Product Info Summary
| SKU: | PA1054 |
|---|---|
| Size: | 100 μg/vial |
| Reactive Species: | Human, Mouse, Rat |
| Host: | Rabbit |
| Application: | Flow Cytometry, IF, IHC, WB |
Customers Who Bought This Also Bought
Product info
Product Name
Anti-Myeloperoxidase/MPO Antibody Picoband®
SKU/Catalog Number
PA1054
BA0544 is an alternative SKU for this antibody, used in previous lots.
Size
100 μg/vial
Form
Lyophilized
Description
MPO is a neutrophil granule enzyme central to oxidative host defense, generating hypohalous acids and serving as a widely used marker of neutrophil activation/degranulation in inflammatory disease contexts. Assay context: Flow Cytometry/IF for leukocyte phenotyping; IHC for tissue neutrophil infiltration; Western blot for lysate confirmation. Often paired with other neutrophil effector proteases such as ELANE (neutrophil elastase), and can be cross-checked against an alternate MPO reagent such as MPO (PB9057) in panel design. "
Storage & Handling
Store at -20˚C for one year from date of receipt. After reconstitution, at 4˚C for one month. It can also be aliquotted and stored frozen at -20˚C for six months. Avoid repeated freeze-thaw cycles.
Cite This Product
Anti-Myeloperoxidase/MPO Antibody Picoband® (Boster Biological Technology, Pleasanton CA, USA, Catalog # PA1054)
Host
Rabbit
Contents
Each vial contains antibody formulated with stabilizing components, 0.9mg NaCl, 0.2mg Na2HPO4, 0.05mg Thimerosal, 0.05mg NaN3.
*This antibody is supplied in a stabilized formulation.
Compatibility with conjugation reactions depends on the chemistry of the conjugation method used.
For conjugation methods that are not compatible with the stabilizing components present in this formulation, a carrier-free antibody format is required.
Clonality
Polyclonal
Isotype
Rabbit IgG
Immunogen
A synthetic peptide corresponding to a sequence at the C-terminus of human MPO, different from the related mouse and rat sequences by one amino acid.
Cross-reactivity
No cross-reactivity with other proteins
Reactive Species
PA1054 is reactive to MPO in Human, Mouse, Rat
Observed Molecular Weight
60 kDa
Calculated molecular weight
83.9 kDa
Background of MPO
Myeloperoxidase (MPO) is a mammalian phagocyte hemoprotein though to primarily mediate host defense reactions. It is abundantly expressed in neutrophils and secreted during their activation. Myeloperoxidase is part of the host defense system of human polymorphonuclear leukocytes, responsible for microbicidal activity against a wide range of organisms. It is located in the nucleus as well as in the cytoplasm. Intranuclear MPO may help to protect DNA against damage resulting from oxygen radicals produced during myeloid cell maturation and function.
Antibody Validation
Boster validates all antibodies on WB, IHC, ICC, Immunofluorescence, and ELISA with known positive control and negative samples to ensure specificity and high affinity, including thorough antibody incubations.
Application & Images
Applications
PA1054 is guaranteed for Flow Cytometry, IF, IHC, WB Boster Guarantee
Recommend Dilution
| Application | Dilution | Species |
|---|---|---|
| Western blot | 0.1-0.5μg/ml | Human, Mouse, Rat |
| Immunohistochemistry (Paraffin-embedded Section) | 2-5μg/ml | Human, Mouse, Rat |
| Immunofluorescence | 5 μg/ml | Human |
| Flow Cytometry (Fixed) | 1-3 μg/1x106 cells | Human |
Tested application
Suggested blocking solution with 5% non-fat milk or BSA; (*)Recommended protein loading: 20-40 µg per lane
Use TE buffer pH 9.0 for antigen retrieval; (*) citrate buffer pH 6.0 is an alternative.
Validation Images & Assay Conditions
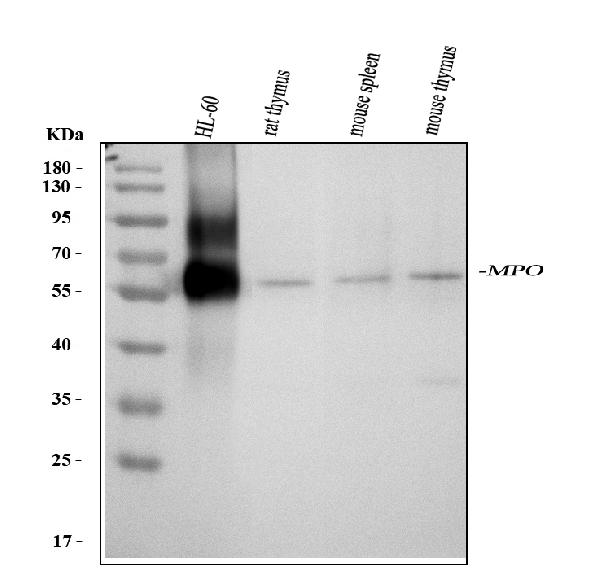
Click image to see more details
Western blot analysis of MPO using anti-MPO antibody (PA1054).
Electrophoresis was performed on a 5-20% SDS-PAGE gel at 70V (Stacking gel) / 90V (Resolving gel) for 2-3 hours. The sample well of each lane was loaded with 30 ug of sample under reducing conditions.
Lane 1: human HL-60 whole cell lysates,
Lane 2: rat thymus tissue lysates,
Lane 3: mouse spleen tissue lysates,
Lane 4: mouse thymus tissue lysates.
After electrophoresis, proteins were transferred to a nitrocellulose membrane at 150 mA for 50-90 minutes. Blocked the membrane with 5% non-fat milk/TBS for 1.5 hour at RT. The membrane was incubated with rabbit anti-MPO antigen affinity purified polyclonal antibody (Catalog # PA1054) at 0.5 μg/mL overnight at 4°C, then washed with TBS-0.1%Tween 3 times with 5 minutes each and probed with a goat anti-rabbit IgG-HRP secondary antibody at a dilution of 1:5000 for 1.5 hour at RT. The signal is developed using an Enhanced Chemiluminescent detection (ECL) kit (Catalog # EK1002) with Tanon 5200 system. A specific band was detected for MPO at approximately 60 kDa. The expected band size for MPO is at 84 kDa.
