Product Info Summary
| SKU: | RP1022 |
|---|---|
| Size: | 100 μg/vial |
| Reactive Species: | Human, Mouse, Rat |
| Host: | Rabbit |
| Application: | ELISA, Flow Cytometry, IHC, WB |
Customers Who Bought This Also Bought
Product info
Product Name
Anti-Transferrin/TF Antibody Picoband®
SKU/Catalog Number
RP1022
Size
100 μg/vial
Form
Lyophilized
Description
Boster Bio Anti-Transferrin/TF Antibody catalog # RP1022. Tested in ELISA, Flow Cytometry, IHC, WB applications. This antibody reacts with Human, Mouse, Rat. The brand Picoband indicates this is a premium antibody that guarantees superior quality, high affinity, and strong signals with minimal background in Western blot applications. Only our best-performing antibodies are designated as Picoband, ensuring unmatched performance.
Storage & Handling
Store at -20˚C for one year from date of receipt. After reconstitution, at 4˚C for one month. It can also be aliquotted and stored frozen at -20˚C for six months. Avoid repeated freeze-thaw cycles.
Cite This Product
Anti-Transferrin/TF Antibody Picoband® (Boster Biological Technology, Pleasanton CA, USA, Catalog # RP1022)
Host
Rabbit
Contents
Each vial contains 4 mg Trehalose, 0.9 mg NaCl and 0.2 mg Na2HPO4.
Clonality
Polyclonal
Isotype
Rabbit IgG
Immunogen
Oryza sativa-derived human Transferrin recombinant protein (Position: V20-P698).
Cross-reactivity
No cross-reactivity with other proteins
Reactive Species
RP1022 is reactive to TF in Human, Mouse, Rat
Observed Molecular Weight
77 kDa
Calculated molecular weight
77.1 kDa
Background of TF
Transferrins are iron-binding blood plasma glycoproteins that control the level of free iron in biological fluids. In humans, it is encoded by the TF gene. In humans, transferrin consists of a polypeptide chain containing 679 amino acids. The protein is composed of alpha helices and beta sheets to form two domains. The N- and C- terminal sequences are represented by globular lobes and between the two lobes is an iron-binding site. Transferrin is a glycoprotein that binds iron very tightly but reversibly. Although iron bound to transferrin is less than 0.1% (4 mg) of the total body iron, it is the most important iron pool, with the highest rate of turnover (25 mg/24 h). Transferrin has a molecular weight of around 80 kDa and contains 2 specific high-affinity Fe (III) binding sites. The affinity of transferrin for Fe (III) is extremely high (1023 M−1 at pH 7.4) but decreases progressively with decreasing pH below neutrality.
Antibody Validation
Boster validates all antibodies on WB, IHC, ICC, Immunofluorescence, and ELISA with known positive control and negative samples to ensure specificity and high affinity, including thorough antibody incubations.
Application & Images
Applications
RP1022 is guaranteed for ELISA, Flow Cytometry, IHC, WB Boster Guarantee
Assay Dilutions Recommendation
The recommendations below provide a starting point for assay optimization. The actual working concentration varies and should be decided by the user.
Western blot, 0.1-0.5μg/ml, Human, Mouse, Rat
Immunohistochemistry (Paraffin-embedded Section), 2-5μg/ml, Mouse
Flow Cytometry(Fixed), 1-3 μg/1x106 cells, Human
ELISA, 0.1-0.5μg/ml, -
Positive Control
WB: human placenta tissue, human HCCT, human HCCP, human HepG2 whole cell, rat liver tissue, rat RH-35 whole cell, mouse liver tissue, mouse Hepa1/6 whole cell
IHC: mouse liver tissuebr>FCM: HepG2 cell
Validation Images & Assay Conditions
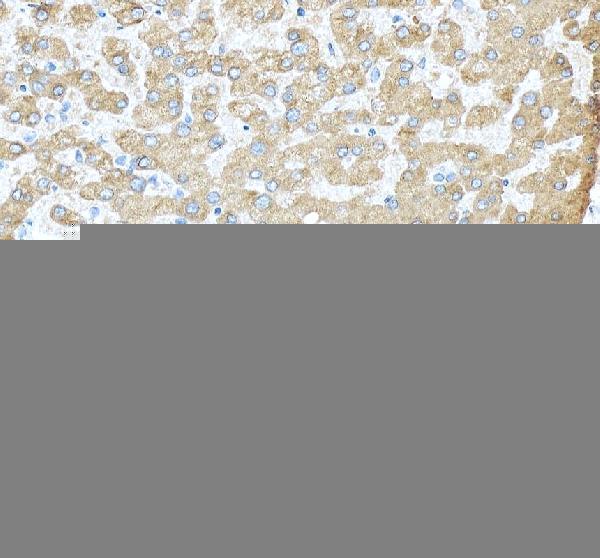
Click image to see more details
IHC analysis of Transferrin/TF using anti-Transferrin/TF antibody (RP1022).
