizz1 expression to further determine polarization state of BMDM. (F) ELISA was used to quantify cytokine levels in the co-culture media. The data are shown as the means ± SD of triplicate experiments. *P < 0.05; **P < 0.01, as assessed via one-way ANOVA (E, F).
Index in PubMed under a CC BY license. PMID: 33204326
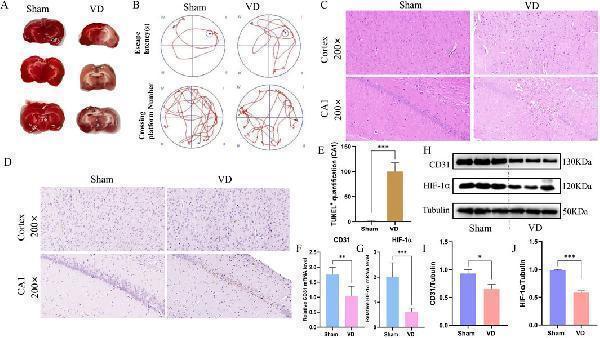
Click image to see more details
Construction of animal models and analysis of pathological damage. (A) TTC staining plots of the Sham and VD group. (B) Motion trajectory diagram. (C) Representative images stained with HE (scale bars = 50 μm). (D) Representative histopathological images obtained from TUNEL staining (scale bars = 50 μm), showing changes in the cortex hippocampus CA1. (E) Quantification of TUNEL+ cells in the hippocampal CA1 (n = 3). (F) Expression profile of CD31 between Sham and VD groups (qPCR). **P < 0.01. (G) Expression profile of HIF-1α between Sham and VD groups (qPCR; n = 6). ***P < 0.001. (H) WB strips. (I) Expression profile of CD31 between Sham and VD groups (WB). *P < 0.05. (J) Expression profile of HIF-1α between Sham and VD groups (WB; n = 3). ***P < 0.001.
Index in PubMed under a CC BY license. PMID: 40988927
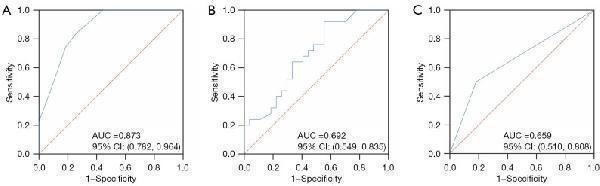
Click image to see more details
Receiver operator characteristic curves of nuclear HIF-1α expression (A), %ΔCA19-9 (B), and tumor differentiation (C) for predicting the post-NAC pathological response. HIF-1α, hypoxia-inducible factor-1α; NAC, neoadjuvant chemotherapy; %Δ, [(post-NAC − pre-NAC)/pre-NAC]; CA19-9, carbohydrate antigen 19-9; AUC, area under the curve; CI, confidence interval.
Index in PubMed under a CC BY license. PMID: 39839013
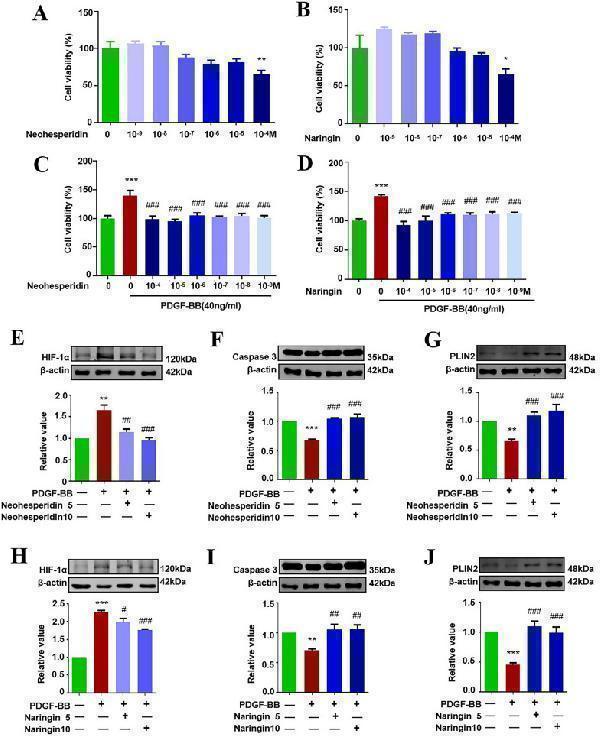
Click image to see more details
