This website uses cookies to ensure you get the best experience on our website.
- Table of Contents
2 Citations 5 Q&As
1 Citations 10 Q&As
1 Citations 5 Q&As
Facts about Insulin-like growth factor-binding protein 3.
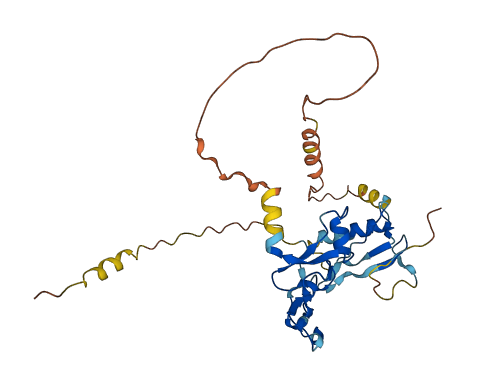
Also exhibits IGF-independent antiproliferative and apoptotic effects mediated by its receptor TMEM219/IGFBP-3R. .
| Human | |
|---|---|
| Gene Name: | IGFBP3 |
| Uniprot: | P17936 |
| Entrez: | 3486 |

| Belongs to: |
|---|
| No superfamily |

acid stable subunit of the 140 K IGF complex; binding protein 29; binding protein 53; growth hormone-dependent binding protein; IBP-3; IBP3BP-53; IGF-binding protein 3; IGFBP3; IGFBP-3; insulin-like growth factor binding protein 3; insulin-like growth factor-binding protein 3
Mass (kDA):
31.674 kDA

| Human | |
|---|---|
| Location: | 7p12.3 |
| Sequence: | 7; NC_000007.14 (45912245..45921272, complement) |
Expressed by most tissues. Present in plasma.
Secreted.





PMID: 2464130 by Wood W.I., et al. Cloning and expression of the growth hormone-dependent insulin-like growth factor-binding protein.
PMID: 1695633 by Cubbage M.L., et al. Insulin-like growth factor binding protein-3. Organization of the human chromosomal gene and demonstration of promoter activity.
*More publications can be found for each product on its corresponding product page