Product Info Summary
| SKU: | PB9887 |
|---|---|
| Size: | 100 μg/vial |
| Reactive Species: | Human, Mouse, Rat |
| Host: | Rabbit |
| Application: | WB |
Customers Who Bought This Also Bought
Product info
Product Name
Anti-Scn1a Antibody Picoband®
SKU/Catalog Number
PB9887
PB0932 is an alternative SKU for this antibody, used in previous lots.
Size
100 μg/vial
Form
Lyophilized
Description
Boster Bio Anti-Scn1a Antibody Picoband® catalog # PB9887. Tested in WB applications. This antibody reacts with Human, Mouse, Rat. The brand Picoband indicates this is a premium antibody that guarantees superior quality, high affinity, and strong signals with minimal background in Western blot applications. Only our best-performing antibodies are designated as Picoband, ensuring unmatched performance.
Storage & Handling
Store at -20˚C for one year from date of receipt. After reconstitution, at 4˚C for one month. It can also be aliquotted and stored frozen at -20˚C for six months. Avoid repeated freeze-thaw cycles.
Cite This Product
Anti-Scn1a Antibody Picoband® (Boster Biological Technology, Pleasanton CA, USA, Catalog # PB9887)
Host
Rabbit
Contents
Each vial contains 4mg Trehalose, 0.9mg NaCl and 0.2mg Na2HPO4.
Clonality
Polyclonal
Isotype
Rabbit IgG
Immunogen
A synthetic peptide corresponding to a sequence at the C-terminus of human Scn1a, identical to the related rat sequence.
Cross-reactivity
No cross-reactivity with other proteins.
Reactive Species
PB9887 is reactive to SCN1A in Human, Mouse, Rat
Observed Molecular Weight
250 kDa
Calculated molecular weight
229.0 kDa
Background of SCN1A
Nav1.1, also known as the sodium channel, voltage-gated, type I, alpha subunit (SCN1A), is a protein which in humans is encoded by the SCN1A gene. Voltage-dependent sodium channels are heteromeric complexes that regulate sodium exchange between intracellular and extracellular spaces and are essential for the generation and propagation of action potentials in muscle cells and neurons. Each sodium channel is composed of a large pore-forming, glycosylated alpha subunit and two smaller beta subunits. This gene encodes a sodium channel alpha subunit, which has four homologous domains, each of which contains six transmembrane regions. Allelic variants of this gene are associated with generalized epilepsy with febrile seizures and epileptic encephalopathy. Alternative splicing results in multiple transcript variants. The RefSeq Project has decided to create four representative RefSeq records. Three of the transcript variants are supported by experimental evidence and the fourth contains alternate 5' untranslated exons, the exact combination of which have not been experimentally confirmed for the full-length transcript.
Antibody Validation
Boster validates all antibodies on WB, IHC, ICC, Immunofluorescence, and ELISA with known positive control and negative samples to ensure specificity and high affinity, including thorough antibody incubations.
Application & Images
Applications
PB9887 is guaranteed for WB Boster Guarantee
Recommend Dilution
| Application | Dilution | Species |
|---|---|---|
| Western blot | 0.1-0.5μg/ml | Human, Mouse, Rat |
Tested application
Suggested blocking solution with 5% non-fat milk or BSA; (*)Recommended protein loading: 20-40 µg per lane
Validation Images & Assay Conditions

Click image to see more details
Western blot analysis of Scn1a using anti-Scn1a antibody (PB9887).
Electrophoresis was performed on a 5-20% SDS-PAGE gel at 70V (Stacking gel) / 90V (Resolving gel) for 2-3 hours. The sample well of each lane was loaded with 30 ug of sample under reducing conditions.
Lane 1: rat brain tissue lysates,
Lane 2: mouse brain tissue lysates,
Lane 3: U87 whole cell lysates.
After electrophoresis, proteins were transferred to a nitrocellulose membrane at 150 mA for 50-90 minutes. Blocked the membrane with 5% non-fat milk/TBS for 1.5 hour at RT. The membrane was incubated with rabbit anti-Scn1a antigen affinity purified polyclonal antibody (Catalog # PB9887) at 0.5 μg/mL overnight at 4°C, then washed with TBS-0.1%Tween 3 times with 5 minutes each and probed with a goat anti-rabbit IgG-HRP secondary antibody at a dilution of 1:5000 for 1.5 hour at RT. The signal is developed using an Enhanced Chemiluminescent detection (ECL) kit (Catalog # EK1002) with Tanon 5200 system. A specific band was detected for Scn1a at approximately 250 kDa. The expected band size for Scn1a is at 229 kDa.