Effect of neohesperidin and naringin on cell viability, HIF-1α, Caspase3, PLIN2 in PASMCs. A – D Effects of neohesperidin and naringin on PASMCs viability were observed by MTT (n = 6). E – J Expression levels of HIF-1α, Caspase3, PLIN2 in PASMCs were evaluated by Western Blot (n ≥ 3). * p < 0.05, ** p < 0.01, *** p < 0.001 vs control, # p < 0.05, ## p < 0.01, ### p < 0.001 vs PDGF-BB. 5 represents 5 µM, 10 represents 10 µM
Index in PubMed under a CC BY license. PMID: 39696593
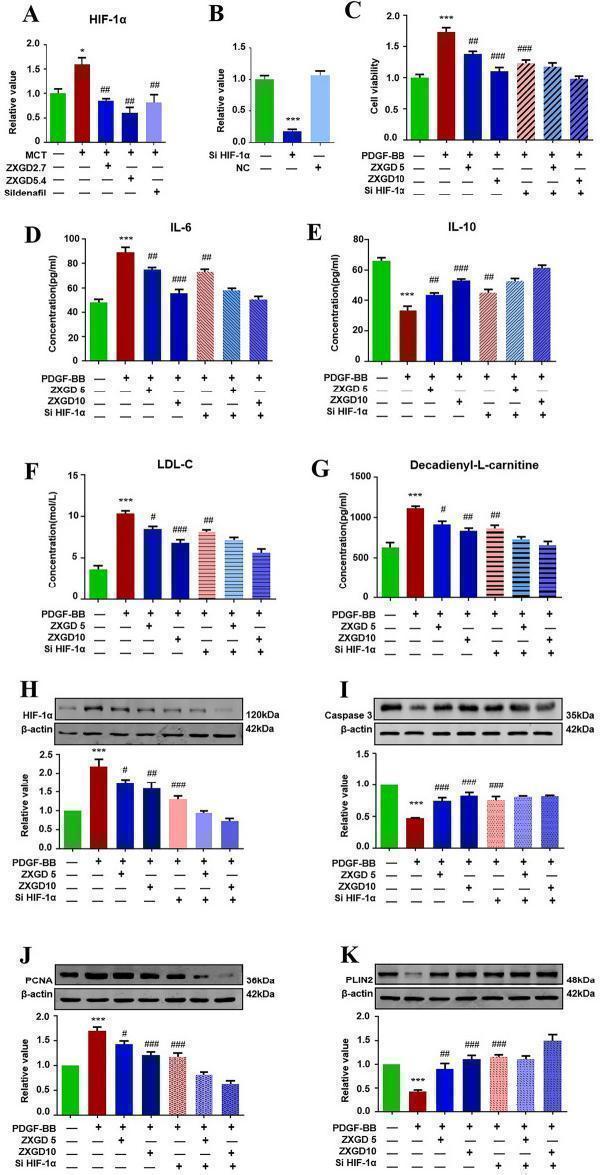
Click image to see more details
ZXGD modulated HIF-1α mediated pulmonary vascular remodeling. A Expression level of HIF-1α in lung tissue of rats was tested with PCR (n = 3). B Expression level of HIF-1α in PASMCs transfected with siRNA (n = 6). C Effects of ZXGD on viability of PASMCs transfected with HIF-1α siRNA was examined by MTT (n = 6). D – G Levels of IL-6, IL-10, LDL-C and decadienyl- l -carnitine in PASMCs transfected with HIF-1α siRNA. H – K Expression level of HIF-1α, Caspase-3, PCNA, PLIN2 in PASMCs transfected with HIF-1α siRNA (n ≥ 3). * p < 0.05, *** p < 0.001 vs control, # p < 0.05, ## p < 0.01, ### p < 0.001 vs PH or PDGF-BB
Index in PubMed under a CC BY license. PMID: 39696593
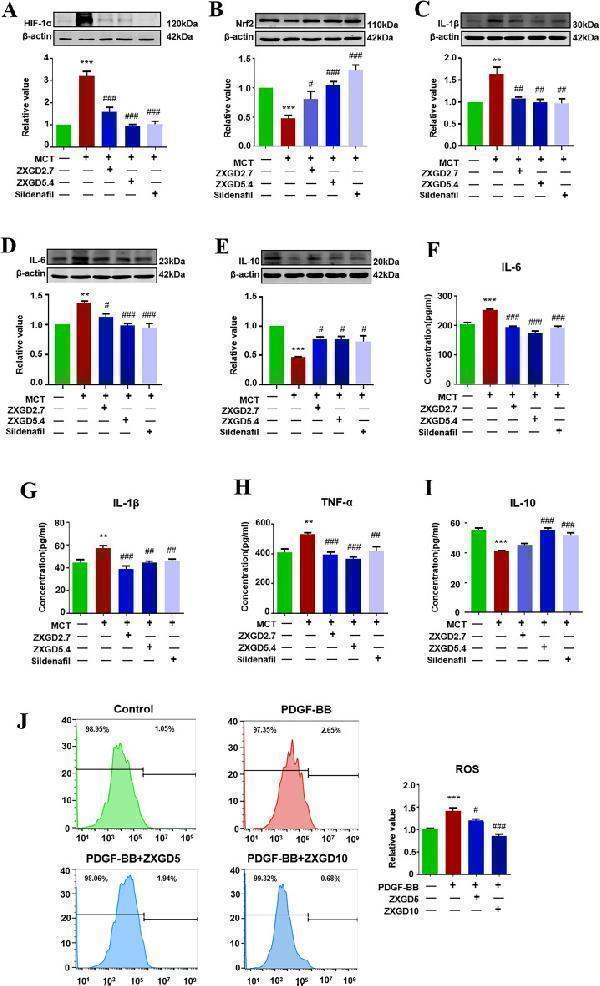
Click image to see more details
ZXGD inhibited hypoxia, oxidative stress and inflammation in rats with PH and PASMCs induced with PDGF-BB. A – E Expression of HIF-1α, Nrf2, IL-1β, IL-6 and IL-10 in lung tissue of rats were detected using Western blot (n = 3 or 6). F – I Concentration of IL-6, IL-1β, TNF-α and IL-10 in lung tissue of rats were examined by ELISA (n = 6). J Level of ROS in PASMCs was assessed with flow cytometry, and statistical data was obtained. ** p < 0.01, *** p < 0.001 vs control, # p < 0.05, ## p < 0.01, ### p < 0.001 vs PH or PDGF-BB
Index in PubMed under a CC BY license. PMID: 39696593
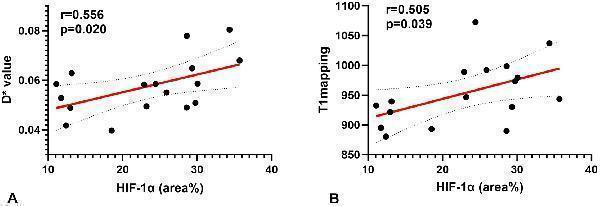
Click image to see more details