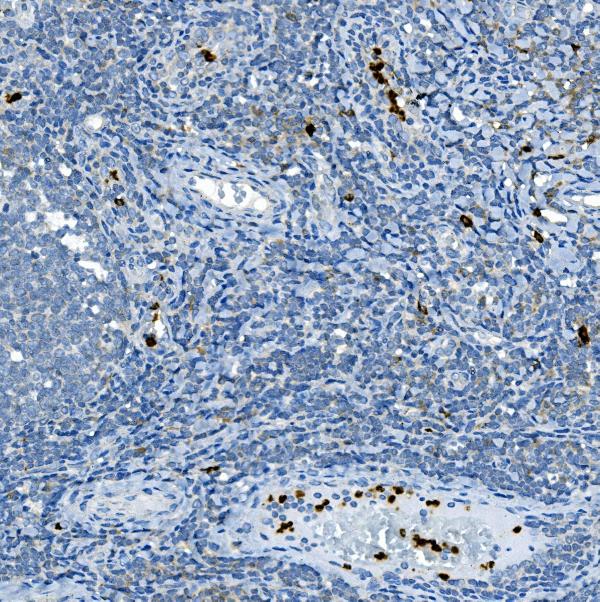
Click image to see more details
IHC analysis of MPO using anti-MPO antibody (PA1054).
MPO was detected in a paraffin-embedded section of human tonsil tissue. Heat mediated antigen retrieval was performed in EDTA buffer (pH 8.0, epitope retrieval solution). The tissue section was blocked with 10% goat serum. The tissue section was then incubated with 2 μg/ml rabbit anti-MPO Antibody (PA1054) overnight at 4°C. Peroxidase Conjugated Goat Anti-rabbit IgG was used as secondary antibody and incubated for 30 minutes at 37°C. The tissue section was developed using HRP Conjugated Rabbit IgG Super Vision Assay Kit (Catalog # SV0002) with DAB as the chromogen.
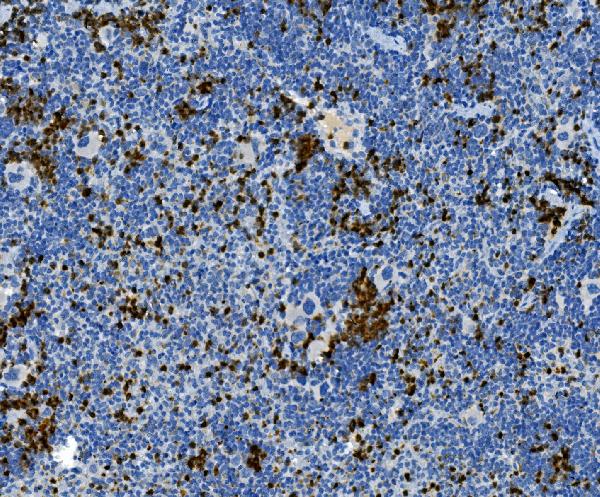
Click image to see more details
IHC analysis of MPO using anti-MPO antibody (PA1054).
MPO was detected in a paraffin-embedded section of mouse spleen tissue. Heat mediated antigen retrieval was performed in EDTA buffer (pH 8.0, epitope retrieval solution). The tissue section was blocked with 10% goat serum. The tissue section was then incubated with 2 μg/ml rabbit anti-MPO Antibody (PA1054) overnight at 4°C. Peroxidase Conjugated Goat Anti-rabbit IgG was used as secondary antibody and incubated for 30 minutes at 37°C. The tissue section was developed using HRP Conjugated Rabbit IgG Super Vision Assay Kit (Catalog # SV0002) with DAB as the chromogen.
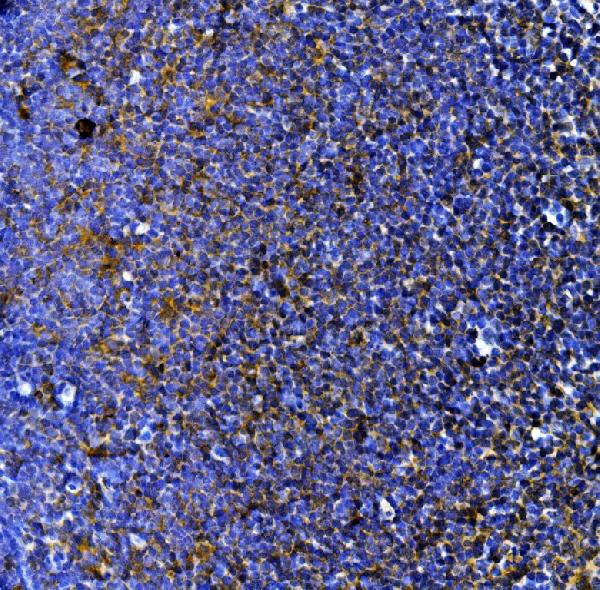
Click image to see more details
IHC analysis of MPO using anti-MPO antibody (PA1054).
MPO was detected in a paraffin-embedded section of rat lymphaden tissue. Heat mediated antigen retrieval was performed in EDTA buffer (pH 8.0, epitope retrieval solution). The tissue section was blocked with 10% goat serum. The tissue section was then incubated with 2 μg/ml rabbit anti-MPO Antibody (PA1054) overnight at 4°C. Peroxidase Conjugated Goat Anti-rabbit IgG was used as secondary antibody and incubated for 30 minutes at 37°C. The tissue section was developed using HRP Conjugated Rabbit IgG Super Vision Assay Kit (Catalog # SV0002) with DAB as the chromogen.
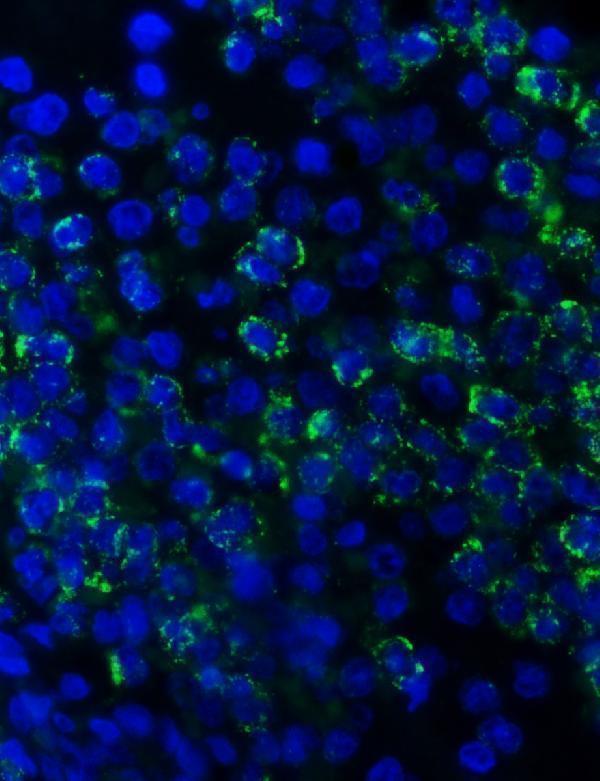
Click image to see more details
IF analysis of MPO using anti-MPO antibody (PA1054).