Transferrin/TF was detected in a paraffin-embedded section of human liver tissue. Heat mediated antigen retrieval was performed in EDTA buffer (pH 8.0, epitope retrieval solution). The tissue section was blocked with 10% goat serum. The tissue section was then incubated with 2 μg/ml rabbit anti-Transferrin/TF Antibody (RP1022) overnight at 4°C. Peroxidase Conjugated Goat Anti-rabbit IgG was used as secondary antibody and incubated for 30 minutes at 37°C. The tissue section was developed using HRP Conjugated Rabbit IgG Super Vision Assay Kit (Catalog # SV0002) with DAB as the chromogen.
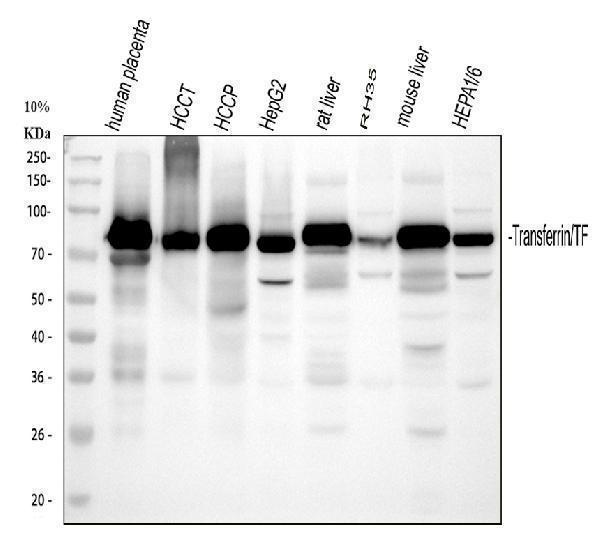
Click image to see more details
Western blot analysis of Transferrin/TF using anti-Transferrin/TF antibody (RP1022).
Electrophoresis was performed on a 5-20% SDS-PAGE gel at 70V (Stacking gel) / 90V (Resolving gel) for 2-3 hours. The sample well of each lane was loaded with 30 ug of sample under reducing conditions.
Lane 1: human placenta tissue lysates,
Lane 2: human hepatocellular carcinoma tumor tissue (HCCT) lysates,
Lane 3: human hepatocellular carcinoma paracancerous tissue (HCCP) lysates,
Lane 4: human HepG2 whole cell lysates,
Lane 5: rat liver tissue lysates,
Lane 6: rat RH-35 whole cell lysates,
Lane 7: mouse liver tissue lysates,
Lane 8: mouse Hepa1/6 whole cell lysates.
After electrophoresis, proteins were transferred to a nitrocellulose membrane at 150 mA for 50-90 minutes. Blocked the membrane with 5% non-fat milk/TBS for 1.5 hour at RT. The membrane was incubated with rabbit anti-Transferrin/TF antigen affinity purified polyclonal antibody (Catalog # RP1022) at 0.5 μg/mL overnight at 4°C, then washed with TBS-0.1%Tween 3 times with 5 minutes each and probed with a goat anti-rabbit IgG-HRP secondary antibody at a dilution of 1:5000 for 1.5 hour at RT. The signal is developed using an Enhanced Chemiluminescent detection (ECL) kit (Catalog # EK1002) with Tanon 5200 system. A specific band was detected for Transferrin/TF at approximately 77 kDa. The expected band size for Transferrin/TF is at 77 kDa.
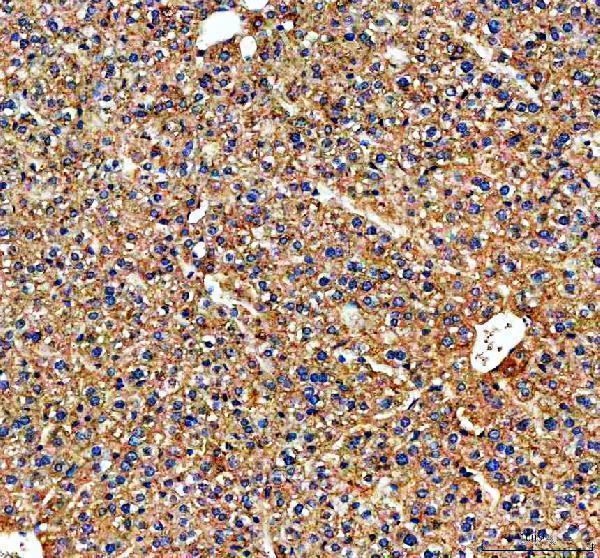
Click image to see more details
IHC analysis of Transferrin/TF using anti-Transferrin/TF antibody (RP1022).