Correlations for the hypoxia score (HIF-1α) with the pseudo-diffusion coefficient (D*) and the T1 mapping. (A) HIF-1α correlated positively with the D* ( r = 0.556, p = 0.020). (B) HIF-1α correlated positively with T1 mapping ( r = 0.505, p = 0.039).
Index in PubMed under a CC BY license. PMID: 39906347

Click image to see more details
Effects of XLW on HIF1α and PTGS2 expressions in rat extraocular muscle tissues. ( A ) The immunohistochemical slice of HIF1α, ( B ) HIF1α immunohistochemical quantitative analysis, ( C ) WB of PTGS2 levels, and ( D ) quantification of the WB in PTGS2. ** p < 0.01, **** p < 0.0001 versus NC group; # p < 0.05, ## p < 0.01 versus HT group; n = 6 or 8
Index in PubMed under a CC BY license. PMID: 40696458
Click image to see more details
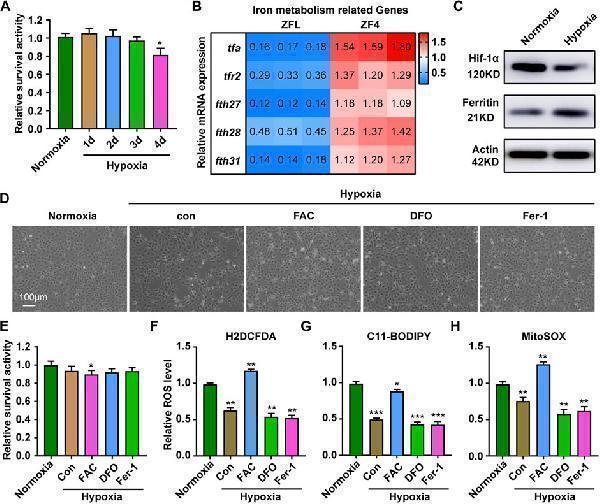
Response of ZF4 cells to hypoxic stress. (A) Cell Viability was analyzed with PrestoBlue™ HS Cell Viability Regent under normoxia or hypoxia treated for 1, 2, 3 and 4 d. (B) The mRNA expression of iron absorption and storage gene in ZFL cells was quantified by real-time RT–PCR under normoxia and hypoxia for 3 days. (C) Western blot analysis of Hif-1α and Ferritin expression in ZF4 cells cultured under normoxia and hypoxia for 3 days. (D) The microscope analysis of morphology changes in ZF4 cells under normoxia or hypoxia treated with or without FAC (2.5 mM), DFO (10 µM) and Fer-1 (2.5 µM) for 3 days. (E) Cell Viability was analyzed with PrestoBlue™ HS Cell Viability Regent under normoxia or hypoxia treated with or without FAC (2.5 mM), DFO (10 µM) and Fer-1 (2.5 µM). (F–H) Analysis of changes in general ROS (F) , mitochondrial-derived ROS (G) and lipid peroxidation (H) levels in cells with H2DCFDA, MitoSOX and C11-BODIPY probe under normoxia and hypoxia for 3 days. Cells under hypoxic stress were rescued with FAC (2.5 mM), DFO (10 µM) and Fer-1 (2.5 µM). Normoxia was used as a control group for significance analysis. Error bars, mean ± s.d., n = 3 (biological replicates).