MPO was detected in a paraffin-embedded section of human spleen tissue. Heat mediated antigen retrieval was performed in EDTA buffer (pH 8.0, epitope retrieval solution). The tissue section was blocked with 10% goat serum. The tissue section was then incubated with 5 μg/mL rabbit anti-MPO Antibody (PA1054) overnight at 4°C. DyLight488 Conjugated Goat Anti-Rabbit IgG (BA1127) was used as secondary antibody at 1:500 dilution and incubated for 30 minutes at 37°C. The section was counterstained with DAPI. Visualize using a fluorescence microscope and filter sets appropriate for the label used.
Click image to see more details
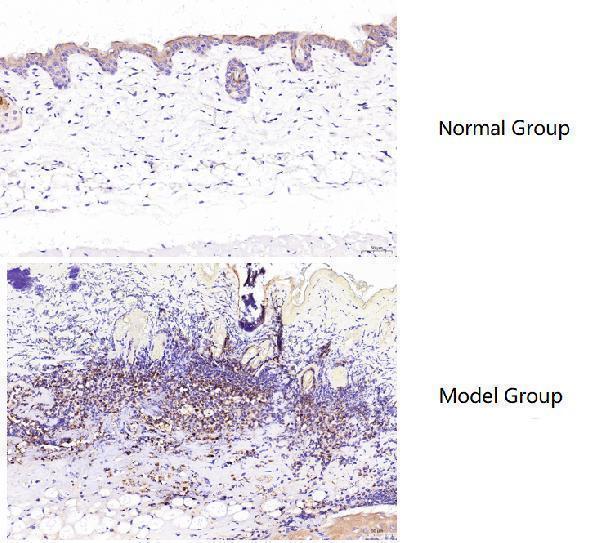
IHC analysis of MPO using anti-MPO antibody (PA1054).
MPO was detected in a paraffin-embedded section of mouse dorsal skin (normal group) and burned skin (model group) tissue. Heat mediated antigen retrieval was performed in EDTA buffer (pH 9.0, epitope retrieval solution). The tissue section was blocked with 10% goat serum. The tissue section was then incubated with 1:100 rabbit anti-MPO Antibody (PA1054) overnight at 4°C. Polymer Anti-Rabbit IgG–HRP IHC Kit was used as secondary antibody and incubated for 30 minutes at 37°C. The tissue section was developed using HRP Conjugated Rabbit IgG Super Vision Assay Kit (Catalog # SV0002) with DAB as the chromogen.
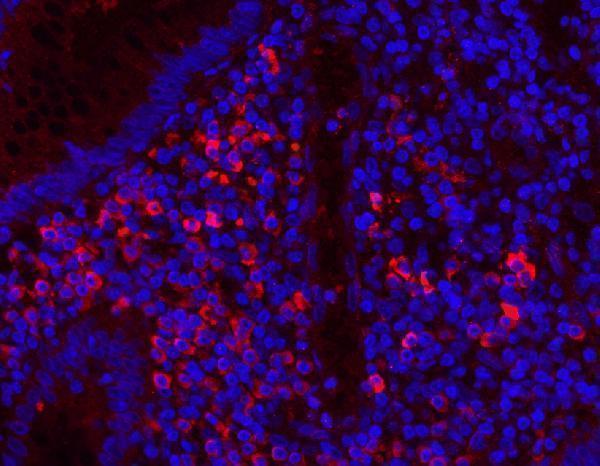
Click image to see more details
IF analysis of MPO using anti-MPO antibody (PA1054).
MPO was detected in a paraffin-embedded section of human appendicitis tissue. Heat mediated antigen retrieval was performed in EDTA buffer (pH 8.0, epitope retrieval solution). The tissue section was blocked with 10% goat serum. The tissue section was then incubated with 5 μg/mL rabbit anti-MPO Antibody (PA1054) overnight at 4°C. Cy3 Conjugated Goat Anti-Rabbit IgG (BA1032) was used as secondary antibody at 1:500 dilution and incubated for 30 minutes at 37°C. The section was counterstained with DAPI. Visualize using a fluorescence microscope and filter sets appropriate for the label used.
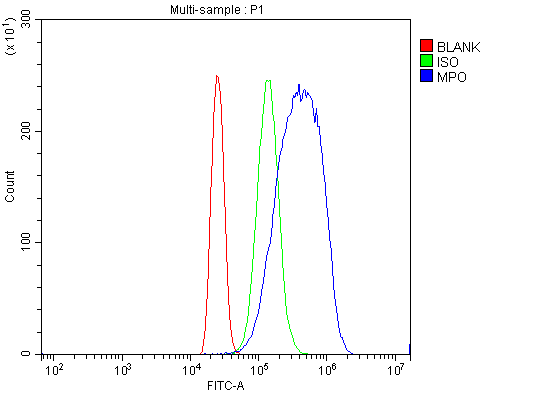
Click image to see more details
Flow Cytometry analysis of HL-60 cells using anti-MPO antibody (PA1054).
Overlay histogram showing HL-60 cells stained with PA1054 (Blue line). To facilitate intracellular staining, cells were fixed with 4% paraformaldehyde and permeabilized with permeabilization buffer. The cells were blocked with 10% normal goat serum. And then incubated with rabbit anti-MPO Antibody (PA1054, 1 μg/1x106 cells) for 30 min at 20°C. DyLight®488 conjugated goat anti-rabbit IgG (BA1127, 5-10 μg/1x106 cells) was used as secondary antibody for 30 minutes at 20°C. Isotype control antibody (Green line) was rabbit IgG (1 μg/1x106) used under the same conditions. Unlabelled sample without incubation with primary antibody and secondary antibody (Red line) was used as a blank control.
