Product Info Summary
| SKU: | PB9457 |
|---|---|
| Size: | 100 μg/vial |
| Reactive Species: | Human, Mouse, Rat |
| Host: | Rabbit |
| Application: | Flow Cytometry, IF, IHC, WB |
Customers Who Bought This Also Bought
Product info
Product Name
Anti-Villin/VIL1 Antibody Picoband®
SKU/Catalog Number
PB9457
Size
100 μg/vial
Form
Lyophilized
Description
Boster Bio Anti-Villin/VIL1 Antibody Picoband® catalog # PB9457. Tested in Flow Cytometry, IF, IHC, WB applications. This antibody reacts with Human, Mouse, Rat. The brand Picoband indicates this is a premium antibody that guarantees superior quality, high affinity, and strong signals with minimal background in Western blot applications. Only our best-performing antibodies are designated as Picoband, ensuring unmatched performance.
Storage & Handling
Store at -20˚C for one year from date of receipt. After reconstitution, at 4˚C for one month. It can also be aliquotted and stored frozen at -20˚C for six months. Avoid repeated freeze-thaw cycles.
Cite This Product
Anti-Villin/VIL1 Antibody Picoband® (Boster Biological Technology, Pleasanton CA, USA, Catalog # PB9457)
Host
Rabbit
Contents
Each vial contains antibody formulated with stabilizing components, 0.9 mg NaCl, 0.2 mg Na2HPO4, and 0.05 mg NaN3.
*This antibody is supplied in a stabilized formulation.
Compatibility with conjugation reactions depends on the chemistry of the conjugation method used.
For conjugation methods that are not compatible with the stabilizing components present in this formulation, a carrier-free antibody format is required.
Clonality
Polyclonal
Isotype
Rabbit IgG
Immunogen
A synthetic peptide corresponding to a sequence at the C-terminus of human Villin, different from the related mouse sequence by three amino acids.
Cross-reactivity
No cross-reactivity with other proteins
Reactive Species
PB9457 is reactive to VIL1 in Human, Mouse, Rat
Observed Molecular Weight
93 kDa
Calculated molecular weight
92.7 kDa
Background of VIL1
Villin is known as VIL1. This gene encodes a member of a family of calcium-regulated actin-binding proteins. This protein represents a dominant part of the brush border cytoskeleton which functions in the capping, severing, and bundling of actin filaments. Two mRNAs of 2.7 kb and 3.5 kb have been observed; they result from utilization of alternate poly-adenylation signals present in the terminal exon. In vertebrates, the villin proteins help to support the microfilaments of the microvilli of the brush border. It may play a role in cell plasticity through F-actin severing.
Antibody Validation
Boster validates all antibodies on WB, IHC, ICC, Immunofluorescence, and ELISA with known positive control and negative samples to ensure specificity and high affinity, including thorough antibody incubations.
Application & Images
Applications
PB9457 is guaranteed for Flow Cytometry, IF, IHC, WB Boster Guarantee
Recommend Dilution
| Application | Dilution | Species |
|---|---|---|
| Western blot | 0.1-0.5μg/ml | Human, Mouse, Rat |
| Immunohistochemistry (Paraffin-embedded Section) | 0.5-1μg/ml | Human, Mouse, Rat |
| Immunofluorescence | 2μg/ml | Human, Mouse, Rat |
| Flow Cytometry (Fixed) | 1-3μg/1x106 cells | Human |
Tested application
Suggested blocking solution with 5% non-fat milk or BSA; (*)Recommended protein loading: 20-40 µg per lane
Use TE buffer pH 9.0 for antigen retrieval; (*) citrate buffer pH 6.0 is an alternative.
Validation Images & Assay Conditions

Click image to see more details
Western blot analysis of Villin using anti-Villin antibody (PB9457).
Electrophoresis was performed on a 5-20% SDS-PAGE gel at 70V (Stacking gel) / 90V (Resolving gel) for 2-3 hours.
Lane 1: Rat Intestine Tissue Lysate at 50ug
Lane 2: Mouse Kidney Tissue Lysate at 50ug
Lane 3: RH35 Whole Cell Lysate at 40ug
Lane 4: HEPG2 Whole Cell Lysate at 40ug,
Lane 5: MCF-7 Whole Cell Lysate at 40ug.