Index in PubMed under a CC BY license. PMID: 36091397
Click image to see more details
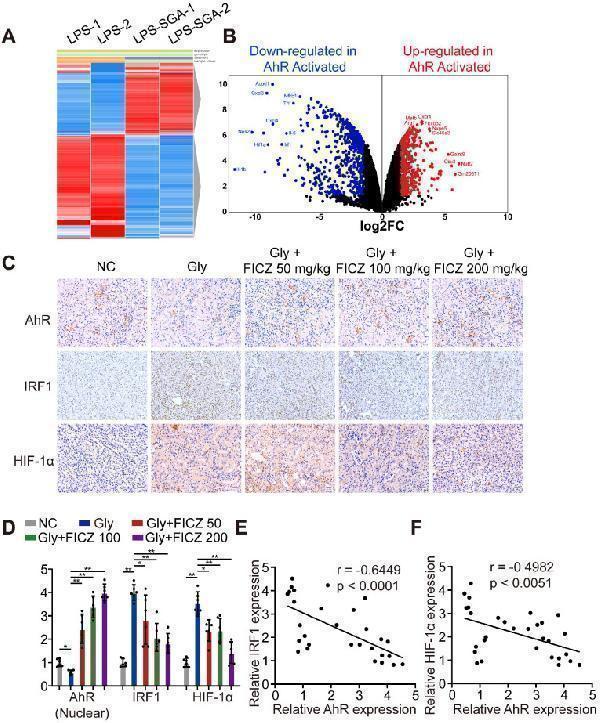
AhR significantly suppressed IRF1 and HIF-1α expression in a murine CaOx nephrocalcinosis model. (A) RNA-seq heatmap showing significantly altered mRNAs in SGA-treated BMDMs. (B) Volcano plots showing mRNA transcripts that were differentially expressed between LPS-treated and SGA-treated BMDMs. Significantly downregulated and upregulated mRNAs are shown in green and red, respectively, whereas genes that were not significantly changed are shown in black. (C) IHC staining for AhR, IRF1, and HIF-1α in the kidneys of FICZ-treated mice with CaOx nephrocalcinosis (200×; scale bar: 20 µm). (D) qRT-PCR was used to assess AhR, IRF1, and HIF-1α expression in kidney samples from FICZ-treated mice (n = 6) with CaOx nephrocalcinosis compared to kidney samples from model mice. (E, F) Pearson's correlation coefficient analysis (n = 30) of the expression levels of AhR and IRF1 (E) or HIF-1α (F). Each dot represents an individual animal. *P < 0.05; **P < 0.01, as assessed via one-way ANOVA (D).
Index in PubMed under a CC BY license. PMID: 33204326
Click image to see more details
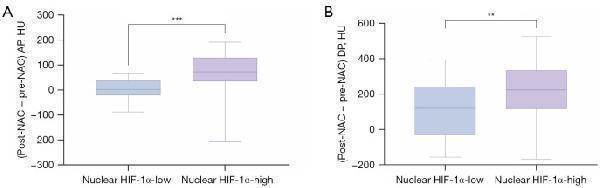
Comparison of CT values between the nuclear HIF-1α-high and -low expression PDAC patients following NAC. ***, P<0.001; **, P=0.003. CT, computed tomography; HIF-1α, hypoxia-inducible factor-1-alpha; PDAC, pancreatic ductal adenocarcinoma; NAC, neoadjuvant chemotherapy; AP, arterial phase; DP, delayed phase; HU, c.