Click image to see more details
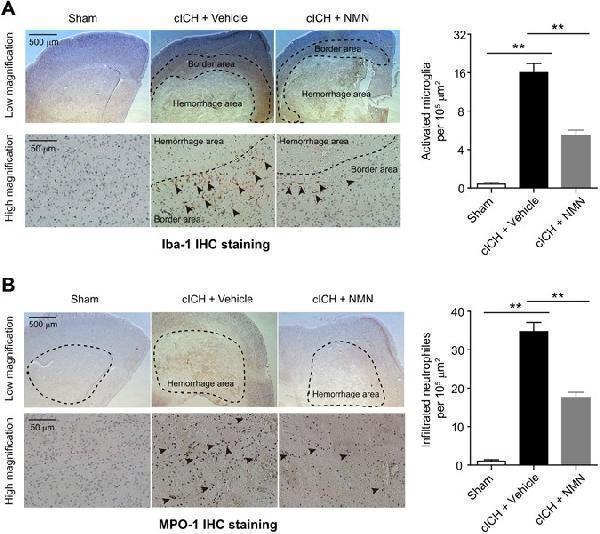
Effects of NMN on microglia activation and neutrophil infiltration in cICH model. ( A ) Representative images and quantitative analysis of Iba-1 (microglia marker) immunohistochemistry staining at 24 hours post cICH. ** P < 0.01, n = 5 per group. ( B ) Representative images and quantitative analysis of MPO-1 (neutrophil marker) immunohistochemistry staining at 24 hours post cICH. ** P < 0.01, n = 5 per group.
Index in PubMed under a CC BY license. PMID: 28386082
Click image to see more details
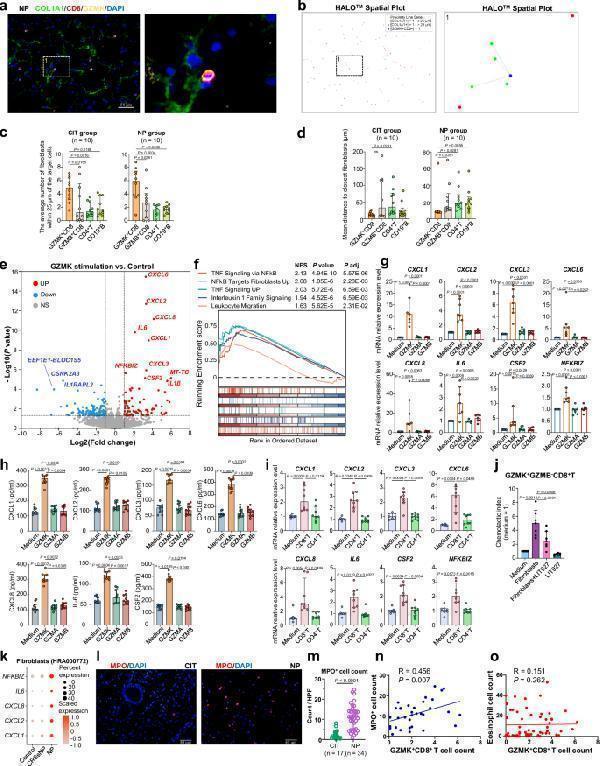
Interaction between GZMK + CD8 + T cells and fibroblasts contributes to neutrophilic inflammation in nasal polyps. a Representative immunofluorescence staining of collagen I (COL1A1, green), CD8 (red), and GZMK (yellow) in NPs. The right image shows a greater magnification of the outlined area. b Spatial distribution analysis of GZMK + CD8 + T and COL1A1 + cells in the same tissue field demonstrated in ( a ) using HALO software. c–d The number of COL1A1 + fibroblasts within a radius of 25 μm from the nuclear center of GZMK + CD8 + T, GZMB + CD8 + T, CD4 + T, or CD19 + B cells in CIT group (left, n = 10 samples) and NP group (right, n = 10 samples) ( c ). Average distance from the indicated cell types to the closest COL1A1 + fibroblasts in CIT group (left, n = 10 samples) and NP group (right, n = 10 samples) ( d ). e DEGs between NP-derived primary fibroblasts (NPDF) treated with and without recombinant human GZMK ( n = 4). Two-sided Wald test (default for DESeq2 r-package) was used for differential expression analysis utilizing standard cutoffs of |log2(fold change) | >1 and P value < 0.05. f GSEA plots showing signaling pathways enriched in the GZMK stimulated NPDF. Two-sided permutation test with Benjamini-Hochberg adjustments was used for GSEA analysis. Normalized enrichment score (NES) > 1 and adjusted P < 0.05 was considered significant. g Expression levels of selected genes in NPDF stimulated with GZMK, GZMA or GZMB ( n = 7 biological replicates). h Protein levels of chemokines and cytokines secreted by NPDF stimulated with GZMK, GZMA, or GZMB ( n = 10 biological replicates). i Gene expression levels in NPDF cultured with supernatants of NP-derived and activated CD4 + T or CD8 + T cells ( n = 8 biological replicates). j Migration of GZMK + CD8 + T cells towards NPDF in the presence or absence of CXCL12 neutraligand LIT-927 ( n = 7 biological replicates). Data are presented as mean with standard deviation; two-sided paired t test. k Expression of neutrophil chemokines and inflammatory marker in fibroblasts from the indicated group (5 controls, 5 CRSsNP, and 11 NP samples) in a public dataset (HRA000772). l Representative immunofluorescence staining of myeloperoxidase (MPO, red) positive cells in CIT and NP. Scale bar: 40 μm. m Quantification of MPO + cells in the indicated group (17 CIT and 34 NP samples). Data are presented as median with interquartile ranges; Two-sided unpaired Wilcoxon test. Spearman correlation between GZMK + CD8 + T cells and MPO + neutrophil counts ( n = 34 samples) ( n ) or eosinophil counts in NP ( n = 57 samples) ( o ). Two-sided Spearman’s rank correlations test. For ( c , d ), and ( g–i ), data are presented as median with interquartile ranges; Two-sided Friedman test with Dunn’s multiple comparisons test. CIT control inferior turbinate sample, NP nasal polyp. Source data are provided as a Source Data file.