Transferrin/TF was detected in a paraffin-embedded section of mouse liver tissue. Heat mediated antigen retrieval was performed in EDTA buffer (pH 8.0, epitope retrieval solution). The tissue section was blocked with 10% goat serum. The tissue section was then incubated with 2 μg/ml rabbit anti-Transferrin/TF Antibody (RP1022) overnight at 4°C. Peroxidase Conjugated Goat Anti-rabbit IgG was used as secondary antibody and incubated for 30 minutes at 37°C. The tissue section was developed using HRP Conjugated Rabbit IgG Super Vision Assay Kit (Catalog # SV0002) with DAB as the chromogen.
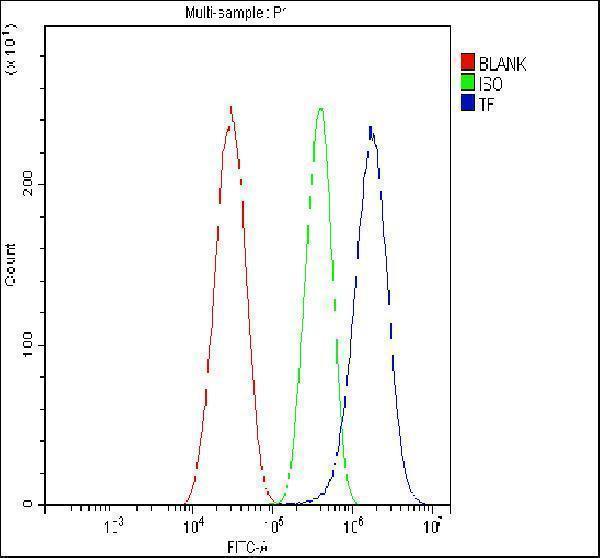
Click image to see more details
Flow Cytometry analysis of HepG2 cells using anti-Transferrin/TF antibody (RP1022).
Overlay histogram showing HepG2 cells stained with RP1022 (Blue line). The cells were fixed with 4% paraformaldehyde and blocked with 10% normal goat serum. And then incubated with rabbit anti-Transferrin/TF Antibody (RP1022, 1 μg/1x106 cells) for 30 min at 20°C. DyLight®488 conjugated goat anti-rabbit IgG (BA1127, 5-10 μg/1x106 cells) was used as secondary antibody for 30 minutes at 20°C. Isotype control antibody (Green line) was rabbit IgG (1 μg/1x106) used under the same conditions. Unlabelled sample without incubation with primary antibody and secondary antibody (Red line) was used as a blank control.
Specific Publications For Anti-Transferrin/TF Antibody Picoband® (RP1022)
Loading publications
Recommended Resources
Here are featured tools and databases that you might find useful.
- Boster's Pathways Library
- Protein Databases
- Bioscience Research Protocol Resources
- Data Processing & Analysis Software
- Photo Editing Software
- Scientific Literature Resources
- Research Paper Management Tools
- Molecular Biology Software
- Primer Design Tools
- Bioinformatics Tools
- Phylogenetic Tree Analysis
Customer Reviews
Have you used Anti-Transferrin/TF Antibody Picoband®?
Share your experimental results or join a short interview to earn up to $1,000 in product credits or other rewards.
0 Reviews For Anti-Transferrin/TF Antibody Picoband®
Customer Q&As
Have a question?
Find answers in Q&As, reviews.
Can't find your answer?
Submit your question
16 Customer Q&As for Anti-Transferrin/TF Antibody Picoband®
Question
I see that the anti-Transferrin/TF antibody RP1022 works with IHC, what is the protocol used to produce the result images on the product page?
Verified Customer
Verified customer
Asked: 2020-04-06
Answer
You can find protocols for IHC on the "support/technical resources" section of our navigation menu. If you have any further questions, please send an email to support@bosterbio.com
Boster Scientific Support
Answered: 2020-04-06
Question
Is this RP1022 anti-Transferrin/TF antibody reactive to the isotypes of TF?