Index in PubMed under a CC BY license. PMID: 39839013
Click image to see more details
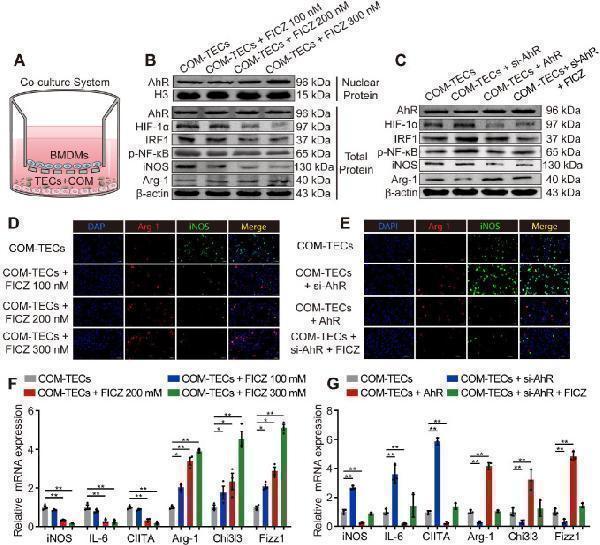
AhR suppressed IRF1 and HIF-1α to attenuate CaOx crystal-stimulated M1 macrophage polarization in vitro . (A) BMDMs and COM-treated TECs co-culture model. (B, C) Western blotting analysis was used to detect AhR, HIF-1α, IRF1, NF-κB p65, iNOS, and Arg-1 expression after FICZ treatment and the upregulation or downregulation of AhR in BMDMs. β-actin served as a normalization control. (D, E) iNOS (M1 macrophage marker, green) and Arg-1 (M2 macrophage marker, red) distribution in BMDMs were detected by immunofluorescence (200×; scale bar: 20 µm). (F, G) qRT-PCR analysis of iNOS, IL-6, CIITA, Arg-1, Chi3l3 and Fizz1 expression to further determine polarization state of BMDM. The data are shown as the means ± SD of triplicate experiments. *P < 0.05; **P < 0.01, as assessed via one-way ANOVA (F, G).
Index in PubMed under a CC BY license. PMID: 33204326
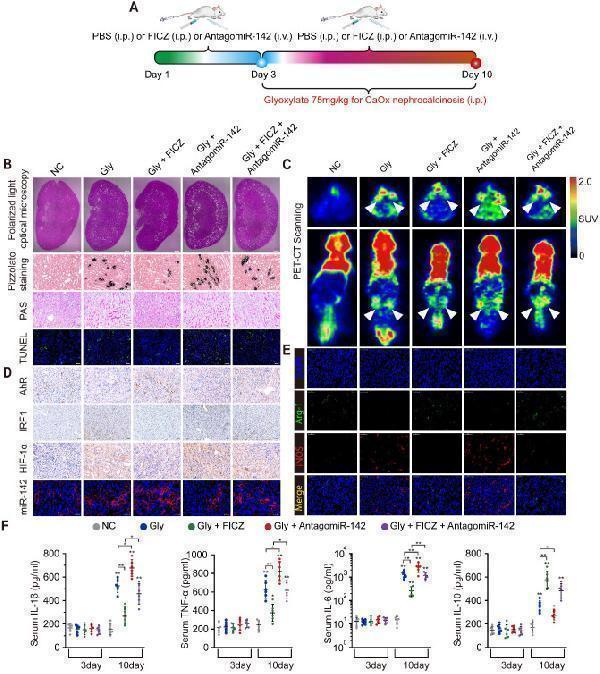
Click image to see more details
AhR activation suppressed the deposition of CaOx crystal and CaOx nephrocalcinosis-mediated kidney inflammation and injury through the AhR-miR-142a-IRF1/HIF-1α axis in vivo . (A) Experimental overview. (B) The deposition of renal CaOx crystal in FICZ- and/or antagomiR-142a-treated mice was assessed via polarized light optical microscopy (20×; scale bar: 500 µm). Pizzolato staining was employed as a means of detecting these CaOx crystal in corticomedullary tissue, while PAS was utilized to evaluate injury to TECs (200×; scale bar: 20 µm), and TUNEL staining was employed to assess renal TECs death (200×; scale bar: 50 µm). (C) PET-CT scanning was employed as a means of assessing renal inflammation state in CaOx nephrocalcinosis mice. (D) IHC was used to analyse AhR, IRF1, and HIF-1α expression, and FISH was used to detect miR-142a expression in renal tissue (200×; scale bar: 20 µm). (E) iNOS (M1 macrophage marker, red) and Arg-1 (M2 macrophage marker, green) distributions in renal tissues were detected by immunofluorescence (200×; scale bar: 50 µm). (F) On days 3 and 10, the serum pro-inflammatory IL-1β, TNF-α, and IL-6 levels and the anti-inflammatory IL-10 levels were measured by ELISA. n = 6 per group. *P < 0.05; **P < 0.01, as assessed via one-way ANOVA (F).