Index in PubMed under a CC BY license. PMID: 39614076
Click image to see more details
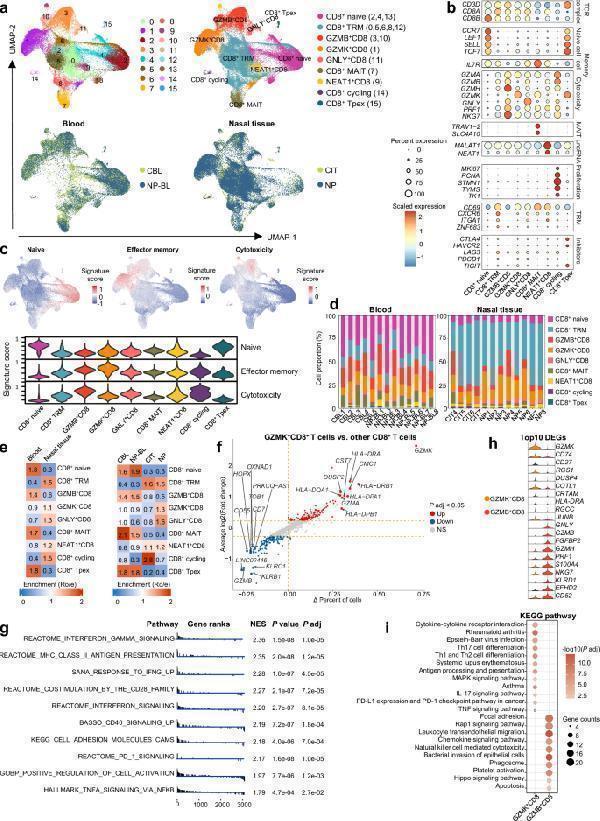
GZMK + CD8 + T cells are preferentially increased in NPs with a distinct transcriptional program. a UMAP plots showing that 81,202 CD8 + T cells from 25 samples (5 CBL, 8 NP-BL, 4 CIT, and 8 NP samples) are separated into 16 clusters (upper left). Clusters are annotated into nine major cell types by canonical markers (upper right) and colored by different sampling locations (lower left and right). b Dot plots showing the scaled expression of selected canonical marker genes in the indicated cell types. c Feature plots and violin plots illustrating expression of naive, effector memory and cytotoxicity curated gene signatures across CD8 + T cell clusters. d Bar plots showing the compositions of major cell types in each sample across different sampling locations in control participants and patients with CRSwNP. e Tissue prevalence of major cell types in the indicated group (13 blood samples (including 5 CBL and 8 NP-BL samples) and 12 nasal tissue samples (including 4 CIT and 8 NP samples)) is estimated by Ro/e score. f Scatter-plot shows differentially expressed genes (DEGs) between GZMK + CD8 + T cells and other CD8 + T cells. Two-sided Wilcoxon rank-sum tests with Bonferroni correction. Genes with |log2(fold change) | > 0.5 and adjusted P < 0.05 were considered significant. NS, no significant difference; P adj, adjusted P value; Δ percent of cells, the difference in the percentage of cells expressing the gene comparing GZMK + CD8 + T versus all other CD8 + T cells. g Gene set enrichment analysis (GSEA) showing significantly differentially upregulated pathways in GZMK + CD8 + T cells compared to other CD8 + T cells. Two-sided permutation test with Benjamini-Hochberg adjustments was used for GSEA analysis. Normalized enrichment score (NES) > 1 and adjusted P < 0.05 was considered significant. h Violin plots displaying top10 differentially expressed genes (DEGs) among GZMK + CD8 + T and GZMB + CD8 + T cells in NPs. Two-sided Wilcoxon rank-sum tests with Bonferroni correction. Genes with |log2(fold change) | > 0.5 and adjusted P < 0.05 were considered significant. i , Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway analysis of DEGs that enriched in indicated CD8 + T clusters. Two-sided Fisher’s Exact test with Benjamini-Hochberg adjustments was used for KEGG analysis. Adjusted P < 0.05 was considered significant. CBL control blood sample, CD8 + Tpex, CD8 + progenitor exhausted cells, CIT control inferior turbinate sample, MAIT mucosal associated invariant T cell, NP nasal polyp, NP-BL blood sample from CRSwNP patient, TRM tissue-resident memory T cell. Source data are provided as a Source Data file.
Index in PubMed under a CC BY license. PMID: 39614076
Click image to see more details
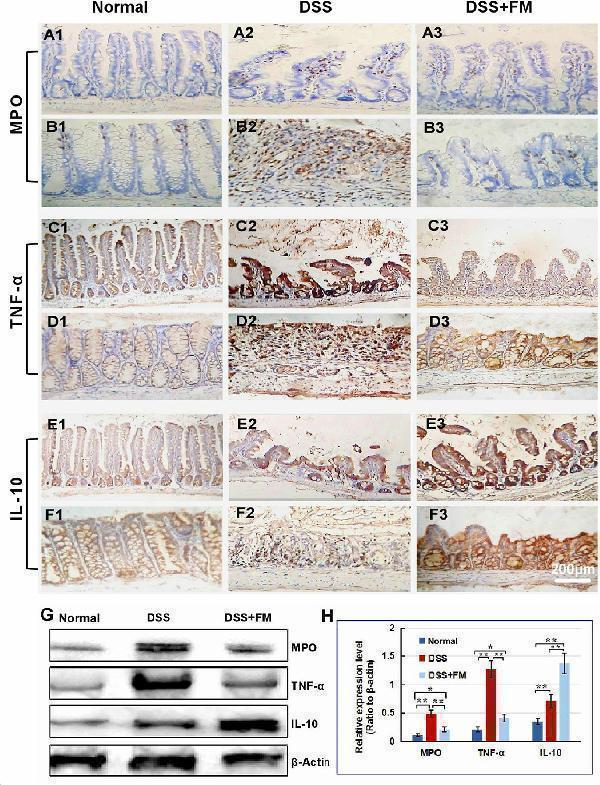
The infiltration of MPO + neutrophils, and the cellular distribution and relative expression level detection of the TNF and IL-10 in the small intestinal and colonic mucosa at 7 days after the termination of DSS administration. (A) The MPO immunohistochemistry staining of the small intestinal mucosa: (A1) the normal group: few neutrophils were observed in the small intestinal mucosa; (A2) the DSS group: a number of accumulative MPO + neutrophils (brown) infiltrated into the mucosa epithelium; (A3) the DSS + B. subtilis- fermented milk group: only limited neutrophil infiltration could be observed in the small intestinal mucosa. (B) The MPO immunohistochemistry staining of the colonic mucosa: (B1) the normal group: few neutrophils were observed in the colonic mucosa; (B2) the DSS group: colonic epithelium and the glands disappeared, and the ulcer was locally replaced by scars and a number of accumulative MPO + neutrophils (brown) were observed in the scars; (B3) the DSS + B. subtilis -fermented milk group: only limited MPO + neutrophils observed in the colonic mucosa. (C) The TNF immunohistochemistry staining of the small intestinal mucosa: (C1) the normal group: the epithelium was integrated with faint yellow staining, suggesting low expression of TNF; (C2) the DSS group: the villus structure is not integrated, and the epithelial cells showed black brown, suggesting overexpression of TNF; (C3) the DSS + B. subtilis -fermented milk group: the villus and the glands were almost integrated, and the staining of epithelial cells was similar to that of the normal group (C1) , suggesting low expression of TNF. (D) The TNF immunohistochemistry staining of the colonic mucosa: (D1) the normal colonic mucosa: the epithelium was integrated with low TNF expression (faint yellow); ( D2 ) the DSS group: the epithelium structure and the glands were destroyed and replaced by a scar, and there were a number of TNF + inflammatory cells (black brown) in the scar; (D3) the DSS + B. subtilis -fermented milk group: the recovered epithelium showed faint yellow, suggesting low TNF expression. (E) The IL-10 immunohistochemistry staining of the small intestinal mucosa: (E1) the normal small intestinal mucosa: the IL-10 staining dispersed in the villi and the crypts with faint yellow, suggesting low-level expression of IL-10; (E2) the DSS group, the residual epithelium and the crypts were light brown, suggesting mid-level of IL-10 expression; (E3) the DSS + B. subtilis -fermented milk group: the dark brown staining of the regenerative epithelium represented high-level expression of IL-10. (F) The IL-10 immunohistochemistry staining of the colonic mucosa: (F1) the normal group: the IL-10 staining dispersed in the glands with bright yellow, suggesting low-level expression of IL-10; (F2) the DSS group: there were few IL-10 + cells in the scars; (F3) the DSS + B. subtilis -fermented milk group, the dark brown staining of the epithelial cells represented high-level expression of IL-10. (G,H) Western blotting analysis for the expression of MPO, TNF, and IL-10 in the samples containing equivalent ileum and colon. The expression level of MPO, TNF, and IL-10 in the DSS group was significantly higher than that of the normal (control) group. The expression level of MPO and TNF in the DSS + B. subtilis -fermented milk (FM) group was significantly lower than that of the DSS group, while the expression level of IL-10 in the DSS + B. subtilis -fermented milk (FM) group was significantly higher than that of the DSS group ( n = 5, * represents p < 0.05, ** represents p < 0.01).
Index in PubMed under a CC BY license. PMID: 33519783
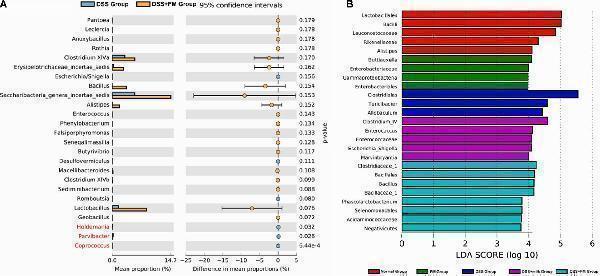
Click image to see more details
Difference in the mean proportions of the major composition of the intestinal flora. (A) Mean proportions of the top 25 genus in the intestinal flora at 7 days after termination of DSS intake and the statistical difference between group 3 (DSS group) and group 5 (DSS + B. subtilis -fermented milk group) ( n = 5). (B) Significantly different taxa as measured by LEfSe analysis (threshold > 3.5).
Index in PubMed under a CC BY license. PMID: 33519783
Specific Publications For Anti-Myeloperoxidase/MPO Antibody Picoband® (PA1054)
Loading publications
Recommended Resources
Here are featured tools and databases that you might find useful.
- Boster's Pathways Library
- Protein Databases
- Bioscience Research Protocol Resources
- Data Processing & Analysis Software
- Photo Editing Software
- Scientific Literature Resources
- Research Paper Management Tools
- Molecular Biology Software
- Primer Design Tools
- Bioinformatics Tools
- Phylogenetic Tree Analysis
Customer Reviews
Have you used Anti-Myeloperoxidase/MPO Antibody Picoband®?
Share your experimental results or join a short interview to earn up to $1,000 in product credits or other rewards.
1 Reviews For Anti-Myeloperoxidase/MPO Antibody Picoband®
Using Myeloperoxidase/MPO Antibody Picoband® (PA1054) in IHC, MPO showed low expression in normal mouse skin and was markedly increased in burned skin, with clear staining and expected results.
Excellent
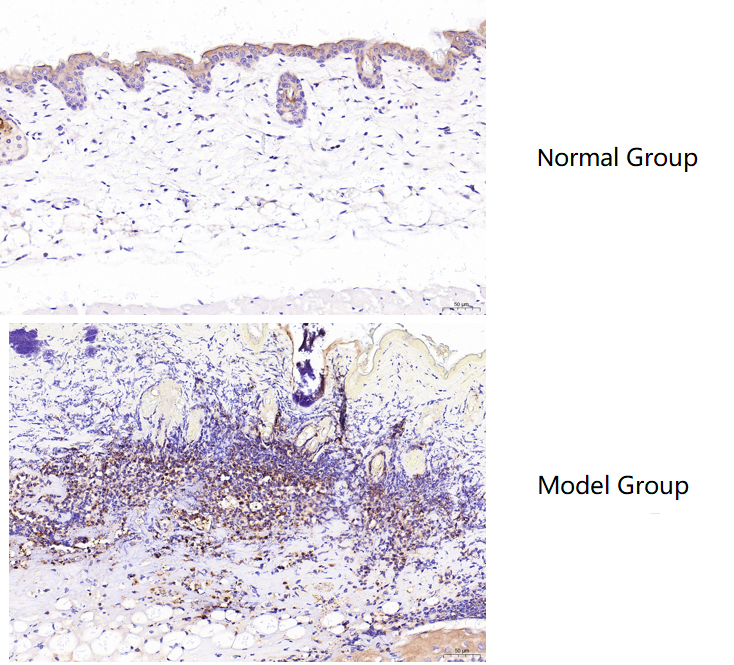
| SKU | PA1054 |
|---|---|
| Application | Immunohistochemistry |
| Sample | mouse skin tissue |
| Sample Processing Description | Male BALB/c mice aged 6–8 weeks were used. (1) Normal dorsal skin was collected from untreated mice. (2) Burn injury was induced on the dorsal skin using a burn device. After one week of housing, hair was removed with depilatory cream. Skin samples were collected from normal mice (dorsal skin) and burned mice (burned area), fixed in formalin for 72 hours, and embedded in paraffin for sectioning./td> |
| Other Reagents | Tris-EDTA Antigen Retrieval Buffer (50×, pH 9.0), DAB Chromogen Kit |
| Primary Antibody | Myeloperoxidase/MPO Antibody Picoband® |
| Primary Incubation | 1:100, overnight at 4 ℃ |
| Secondary Antibody | Polymer Anti-Rabbit IgG–HRP Immunohistochemistry Kit |
| Detection | Imaging system:Leica DM2500 |
| Results Summary | MPO (myeloperoxidase) is a lysosomal enzyme mainly present in neutrophils and monocytes/macrophages. Its core function is to generate reactive oxygen species, and therefore it plays a “double-edged sword” role in innate immune defense and inflammation-related diseases. MPO is present at very low levels in normal skin, but in burned skin, a large number of macrophages accumulate around the injured tissue, accompanied by a significant increase in MPO expression. Based on the immunohistochemical results, the staining is clear and the findings are consistent with expectations. |
Ruibo Zhao, South China Agricultural University
Verified customer
Submitted 2026-01-26
Customer Q&As
Have a question?