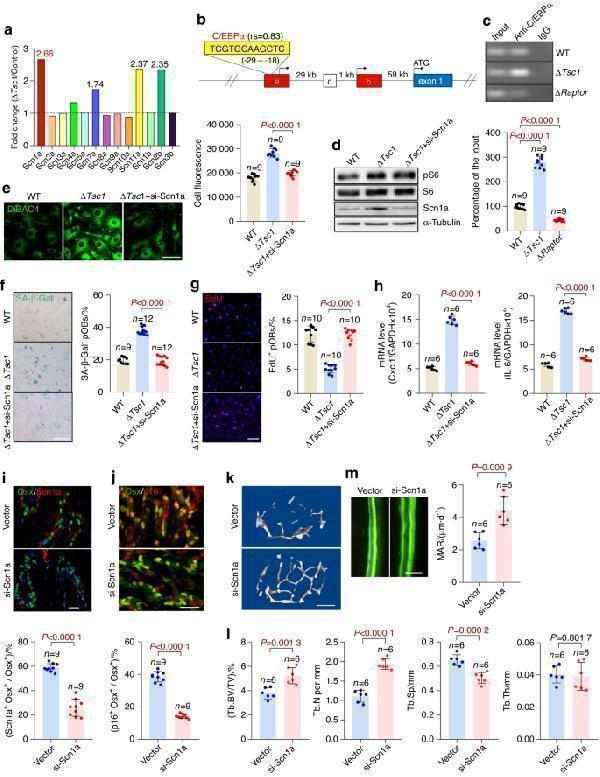
Click image to see more details
The sodium channel Scn1a mediates plasma membrane depolarization in senescent preosteoblasts. a mRNA expression of voltage-sensitive sodium channels in the Δ Tsc1 and control calvarial preosteoblasts indicated by our previous global mRNA expression profile (GSE74781). b Binding site of C/EBPα in the 5′-UE of Scn1a (yellow box) predicted by the JASPAR database ( ). rs: JASPAR relative scores, which are defined as 1 for the maximum-likelihood sequence. Boxes represent exons: blue coding exons, red noncoding exons conserved between humans and mice, white noncoding exons identified in either human or mouse transcripts. Noncoding exons are named alphabetically, and the first coding exon (exon 1) of the Scn1a gene is indicated. Genomic distances between exons are indicated. c Binding of C/EBPα to the 5′-UE sequence of Scn1a in vivo, determined by a ChIP assay using the Δ Tsc1 and Δ Raptor cells and anti-C/EBPα antibody or IgG. The ChIP samples were then subjected to qPCR with the Scn1a 5′-UE primers. The percentage of the input of the sample by using the anti-C/EBPα antibody in the wild-type cells was normalized to 100. The Δ Tsc1 osteoblasts were transfected with Scn1a siRNA and subjected to ( d ) Scn1a detection with western blotting, ( e ) measurement of relative plasma membrane potential with DiBAC4 dye, ( f ) SA-β-gal staining and quantification of the proportion of SA-β-gal-positive cells, ( g ) immunostaining of EdU and quantitative analysis of EdU + cells relative to total cells, and ( h ) qPCR analysis of IL-6 and Cxcl1 mRNA. Double immunostaining of Osx plus Scn1a ( i ) and Osx plus p16 ( j ) in the tibias of the 18-month-old Δ Tsc1 mice injected with adenovirus encoding si-Scn1a for 1 month. Double positively stained cells were quantified. Representative μCT images ( k ) and quantification of trabecular bone ( l ) in the mice. ( m ) Representative images of calcein labels and quantification of the mineral apposition rate (MAR) in femurs from the 18-month-old Δ Tsc1 mice receiving si-Scn1a. Scale bars: 50 μm in e , g , m ; 100 μm in i , j ; and 500 μm in k . Data are shown as the mean ± SD. The numbers of samples ( n ) are indicated in each figure panel. P values were determined with two-tailed Student’s t test for single comparisons
Index in PubMed under a CC BY license. PMID: 35256591
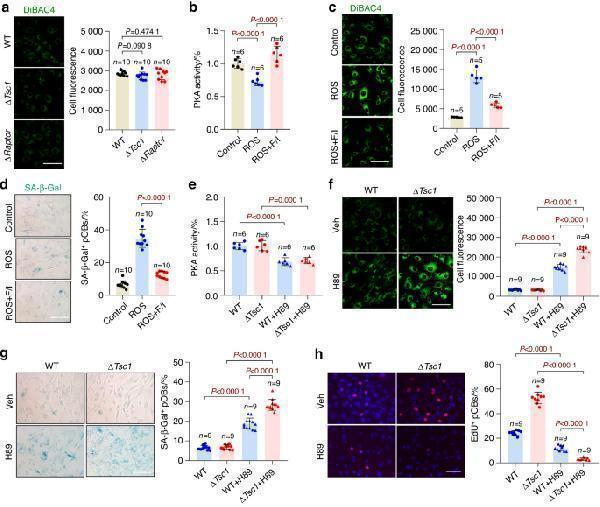
Click image to see more details
