Product Info Summary
| SKU: | PB9087 |
|---|---|
| Size: | 100 μg/vial |
| Reactive Species: | Human |
| Host: | Rabbit |
| Application: | WB |
Customers Who Bought This Also Bought
Product info
Product Name
Anti-NF-kB p65/RELA Antibody Picoband®
SKU/Catalog Number
PB9087
PB0073 is an alternative SKU for this antibody, used in previous lots.
Size
100 μg/vial
Form
Lyophilized
Description
Boster Bio Anti-NF-kB p65/RELA Antibody Picoband® catalog # PB9087. Tested in WB applications. This antibody reacts with Human. The brand Picoband indicates this is a premium antibody that guarantees superior quality, high affinity, and strong signals with minimal background in Western blot applications. Only our best-performing antibodies are designated as Picoband, ensuring unmatched performance.
Storage & Handling
Store at -20˚C for one year from date of receipt. After reconstitution, at 4˚C for one month. It can also be aliquotted and stored frozen at -20˚C for six months. Avoid repeated freeze-thaw cycles.
Cite This Product
Anti-NF-kB p65/RELA Antibody Picoband® (Boster Biological Technology, Pleasanton CA, USA, Catalog # PB9087)
Host
Rabbit
Contents
Each vial contains antibody formulated with stabilizing components, 0.9 mg NaCl, 0.2 mg Na2HPO4, and 0.05 mg NaN3.
*This antibody is supplied in a stabilized formulation.
Compatibility with conjugation reactions depends on the chemistry of the conjugation method used.
For conjugation methods that are not compatible with the stabilizing components present in this formulation, a carrier-free antibody format is required.
Clonality
Polyclonal
Isotype
Rabbit IgG
Immunogen
E.coli-derived human NF-kB p65 recombinant protein (Position: F99-S551). Human NF-kB p65 shares 86% amino acid (aa) sequence identity with mouse NF-kB p65.
Cross-reactivity
No cross-reactivity with other proteins
Reactive Species
PB9087 is reactive to RELA in Human
Observed Molecular Weight
65 kDa
Calculated molecular weight
60.2 kDa
Background of RELA
Transcription factor p65, also known as NFKB3 or NF-kB p65, is a protein that in humans is encoded by the RELA gene. It is mapped to 11q13.1. NFKB is an essential transcription factor complex involved in all types of cellular processes, including cellular metabolism, chemotaxis, etc, and it may play a role in inflammatory conditions of the peripheral nervous system. Phosphorylation and acetylation of NFKB3 are crucial post-translational modifications required for NFKB activation. It has also been shown to modulate immune responses, and activation of NFKB3 is positively associated with multiple types of cancer. In addition to that, NFKB3 antagonizes TNFR1-JNK proliferative signals in epidermis and plays a nonredundant role in restraining epidermal growth.
Antibody Validation
Boster validates all antibodies on WB, IHC, ICC, Immunofluorescence, and ELISA with known positive control and negative samples to ensure specificity and high affinity, including thorough antibody incubations.
Application & Images
Applications
PB9087 is guaranteed for WB Boster Guarantee
Recommend Dilution
| Application | Dilution | Species |
|---|---|---|
| Western blot | 0.1-0.5μg/ml | Human |
Tested application
Suggested blocking solution with 5% non-fat milk or BSA; (*)Recommended protein loading: 20-40 µg per lane
Validation Images & Assay Conditions

Click image to see more details
Western blot analysis of NF-kB p65/RELA using anti-NF-kB p65/RELA antibody (PB9087).
Electrophoresis was performed on a 5-20% SDS-PAGE gel at 70V (Stacking gel) / 90V (Resolving gel) for 2-3 hours. The sample well of each lane was loaded with 30 ug of sample under reducing conditions.
Lane 1: human Hela whole cell lysates,
Lane 2: human HepG2 whole cell lysates,
Lane 3: human Caco-2 whole cell lysates,
Lane 4: human Raji whole cell lysates.