Find answers in Q&As, reviews.
Can't find your answer?
Submit your question
18 Customer Q&As for Anti-Myeloperoxidase/MPO Antibody Picoband®
Question
Does anti-Myeloperoxidase/MPO antibody PA1054 work on canine IHC-P with bone marrow cell?
Verified Customer
Verified customer
Asked: 2020-04-14
Answer
Our lab technicians have not tested anti-Myeloperoxidase/MPO antibody PA1054 on canine. You can run a BLAST between canine and the immunogen sequence of anti-Myeloperoxidase/MPO antibody PA1054 to see if they may cross-react. If the sequence homology is close, then you can perform a pilot test. Keep in mind that since we have not validated canine samples, this use of the antibody is not covered by our guarantee. However we have an innovator award program that if you test this antibody and show it works in canine bone marrow cell in IHC-P, you can get your next antibody for free.
Boster Scientific Support
Answered: 2020-04-14
Question
Is a blocking peptide available for product anti-Myeloperoxidase/MPO antibody (PA1054)?
Verified Customer
Verified customer
Asked: 2020-02-27
Answer
We do provide the blocking peptide for product anti-Myeloperoxidase/MPO antibody (PA1054). If you would like to place an order for it please contact support@bosterbio.com and make a special request.
Boster Scientific Support
Answered: 2020-02-27
Question
My question regarding product PA1054, anti-Myeloperoxidase/MPO antibody. I was wondering if it would be possible to conjugate this antibody with biotin. I would need it to be without BSA or sodium azide. I am planning on using a buffer exchange of sodium azide with PBS only. Would there be problems for me to conjugate the antibody and store it in -20 degrees in small aliquots?
Verified Customer
Verified customer
Asked: 2020-01-20
Answer
We suggest not storing this antibody with PBS buffer only in -20 degrees. If you want to store it in -20 degrees it is best to add some cryoprotectant like glycerol. If you want carrier free PA1054 anti-Myeloperoxidase/MPO antibody, we can provide it to you in a special formula with trehalose and/or glycerol. These molecules will not interfere with conjugation chemistry and provide a good level of protection for the antibody from degradation. Please be sure to specify this in your purchase order.
Boster Scientific Support
Answered: 2020-01-20
Question
Our lab want to know about to test anti-Myeloperoxidase/MPO antibody PA1054 on human leukemia for research purposes, then I may be interested in using anti-Myeloperoxidase/MPO antibody PA1054 for diagnostic purposes as well. Is the antibody suitable for diagnostic purposes?
Verified Customer
Verified customer
Asked: 2019-08-30
Answer
The products we sell, including anti-Myeloperoxidase/MPO antibody PA1054, are only intended for research use. They would not be suitable for use in diagnostic work. If you have the means to develop a product into diagnostic use, and are interested in collaborating with us and develop our product into an IVD product, please contact us for more discussions.
Boster Scientific Support
Answered: 2019-08-30
Question
See attached the WB image, lot number and protocol we used for leukemia using anti-Myeloperoxidase/MPO antibody PA1054. Please let me know if you require anything else.
Verified Customer
Verified customer
Asked: 2019-08-12
Answer
Thank you very much for the data. Our lab team are working to resolve this as quickly as possible, and we appreciate your patience and understanding! You have provided everything we needed. Please let me know if there is anything you need in the meantime.
Boster Scientific Support
Answered: 2019-08-12
Question
We appreciate helping with my inquiry over the phone. Here are the WB image, lot number and protocol we used for leukemia using anti-Myeloperoxidase/MPO antibody PA1054. Let me know if you need anything else.
Verified Customer
Verified customer
Asked: 2019-07-10
Answer
Thank you for the data. You have provided everything we needed. Our lab team are working to resolve your inquiry as quickly as possible, and we appreciate your patience and understanding! Please let me know if there is anything you need in the meantime.
Boster Scientific Support
Answered: 2019-07-10
Question
Can PA1054 is antibody be provided in a fluorescence conjugation format? If yes, what is the minimum size and the lead time?
Verified customer
Asked: 2019-07-05
Answer
We have to manufacture a new lot of Anti-Myeloperoxidase/MPO Antibody PA1054 for conjugation. The minimum size is 1mg and the lead time would be 3-4 months.
Boster Scientific Support
Answered: 2019-07-08
Question
Does PA1054 anti-Myeloperoxidase/MPO antibody work on parafin embedded sections? If so, which fixation method do you recommend we use (PFA, paraformaldehyde, other)?
Verified Customer
Verified customer
Asked: 2019-06-17
Answer
It shows on the product datasheet, PA1054 anti-Myeloperoxidase/MPO antibody as been tested on WB. It is best to use PFA for fixation because it has better tissue penetration ability. PFA needs to be prepared fresh before use. Long term stored PFA turns into formalin, as the PFA molecules congregate and become formalin.