Prosenescent stress activates Scn1a by inhibiting PKA. a Replicative wild-type (WT), Δ Tsc1 and Δ Raptor calvarial osteoblasts were incubated with the fluorescent DiBAC4 dye and photographed under a confocal microscope. Scale bar, 50 μm. Relative plasma membrane potentials were measured. ROS-induced senescent wild-type preosteoblasts were treated with F/I (forskolin + IBMx, PKA activator) or left untreated. The cells were subjected to ( b ) measurement of total cellular PKA activities, ( c ) DiBAC4 staining and measurement of relative plasma membrane potentials. Scale bar, 50 μm. d SA-β-gal staining of cells in b and quantification of the proportion of SA-β-gal-positive cells in each population. Scale bar, 100 μm. Replicative WT and Δ Tsc1 osteoblasts were treated with H-89 (PKA inhibitor) and were subjected to ( e ) measurement of total cellular PKA activities, ( f ) DiBAC4 staining and measurement of relative plasma membrane potentials. Scale bar, 50 μm. g SA-β-gal staining of cells in e and quantification of the proportion of SA-β-gal-positive cells in each population. Scale bar, 100 μm. h Immunostaining of EdU in cells in e and quantitative analysis of EdU + cells relative to total cells. Scale bar, 100 μm. Data are shown as the mean ± SD. The numbers of samples ( n ) are indicated in each figure panel. P values were determined by two-tailed Student’s t test for single comparisons
Index in PubMed under a CC BY license. PMID: 35256591
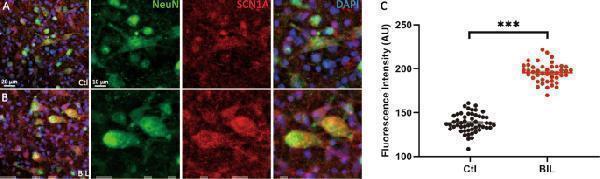
Click image to see more details
BIL elevated the expression level of SCN1A in MVN neurons. a , b Immunofluorescence labeling images of nuclei (blue), NeuN (green), and SCN1A (red) of control ( a ) and BIL group ( b ). Neurons with co-labeling of NeuN and SCN1A were chosen as AOI, and the mean fluorescence intensities of SCN1A from AOI were quantified in artificial units (AUs). c Statistical comparison showing larger MISCN1A in the BIL group, implicating elevated expression level of Nav1.1 after BIL treatment. *** p < 0.001, independent-samples t- test.
Index in PubMed under a CC BY license. PMID: 31601780
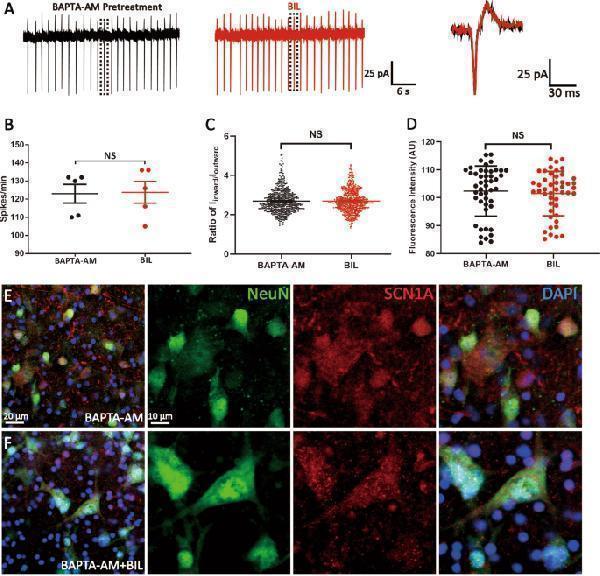
Click image to see more details
Ca 2+ was required for BIL-induced upregulation of activity and level of VGSCs. a An example recording showing that neither the frequency of spontaneous firings nor the spike waveform of a MVN neuron was altered in slices preincubated in BAPTA-AM (40 μM, 30 min). b , c Statistical results showing no significant change in spike frequency, ratio of I inward / I outward . d – f MI SCN1A was not statistically different between control and BIL groups ( d ) as exemplified by the immunofluorescence staining of nuclei (blue), NeuN (green), and SCN1A (red) in control ( e ) and BIL groups ( f ). NS no statistical difference, independent-samples t test.