After electrophoresis, proteins were transferred to a nitrocellulose membrane at 150 mA for 50-90 minutes. Blocked the membrane with 5% non-fat milk/TBS for 1.5 hour at RT. The membrane was incubated with rabbit anti-NF-kB p65/RELA antigen affinity purified polyclonal antibody (Catalog # PB9087) at 0.5 μg/mL overnight at 4°C, then washed with TBS-0.1%Tween 3 times with 5 minutes each and probed with a goat anti-rabbit IgG-HRP secondary antibody at a dilution of 1:5000 for 1.5 hour at RT. The signal is developed using an Enhanced Chemiluminescent detection (ECL) kit (Catalog # EK1002) with Tanon 5200 system. A specific band was detected for NF-kB p65/RELA at approximately 65 kDa. The expected band size for NF-kB p65/RELA is at 60 kDa.
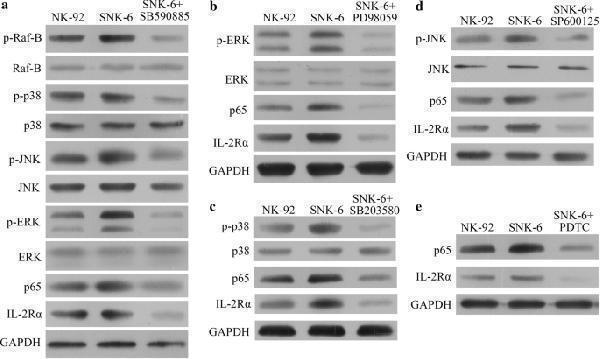
Click image to see more details
Treating SNK-6 cells constitutively expressing LMP1 for 1 h with the following selective inhibitors of proteins in the MAPK/NF-κB pathway down-regulated IL-2Rα: a 0.1 μM SB590885 (B-Raf inhibitor), b 20 μM PD98059 (ERK inhibitor), c 10 μM SB203580 (p38 inhibitor), d 20 μM SP600125 (JNK inhibitor), or e 100 μM pyrrolidine dithiocarbamate (PDTC, NF-κB inhibitor)
Index in PubMed under a CC BY license. PMID: 30340635
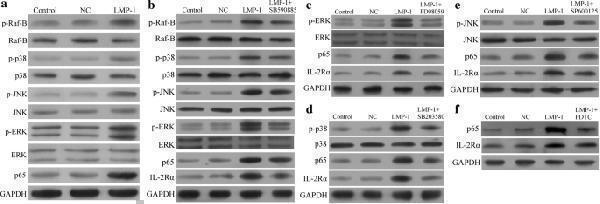
Click image to see more details
LMP1 up-regulated IL-2Rα expression through the MAPK/NF-κB pathway in NK-92 cells. a NK-92 cells transduced with LMP1-encoding lentivirus led to higher levels of p-Raf-B, p-p38, p-JNK, p-ERK, and p65 than transduction with NC lentivirus. b – f NK-92 cells transduced with LMP1-encoding lentivirus were treated for 1 h with the following selective inhibitors of proteins in the MAPK/NF-κB pathway, leading to IL-2Rα down-regulation: b 0.1 μM SB590885 (B-Raf inhibitor), c 20 μM PD98059 (ERK inhibitor), d 10 μM SB203580 (p38 inhibitor), e 20 μM SP600125 (JNK inhibitor), and f 100 μM pyrrolidine dithiocarbamate (PDTC; NF-κB inhibitor)
Index in PubMed under a CC BY license. PMID: 30340635
Specific Publications For Anti-NF-kB p65/RELA Antibody Picoband® (PB9087)
Loading publications
Recommended Resources
Here are featured tools and databases that you might find useful.
- Boster's Pathways Library
- Protein Databases
- Bioscience Research Protocol Resources
- Data Processing & Analysis Software
- Photo Editing Software
- Scientific Literature Resources
- Research Paper Management Tools
- Molecular Biology Software
- Primer Design Tools
- Bioinformatics Tools
- Phylogenetic Tree Analysis
Customer Reviews
Have you used Anti-NF-kB p65/RELA Antibody Picoband®?
Share your experimental results or join a short interview to earn up to $1,000 in product credits or other rewards.
0 Reviews For Anti-NF-kB p65/RELA Antibody Picoband®
Customer Q&As
Have a question?
Find answers in Q&As, reviews.
Can't find your answer?
Submit your question