Index in PubMed under a CC BY license. PMID: 33204326
Specific Publications For Anti-HIF-1-alpha/HIF1A Antibody Picoband® (PB9253)
Loading publications
Recommended Resources
Here are featured tools and databases that you might find useful.
- Boster's Pathways Library
- Protein Databases
- Bioscience Research Protocol Resources
- Data Processing & Analysis Software
- Photo Editing Software
- Scientific Literature Resources
- Research Paper Management Tools
- Molecular Biology Software
- Primer Design Tools
- Bioinformatics Tools
- Phylogenetic Tree Analysis
Customer Reviews
Have you used Anti-HIF-1-alpha/HIF1A Antibody Picoband®?
Share your experimental results or join a short interview to earn up to $1,000 in product credits or other rewards.
1 Reviews For Anti-HIF-1-alpha/HIF1A Antibody Picoband®
The antibody shows a clear target band, correct positioning, minimal background, and overall good results.
Excellent

| SKU | PB9253 |
|---|---|
| Application | Western Blot |
| Sample | mouse 4T1 cells |
| Sample Processing Description | normal 4T1 cells, LPS-stimulated 4T1 cells, cells treated with low, medium, or high doses of the drug, and cells treated with a positive control drug. |
| Other Reagents | RIPA lysis buffer, Protease inhibitor, Electrophoresis buffer, Transfer buffer, Blocking buffer |
| Primary Antibody | HIF-1-alpha/HIF1A Antibody Picoband® |
| Primary Incubation | 1:2500, overnight at 4 ℃ |
| Secondary Antibody | HRP Goat Anti-Rabbit IgG |
| Secondary Incubation | 1:10000, 1 hour in room temperature |
| Detection | Substrate: ECL, Imaging system:ChemiDoc MP |
| Results Summary | With equal loading amounts, the HIF1A protein levels showed clear differences among the groups: the blank group had the lowest level, the model group the highest, and in the drug-treated groups, the levels decreased sequentially from low, medium, to high dose. The positive control group was comparable to the high-dose group. |
Bolan Guan, Harbin Medical University
Verified customer
Submitted 2025-12-01
Customer Q&As
Have a question?
Find answers in Q&As, reviews.
Can't find your answer?
Submit your question
18 Customer Q&As for Anti-HIF-1-alpha/HIF1A Antibody Picoband®
Question
Our lab used your anti-HIF-1-alpha/HIF1A antibody for WB on liver last year. I am using rat, and We intend to use the antibody for IHC next. My lab would like examining liver as well as glial tumor in our next experiment. Could give a recommendation on which antibody would work the best for IHC?
Verified Customer
Verified customer
Asked: 2020-04-16
Answer
I have checked the website and datasheets of our anti-HIF-1-alpha/HIF1A antibody and I see that PB9253 has been validated on rat in both WB and IHC. Thus PB9253 should work for your application. Our Boster satisfaction guarantee will cover this product for IHC in rat even if the specific tissue type has not been validated. We do have a comprehensive range of products for IHC detection and you can check out our website bosterbio.com to find out more information about them.
Boster Scientific Support
Answered: 2020-04-16
Question
Please see the WB image, lot number and protocol we used for visceral pleura using anti-HIF-1-alpha/HIF1A antibody PB9253. Please let me know if you require anything else.
Verified Customer
Verified customer
Asked: 2020-01-27
Answer
Thank you very much for the data. Our lab team are working to resolve this as quickly as possible, and we appreciate your patience and understanding! You have provided everything we needed. Please let me know if there is anything you need in the meantime.
Boster Scientific Support
Answered: 2020-01-27
Question
We appreciate helping with my inquiry over the phone. Here are the WB image, lot number and protocol we used for visceral pleura using anti-HIF-1-alpha/HIF1A antibody PB9253. Let me know if you need anything else.
Verified Customer
Verified customer
Asked: 2020-01-13
Answer
I appreciate the data. You have provided everything we needed. Our lab team are working to resolve your inquiry as quickly as possible, and we appreciate your patience and understanding! Please let me know if there is anything you need in the meantime.