After electrophoresis, proteins were transferred to a nitrocellulose membrane at 150 mA for 50-90 minutes. Blocked the membrane with 5% non-fat milk/TBS for 1.5 hour at RT. The membrane was incubated with rabbit anti-Villin antigen affinity purified polyclonal antibody (Catalog # PB9457) at 0.5 μg/mL overnight at 4°C, then washed with TBS-0.1%Tween 3 times with 5 minutes each and probed with a goat anti-rabbit IgG-HRP secondary antibody at a dilution of 1:5000 for 1.5 hour at RT. The signal is developed using an Enhanced Chemiluminescent detection (ECL) kit (Catalog # EK1002) with Tanon 5200 system. A specific band was detected for Villin at approximately 93 kDa. The expected band size for Villin is at 93 kDa.

Click image to see more details
IHC analysis of Villin using anti-Villin antibody (PB9457).
Villin was detected in a paraffin-embedded section of mouse intestine tissue. Heat mediated antigen retrieval was performed in EDTA buffer (pH 8.0, epitope retrieval solution). The tissue section was blocked with 10% goat serum. The tissue section was then incubated with 1 μg/ml rabbit anti-Villin Antibody (PB9457) overnight at 4°C. Biotinylated goat anti-rabbit IgG was used as secondary antibody and incubated for 30 minutes at 37°C. The tissue section was developed using Strepavidin-Biotin-Complex (SABC) (Catalog # SA1022) with DAB as the chromogen.
Click image to see more details
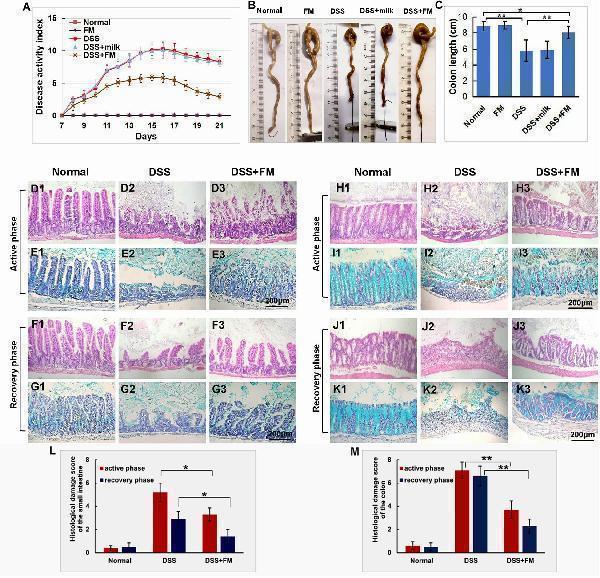
Effects of the B. subtilis -fermented milk on the disease severity and the pathological changes in the small intestine and the colon of the DSS-induced IBD. (A) The DAI dynamic changes of the DSS-induced IBD animal models. During the second 7-day period (active phase of IBD), DAI in the DSS + B. subtilis -fermented milk group was lower than those of the DSS group and the DSS + milk group ( n = 15, p < 0.01). In the third 7-day period (recovery phase of IBD), after termination of DSS intake, DAI recovery with varying degrees was observed in the DSS group, the DSS + milk group, and the DSS + B. subtilis -fermented milk group. However, in the DSS + B. subtilis -fermented milk group, the DAI index recovered faster and was significantly lower than those of the DSS group and the DSS + milk group ( n = 15, p < 0.01). The DSS + milk group showed no obvious difference from the DSS group ( n = 15, p > 0.05). (B) The colonic appearance of the model animals at 7 days after termination of DSS administration. (C) Quantification analysis of the length of the colons in different groups. At 7 days after the termination of DSS intake, the colons in the DSS group and the DSS + milk group were shortened significantly compared with those in the normal group and B. subtilis -fermented milk group. However, in the DSS + B. subtilis -fermented milk, the colons were shortened only slightly and were significantly longer than those of the DSS group and the DSS + milk group. The B. subtilis -fermented milk group showed no obvious difference from the normal group, and the DSS + milk group showed no obvious difference from the DSS group ( n = 15, * represents p < 0.05, ** represents p < 0.01). (D,E) Changes in the histological structure of the small intestinal mucosa in the active phase of the DSS-induced IBD observed with HE staining and Alcian blue staining, respectively. (D1,E1) The normal group: the villi in the small intestinal mucosa were finger-like, which were arranged compactly and tidily. The crypt structure was integrated among the base of villi. The surface of the villi and the crypt were covered