Verified Customer
Verified customer
Asked: 2020-03-18
Answer
The immunogen of RP1022 anti-Transferrin/TF antibody is Oryza sativa-derived human Transferrin recombinant protein(Position: V20-P698). Could you tell me which isotype you are interested in so I can help see if the immunogen is part of this isotype?
Boster Scientific Support
Answered: 2020-03-18
Question
We have tried in the past anti-Transferrin/TF antibody for ELISA on liver in a previous experiment. I am using human, and We are going to use the antibody for IHC next. I am looking for examining liver as well as inferior vagus x ganglion in our next experiment. Could you please give me some suggestion on which antibody would work the best for IHC?
Verified Customer
Verified customer
Asked: 2020-01-31
Answer
I looked at the website and datasheets of our anti-Transferrin/TF antibody and it appears that RP1022 has been validated on human in both ELISA and IHC. Thus RP1022 should work for your application. Our Boster satisfaction guarantee will cover this product for IHC in human even if the specific tissue type has not been validated. We do have a comprehensive range of products for IHC detection and you can check out our website bosterbio.com to find out more information about them.
Boster Scientific Support
Answered: 2020-01-31
Question
Will RP1022 anti-Transferrin/TF antibody work on parafin embedded sections? If so, which fixation method do you recommend we use (PFA, paraformaldehyde, other)?
Verified Customer
Verified customer
Asked: 2020-01-29
Answer
It shows on the product datasheet, RP1022 anti-Transferrin/TF antibody as been validated on IHC. It is best to use PFA for fixation because it has better tissue penetration ability. PFA needs to be prepared fresh before use. Long term stored PFA turns into formalin, as the PFA molecules congregate and become formalin.
Boster Scientific Support
Answered: 2020-01-29
Question
Would anti-Transferrin/TF antibody RP1022 work for IHC with bile?
Verified Customer
Verified customer
Asked: 2020-01-01
Answer
According to the expression profile of bile, TF is highly expressed in bile. So, it is likely that anti-Transferrin/TF antibody RP1022 will work for IHC with bile.
Boster Scientific Support
Answered: 2020-01-01
Question
Will anti-Transferrin/TF antibody RP1022 work on zebrafish ELISA with saliva?
Verified Customer
Verified customer
Asked: 2019-11-14
Answer
Our lab technicians have not validated anti-Transferrin/TF antibody RP1022 on zebrafish. You can run a BLAST between zebrafish and the immunogen sequence of anti-Transferrin/TF antibody RP1022 to see if they may cross-react. If the sequence homology is close, then you can perform a pilot test. Keep in mind that since we have not validated zebrafish samples, this use of the antibody is not covered by our guarantee. However we have an innovator award program that if you test this antibody and show it works in zebrafish saliva in ELISA, you can get your next antibody for free.
Boster Scientific Support
Answered: 2019-11-14
Question
I have a question about product RP1022, anti-Transferrin/TF antibody. I was wondering if it would be possible to conjugate this antibody with biotin. I would need it to be without BSA or sodium azide. I am planning on using a buffer exchange of sodium azide with PBS only. Would there be problems for me to conjugate the antibody and store it in -20 degrees in small aliquots?
Verified Customer
Verified customer
Asked: 2019-11-06
Answer
We do not recommend storing this antibody with PBS buffer only in -20 degrees. If you want to store it in -20 degrees it is best to add some cryoprotectant like glycerol. If you want carrier free RP1022 anti-Transferrin/TF antibody, we can provide it to you in a special formula with trehalose and/or glycerol. These molecules will not interfere with conjugation chemistry and provide a good level of protection for the antibody from degradation. Please be sure to specify this in your purchase order.
Boster Scientific Support
Answered: 2019-11-06
Question
Thank you for helping with my inquiry over the phone. Here are the WB image, lot number and protocol we used for bile using anti-Transferrin/TF antibody RP1022. Let me know if you need anything else.
Verified Customer
Verified customer
Asked: 2019-10-24
Answer
We appreciate the data. You have provided everything we needed. Our lab team are working to resolve your inquiry as quickly as possible, and we appreciate your patience and understanding! Please let me know if there is anything you need in the meantime.
Boster Scientific Support
Answered: 2019-10-24
Question
Is a blocking peptide available for product anti-Transferrin/TF antibody (RP1022)?
Verified Customer
Verified customer
Asked: 2018-02-21
Answer
We do provide the blocking peptide for product anti-Transferrin/TF antibody (RP1022). If you would like to place an order for it please contact support@bosterbio.com and make a special request.
Boster Scientific Support
Answered: 2018-02-21
Question
We have been able to see staining in human liver. What should we do? Is anti-Transferrin/TF antibody supposed to stain liver positively?