Boster Scientific Support
Answered: 2020-01-13
Question
you antibody to test anti-HIF-1-alpha/HIF1A antibody PB9253 on human visceral pleura for research purposes, then I may be interested in using anti-HIF-1-alpha/HIF1A antibody PB9253 for diagnostic purposes as well. Is the antibody suitable for diagnostic purposes?
Verified Customer
Verified customer
Asked: 2020-01-09
Answer
The products we sell, including anti-HIF-1-alpha/HIF1A antibody PB9253, are only intended for research use. They would not be suitable for use in diagnostic work. If you have the means to develop a product into diagnostic use, and are interested in collaborating with us and develop our product into an IVD product, please contact us for more discussions.
Boster Scientific Support
Answered: 2020-01-09
Question
We want using your anti-HIF-1-alpha/HIF1A antibody for positive regulation of blood vessel endothelial cell migration studies. Has this antibody been tested with western blotting on mouse intestine tissue? We would like to see some validation images before ordering.
Verified Customer
Verified customer
Asked: 2019-08-09
Answer
Thank you for your inquiry. This PB9253 anti-HIF-1-alpha/HIF1A antibody is validated on mouse intestine tissue, intestinal cancer tissue, rat intestine tissue. It is guaranteed to work for IHC, WB in human, mouse, rat. Our Boster guarantee will cover your intended experiment even if the sample type has not been be directly tested.
Boster Scientific Support
Answered: 2019-08-09
Question
What specific lysis buffer and blocking buffer was used in the PB9253 Antibody protocol?
Verified customer
Asked: 2019-07-15
Answer
The Lysis buffer used in the Anti-HIF-1-alpha/HIF1A Antibody Picoband PB9253 protocol was RIPA lysis buffer. The Blocking buffer was 5% Non-fat Milk/ TBS.
Boster Scientific Support
Answered: 2019-07-15
Question
Do you have a WB testing with cobalt chloride treatment of the cells prior to lysing/testing for PB9253 Antibody?
Verified customer
Asked: 2019-07-10
Answer
Our lab did not perform a WB test on PON1 Paraoxonase-1 Human Recombinant Protein PROTP27169 with cobalt chloride treatment of the cells prior to lysing/testing.
Boster Scientific Support
Answered: 2019-07-10
Question
We have observed staining in human glial tumor. Are there any suggestions? Is anti-HIF-1-alpha/HIF1A antibody supposed to stain glial tumor positively?
Verified Customer
Verified customer
Asked: 2019-06-17
Answer
Based on literature glial tumor does express HIF1A. Based on Uniprot.org, HIF1A is expressed in visceral pleura, hepatoma, glial tumor, liver, choriocarcinoma placenta, among other tissues. Regarding which tissues have HIF1A expression, here are a few articles citing expression in various tissues:
Choriocarcinoma, and Placenta, Pubmed ID: 15489334
Hepatoma, Pubmed ID: 9079689
Liver, Pubmed ID: 12508121
Boster Scientific Support
Answered: 2019-06-17
Question
Is a blocking peptide available for product anti-HIF-1-alpha/HIF1A antibody (PB9253)?
Verified Customer
Verified customer
Asked: 2019-05-13
Answer
We do provide the blocking peptide for product anti-HIF-1-alpha/HIF1A antibody (PB9253). If you would like to place an order for it please contact support@bosterbio.com and make a special request.
Boster Scientific Support
Answered: 2019-05-13
Question
I see that the anti-HIF-1-alpha/HIF1A antibody PB9253 works with IHC, what is the protocol used to produce the result images on the product page?
Verified Customer
Verified customer
Asked: 2018-06-15
Answer
You can find protocols for IHC on the "support/technical resources" section of our navigation menu. If you have any further questions, please send an email to support@bosterbio.com
Boster Scientific Support
Answered: 2018-06-15
Question
Is this PB9253 anti-HIF-1-alpha/HIF1A antibody reactive to the isotypes of HIF1A?
Verified Customer
Verified customer
Asked: 2018-05-29
Answer
The immunogen of PB9253 anti-HIF-1-alpha/HIF1A antibody is A synthetic peptide corresponding to a sequence at the C-terminal of human HIF-1-alpha (703-732aa EEELNPKILALQNAQRKRKMEHDGSLFQAV), different from the related mouse and rat sequences by three amino acids. Could you tell me which isotype you are interested in so I can help see if the immunogen is part of this isotype?
Boster Scientific Support
Answered: 2018-05-29
Question
Will anti-HIF-1-alpha/HIF1A antibody PB9253 work for IHC with visceral pleura?