Index in PubMed under a CC BY license. PMID: 31601780
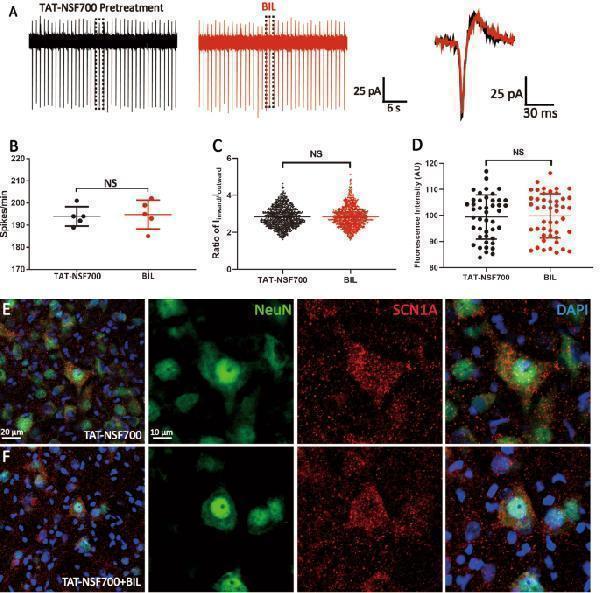
Click image to see more details
Blocking exocytosis with TAT-NSF700 precluded the effects of BIL on MVN neurons. a – c Example recording traces ( a ) showing that when the slice was preincubated with TAT-NSF700 (5 μM, 30 min), a permeable thrombin-induced exocytosis inhibitor, neither the discharge frequency of MVN neuron ( b ) nor the ratio of I inward / I outward ( c ) was altered by BIL (3 μM, 3 min). d–f MI SCN1A was not statistically different between control and BIL groups ( d ) in slices pretreated with TAT-NSF700, as illustrated by the immunofluorescence images comparing staining of nuclei (blue), NeuN (green), and SCN1A (red) in control ( e ) and BIL group ( f ). NS no statistical difference, independent-samples t- test.
Index in PubMed under a CC BY license. PMID: 31601780
Specific Publications For Anti-Scn1a Antibody Picoband® (PB9887)
Loading publications
Recommended Resources
Here are featured tools and databases that you might find useful.
- Boster's Pathways Library
- Protein Databases
- Bioscience Research Protocol Resources
- Data Processing & Analysis Software
- Photo Editing Software
- Scientific Literature Resources
- Research Paper Management Tools
- Molecular Biology Software
- Primer Design Tools
- Bioinformatics Tools
- Phylogenetic Tree Analysis
Customer Reviews
Have you used Anti-Scn1a Antibody Picoband®?
Share your experimental results or join a short interview to earn up to $1,000 in product credits or other rewards.
0 Reviews For Anti-Scn1a Antibody Picoband®
Customer Q&As
Have a question?
Find answers in Q&As, reviews.
Can't find your answer?
Submit your question
1 Customer Q&As for Anti-Scn1a Antibody Picoband®
Question
We are currently using anti-Scn1a antibody PB9887 for mouse tissue, and we are content with the WB results. The species of reactivity given in the datasheet says human, mouse, rat. Is it possible that the antibody can work on bovine tissues as well?
J. Parker
Verified customer
Asked: 2017-11-27
Answer
The anti-Scn1a antibody (PB9887) has not been tested for cross reactivity specifically with bovine tissues, though there is a good chance of cross reactivity. We have an innovator award program that if you test this antibody and show it works in bovine you can get your next antibody for free. Please contact me if I can help you with anything.
Boster Scientific Support
Answered: 2017-11-27