Boster Scientific Support
Answered: 2019-06-17
Question
Is this PA1054 anti-Myeloperoxidase/MPO antibody reactive to the isotypes of MPO?
Verified Customer
Verified customer
Asked: 2018-08-22
Answer
The immunogen of PA1054 anti-Myeloperoxidase/MPO antibody is A synthetic peptide corresponding to a sequence at the C-terminus of human MPO (714-728aa KNNIFMSNSYPRDFV), different from the related mouse and rat sequences by one amino acid. Could you tell me which isotype you are interested in so I can help see if the immunogen is part of this isotype?
Boster Scientific Support
Answered: 2018-08-22
Question
I would like using your anti-Myeloperoxidase/MPO antibody for oxidation-reduction process studies. Has this antibody been tested with western blotting on rat brain tissue? We would like to see some validation images before ordering.
Verified Customer
Verified customer
Asked: 2018-01-09
Answer
We appreciate your inquiry. This PA1054 anti-Myeloperoxidase/MPO antibody is tested on rat brain tissue. It is guaranteed to work for IF, IHC-P, WB in human, mouse, rat. Our Boster guarantee will cover your intended experiment even if the sample type has not been be directly tested.
Boster Scientific Support
Answered: 2018-01-09
Question
Do you have a BSA free version of anti-Myeloperoxidase/MPO antibody PA1054 available?
Verified Customer
Verified customer
Asked: 2018-01-03
Answer
Thanks for your recent telephone inquiry. I can confirm that some lots of this anti-Myeloperoxidase/MPO antibody PA1054 are BSA free. For now, these lots are available and we can make a BSA free formula for you free of charge. It will take 3 extra days to prepare. If you require this antibody BSA free again in future, please do not hesitate to contact me and I will be pleased to check which lots we have in stock that are BSA free.
Boster Scientific Support
Answered: 2018-01-03
Question
We have been able to see staining in rat plasma. Are there any suggestions? Is anti-Myeloperoxidase/MPO antibody supposed to stain plasma positively?
D. Parker
Verified customer
Asked: 2017-12-12
Answer
From what I have seen in literature plasma does express MPO. From what I have seen in Uniprot.org, MPO is expressed in bone marrow cell, leukemia, leukocyte, plasma, saliva, liver, among other tissues. Regarding which tissues have MPO expression, here are a few articles citing expression in various tissues:
Leukemia, Pubmed ID: 2903767
Leukocyte, Pubmed ID: 1334087, 8390465, 2884926
Liver, Pubmed ID: 19159218
Plasma, Pubmed ID: 16335952
Saliva, Pubmed ID: 16740002
Boster Scientific Support
Answered: 2017-12-12
Question
We are currently using anti-Myeloperoxidase/MPO antibody PA1054 for rat tissue, and we are satisfied with the IHC-P results. The species of reactivity given in the datasheet says human, mouse, rat. Is it possible that the antibody can work on horse tissues as well?
A. Gonzalez
Verified customer
Asked: 2017-10-26
Answer
The anti-Myeloperoxidase/MPO antibody (PA1054) has not been validated for cross reactivity specifically with horse tissues, though there is a good chance of cross reactivity. We have an innovator award program that if you test this antibody and show it works in horse you can get your next antibody for free. Please contact me if I can help you with anything.
Boster Scientific Support
Answered: 2017-10-26
Question
Our lab were well pleased with the WB result of your anti-Myeloperoxidase/MPO antibody. However we have seen positive staining in leukemia lysosome. using this antibody. Is that expected? Could you tell me where is MPO supposed to be expressed?
Verified Customer
Verified customer
Asked: 2017-08-16
Answer
From what I have seen in literature, leukemia does express MPO. Generally MPO expresses in lysosome. Regarding which tissues have MPO expression, here are a few articles citing expression in various tissues:
Leukemia, Pubmed ID: 2903767
Leukocyte, Pubmed ID: 1334087, 8390465, 2884926
Liver, Pubmed ID: 19159218
Plasma, Pubmed ID: 16335952
Saliva, Pubmed ID: 16740002
Boster Scientific Support
Answered: 2017-08-16
Question
Does anti-Myeloperoxidase/MPO antibody PA1054 work for WB with leukemia?
Verified Customer
Verified customer
Asked: 2017-06-06
Answer
According to the expression profile of leukemia, MPO is highly expressed in leukemia. So, it is likely that anti-Myeloperoxidase/MPO antibody PA1054 will work for WB with leukemia.
Boster Scientific Support
Answered: 2017-06-06
Question
We ordered your anti-Myeloperoxidase/MPO antibody for IF on bone marrow cell in the past. I am using mouse, and We are going to use the antibody for WB next. I am interested in examining bone marrow cell as well as leukocyte in our next experiment. Could you please give me some suggestion on which antibody would work the best for WB?
J. Carter
Verified customer
Asked: 2015-04-03
Answer
I looked at the website and datasheets of our anti-Myeloperoxidase/MPO antibody and it seems that PA1054 has been validated on mouse in both IF and WB. Thus PA1054 should work for your application. Our Boster satisfaction guarantee will cover this product for WB in mouse even if the specific tissue type has not been validated. We do have a comprehensive range of products for WB detection and you can check out our website bosterbio.com to find out more information about them.
Boster Scientific Support
Answered: 2015-04-03
Question
I was wanting to use your anti-Myeloperoxidase/MPO antibody for WB for human leukemia on frozen tissues, but I want to know if it has been validated for this particular application. Has this antibody been validated and is this antibody a good choice for human leukemia identification?
R. Brown
Verified customer
Asked: 2014-09-01
Answer
You can see on the product datasheet, PA1054 anti-Myeloperoxidase/MPO antibody has been tested for IF, IHC-P, WB on human, mouse, rat tissues. We have an innovator award program that if you test this antibody and show it works in human leukemia in IHC-frozen, you can get your next antibody for free.
Boster Scientific Support
Answered: 2014-09-01
Question
I see that the anti-Myeloperoxidase/MPO antibody PA1054 works with WB, what is the protocol used to produce the result images on the product page?
N. Parker
Verified customer
Asked: 2013-11-01
Answer
You can find protocols for WB on the "support/technical resources" section of our navigation menu. If you have any further questions, please send an email to support@bosterbio.com
Boster Scientific Support
Answered: 2013-11-01