Verified Customer
Verified customer
Asked: 2018-01-30
Answer
According to the expression profile of visceral pleura, HIF1A is highly expressed in visceral pleura. So, it is likely that anti-HIF-1-alpha/HIF1A antibody PB9253 will work for IHC with visceral pleura.
Boster Scientific Support
Answered: 2018-01-30
Question
Does PB9253 anti-HIF-1-alpha/HIF1A antibody work on parafin embedded sections? If so, which fixation method do you recommend we use (PFA, paraformaldehyde, other)?
Verified Customer
Verified customer
Asked: 2017-09-13
Answer
You can see on the product datasheet, PB9253 anti-HIF-1-alpha/HIF1A antibody as been tested on IHC. It is best to use PFA for fixation because it has better tissue penetration ability. PFA needs to be prepared fresh before use. Long term stored PFA turns into formalin, as the PFA molecules congregate and become formalin.
Boster Scientific Support
Answered: 2017-09-13
Question
Is there a BSA free version of anti-HIF-1-alpha/HIF1A antibody PB9253 available?
D. Parker
Verified customer
Asked: 2017-08-11
Answer
I appreciate your recent telephone inquiry. I can confirm that some lots of this anti-HIF-1-alpha/HIF1A antibody PB9253 are BSA free. For now, these lots are available and we can make a BSA free formula for you free of charge. It will take 3 extra days to prepare. If you require this antibody BSA free again in future, please do not hesitate to contact me and I will be pleased to check which lots we have in stock that are BSA free.
Boster Scientific Support
Answered: 2017-08-11
Question
We were content with the WB result of your anti-HIF-1-alpha/HIF1A antibody. However we have seen positive staining in glial tumor cytoplasm. using this antibody. Is that expected? Could you tell me where is HIF1A supposed to be expressed?
J. Bhatt
Verified customer
Asked: 2015-04-20
Answer
From what I have seen in literature, glial tumor does express HIF1A. Generally HIF1A expresses in cytoplasm. Regarding which tissues have HIF1A expression, here are a few articles citing expression in various tissues:
Choriocarcinoma, and Placenta, Pubmed ID: 15489334
Hepatoma, Pubmed ID: 9079689
Liver, Pubmed ID: 12508121
Boster Scientific Support
Answered: 2015-04-20
Question
I was wanting to use your anti-HIF-1-alpha/HIF1A antibody for IHC for human visceral pleura on frozen tissues, but I want to know if it has been validated for this particular application. Has this antibody been validated and is this antibody a good choice for human visceral pleura identification?
P. Evans
Verified customer
Asked: 2014-01-24
Answer
As indicated on the product datasheet, PB9253 anti-HIF-1-alpha/HIF1A antibody has been tested for IHC, WB on human, mouse, rat tissues. We have an innovator award program that if you test this antibody and show it works in human visceral pleura in IHC-frozen, you can get your next antibody for free.
Boster Scientific Support
Answered: 2014-01-24
Question
I have a question about product PB9253, anti-HIF-1-alpha/HIF1A antibody. I was wondering if it would be possible to conjugate this antibody with biotin. I would need it to be without BSA or sodium azide. I am planning on using a buffer exchange of sodium azide with PBS only. Would there be problems for me to conjugate the antibody and store it in -20 degrees in small aliquots?
E. Mitchell
Verified customer
Asked: 2013-06-27
Answer
We do not advise storing this antibody with PBS buffer only in -20 degrees. If you want to store it in -20 degrees it is best to add some cryoprotectant like glycerol. If you want carrier free PB9253 anti-HIF-1-alpha/HIF1A antibody, we can provide it to you in a special formula with trehalose and/or glycerol. These molecules will not interfere with conjugation chemistry and provide a good level of protection for the antibody from degradation. Please be sure to specify this in your purchase order.
Boster Scientific Support
Answered: 2013-06-27
Question
We are currently using anti-HIF-1-alpha/HIF1A antibody PB9253 for human tissue, and we are happy with the IHC results. The species of reactivity given in the datasheet says human, mouse, rat. Is it true that the antibody can work on monkey tissues as well?
C. Thomas
Verified customer
Asked: 2013-01-22
Answer
The anti-HIF-1-alpha/HIF1A antibody (PB9253) has not been validated for cross reactivity specifically with monkey tissues, but there is a good chance of cross reactivity. We have an innovator award program that if you test this antibody and show it works in monkey you can get your next antibody for free. Please contact me if I can help you with anything.
Boster Scientific Support
Answered: 2013-01-22