C. Johnson
Verified customer
Asked: 2018-01-29
Answer
From literature liver does express TF. From Uniprot.org, TF is expressed in inferior vagus x ganglion, liver, brain, fetal liver, brain, cajal-retzius cell fetal brain cortex, heart, bile, plasma, saliva, cerebrospinal fluid, among other tissues. Regarding which tissues have TF expression, here are a few articles citing expression in various tissues:
Bile, Pubmed ID: 15084671
Brain, Pubmed ID: 9272172, 15489334
Cerebrospinal fluid, Pubmed ID: 19838169
Heart, Pubmed ID: 7498159
Liver, Pubmed ID: 1809186, 19159218, 24275569
Plasma, Pubmed ID: 14760718, 16335952
Saliva, Pubmed ID: 16740002
Boster Scientific Support
Answered: 2018-01-29
Question
See attached the WB image, lot number and protocol we used for bile using anti-Transferrin/TF antibody RP1022. Please let me know if you require anything else.
Verified Customer
Verified customer
Asked: 2018-01-11
Answer
Thank you very much for the data. Our lab team are working to resolve this as quickly as possible, and we appreciate your patience and understanding! You have provided everything we needed. Please let me know if there is anything you need in the meantime.
Boster Scientific Support
Answered: 2018-01-11
Question
We are currently using anti-Transferrin/TF antibody RP1022 for human tissue, and we are content with the WB results. The species of reactivity given in the datasheet says human. Is it true that the antibody can work on monkey tissues as well?
Verified Customer
Verified customer
Asked: 2017-12-26
Answer
The anti-Transferrin/TF antibody (RP1022) has not been tested for cross reactivity specifically with monkey tissues, but there is a good chance of cross reactivity. We have an innovator award program that if you test this antibody and show it works in monkey you can get your next antibody for free. Please contact me if I can help you with anything.
Boster Scientific Support
Answered: 2017-12-26
Question
Do you have a BSA free version of anti-Transferrin/TF antibody RP1022 available?
Verified Customer
Verified customer
Asked: 2017-12-07
Answer
We appreciate your recent telephone inquiry. I can confirm that some lots of this anti-Transferrin/TF antibody RP1022 are BSA free. For now, these lots are available and we can make a BSA free formula for you free of charge. It will take 3 extra days to prepare. If you require this antibody BSA free again in future, please do not hesitate to contact me and I will be pleased to check which lots we have in stock that are BSA free.
Boster Scientific Support
Answered: 2017-12-07
Question
I am looking for to test anti-Transferrin/TF antibody RP1022 on human bile for research purposes, then I may be interested in using anti-Transferrin/TF antibody RP1022 for diagnostic purposes as well. Is the antibody suitable for diagnostic purposes?
P. Johnson
Verified customer
Asked: 2016-10-06
Answer
The products we sell, including anti-Transferrin/TF antibody RP1022, are only intended for research use. They would not be suitable for use in diagnostic work. If you have the means to develop a product into diagnostic use, and are interested in collaborating with us and develop our product into an IVD product, please contact us for more discussions.
Boster Scientific Support
Answered: 2016-10-06
Question
I was wanting to use your anti-Transferrin/TF antibody for IHC for human bile on frozen tissues, but I want to know if it has been validated for this particular application. Has this antibody been validated and is this antibody a good choice for human bile identification?
J. Carter
Verified customer
Asked: 2016-01-18
Answer
As indicated on the product datasheet, RP1022 anti-Transferrin/TF antibody has been tested for ELISA, IHC, WB on human tissues. We have an innovator award program that if you test this antibody and show it works in human bile in IHC-frozen, you can get your next antibody for free.
Boster Scientific Support
Answered: 2016-01-18
Question
My team were well pleased with the WB result of your anti-Transferrin/TF antibody. However we have been able to see positive staining in liver secreted. using this antibody. Is that expected? Could you tell me where is TF supposed to be expressed?
M. Collins
Verified customer
Asked: 2013-12-12
Answer
From literature, liver does express TF. Generally TF expresses in secreted. Regarding which tissues have TF expression, here are a few articles citing expression in various tissues:
Bile, Pubmed ID: 15084671
Brain, Pubmed ID: 9272172, 15489334
Cerebrospinal fluid, Pubmed ID: 19838169
Heart, Pubmed ID: 7498159
Liver, Pubmed ID: 1809186, 19159218, 24275569
Plasma, Pubmed ID: 14760718, 16335952
Saliva, Pubmed ID: 16740002
Boster Scientific Support
Answered: 2013-12-12