with epithelial cells and goblet cells, with abundant mucus; (D2,E2) the DSS group: the necrosis and ulcer of the mucosa were observed, the villi were disintegrated, and only some residual crypts were observed; (D3,E3) the DSS + B. subtilis -fermented milk group: damage in the small intestinal mucosa was relatively slight, and the crypt structure was almost integrated. (F,G) Changes in the histological structure of the small intestinal mucosa in the recovery phase of the DSS-induced IBD observed with HE staining and Alcian blue staining, respectively. (F1,G1) The normal group: no significant change was observed in the mucosa; (F2,G2) the DSS group: the small intestinal mucosa was partially recovered, and the villi were short and scattered; (F3,G3) the DSS + B. subtilis -fermented milk group: the intestinal mucosa epithelium and the crypts were integrated, although the regenerative villi were shorter than those of the normal small intestines. (H,I) Changes in the histological structure of the colonic mucosa in the active phase of the DSS-induced IBD observed with HE staining and Alcian blue staining, respectively. (H1,I1) The normal group: the colonic mucus were intact; (H2,I2) the DSS group: the deep ulcer was observed due to the damage of the epithelium and glands; (H3,I3) the DSS + B. subtilis -fermented milk group: fewer bleeding and necrosis were observed. The damage of the colonic mucosa was relatively slight, and only local superficial ulcers were observed. A major part of the epithelium and glands was integrated in the structure, and the goblet cells were filled with abundant mucus. (J,K) Changes in the histological structure of the colonic mucosa in the recovery phase of the DSS-induced IBD observed with HE staining and Alcian blue staining, respectively. (J1,K1) The normal group: no significant change was observed in the colonic mucosa; (J2,K2) the DSS group: ulcer of the colonic mucosa was replaced by an inflammatory scar; (J3,K3) the DSS + B. subtilis -fermented milk group: the epithelium was almost integrated and the surficial ulcer was replaced by regenerated regenerative colonic glands consisting of goblet cells, which were filled with abundant mucus. (L) Histological damage scores of different groups evaluated with H&E-stained sections of the small intestines. The histological damage scores of the DSS + B. subtilis -fermented milk group in both the active phase and the recovery phase were significantly lower than those of the DSS group ( n = 5, * represents p < 0.05). (M) Histological damage scores of different groups evaluated with H&E-stained sections of the colons. The histological damage scores of the DSS + B. subtilis -fermented milk group in both the active phase and the recovery phase were significantly lower than those of the DSS group ( n = 5, ** represents p < 0.01).
Index in PubMed under a CC BY license. PMID: 33519783
Click image to see more details
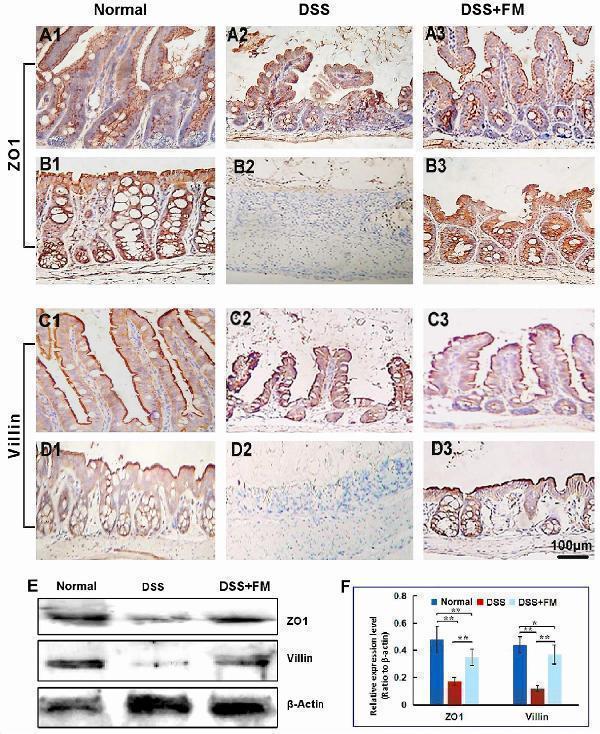
The subcellular localization and relative expression level detection of ZO-1 and villin in the intestinal mucosa of IBD at 7 days after termination of DSS intake. (A) The ZO-1 immunohistochemistry staining of the small intestinal epithelial TJP (brown dots): (A1) the normal group: the villi and crypts were arranged compactly, and the ZO-1-positive staining (representing the TJP) showed the dotted line (brown) along the surface of the villi and the crypts; (A2) the DSS group: ZO-1 distributed dispersively in the residual villi of the small intestinal mucosa; (A3) the DSS + B. subtilis -fermented milk group: the ZO-1 staining formed the dotted line (brown, representing the TJP) at the subsurface of the regenerative villi. (B) The ZO-1 immunohistochemistry staining of the colonic epithelial TJP (brown dots): (B1) the normal group: ZO-1-positive staining distributed on the inner side of the epithelial cell membrane (representing the TJP); (B2) the DSS group: there was no ZO-1-positive staining in the scar; (B3) the DSS + B. subtilis -fermented milk group: the ZO-1-positive staining distributed on the inner side of the regenerative epithelial cell membrane (representing the TJP). (C) The villin immunohistochemistry staining (brown strip) of the small intestinal microvilli: (C1) the normal group: villin-positive staining showed a strip-like distribution on the surface of the villi in the normal small intestinal mucosa; (C2) the DSS group: villin distributed at the surface of the residual villi; (C3) the DSS + B. subtilis -fermented milk group: villin-positive staining formed an integrative strip (brown) enclosing the surface of the regenerative villi. (D) The villin immunohistochemistry staining of the colonic epithelium: (D1) the normal group: villin-positive staining (brown) showed banded distribution on the surface of the epithelium; (D2) the DSS group: almost no villin-positive staining was observed in the scar due to damage of the epithelium; (D3) the DSS + B. subtilis -fermented milk group: the villin-positive staining (brown) showed banded distribution on the surface of the regenerated epithelium in the colonic mucosa. (E,F) The western blotting analysis for the relative expression level of ZO-1 and villin in the samples contained equivalent ileum and colon. The expression level of ZO-1 and villin in the DSS group was significantly lower than that of the normal (control) group. The expression level of ZO-1 and villin and in the DSS + B. subtilis -fermented milk (FM) group was significantly higher than that of the DSS group ( n = 5, * represents p < 0.05, ** represents p < 0.01).
Index in PubMed under a CC BY license. PMID: 33519783
Click image to see more details
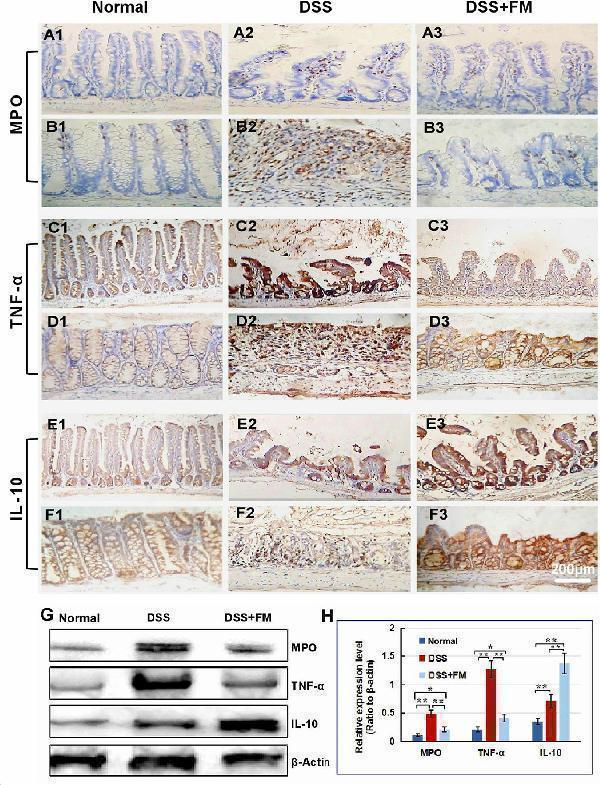
The infiltration of MPO + neutrophils, and the cellular distribution and relative expression level detection of the TNF and IL-10 in the small intestinal and colonic mucosa at 7 days after the termination of DSS administration. (A) The MPO immunohistochemistry staining of the small intestinal mucosa: (A1) the normal group: few neutrophils were observed in the small intestinal mucosa; (A2) the DSS group: a number of accumulative MPO + neutrophils (brown) infiltrated into the mucosa epithelium; (A3) the DSS + B. subtilis- fermented milk group: only limited neutrophil infiltration could be observed in the small intestinal mucosa. (B) The MPO immunohistochemistry staining of the colonic mucosa: (B1) the normal group: few neutrophils were observed in the colonic mucosa; (B2) the DSS group: colonic epithelium and the glands disappeared, and the ulcer was locally replaced by scars and a number of accumulative MPO + neutrophils (brown) were observed in the scars; (B3) the DSS + B. subtilis -fermented milk group: only limited MPO + neutrophils observed in the colonic mucosa. (C) The TNF immunohistochemistry staining of the small intestinal mucosa: (C1) the normal group: the epithelium was integrated with faint yellow staining, suggesting low expression of TNF; (C2) the DSS group: the villus structure is not integrated, and the epithelial cells showed black brown, suggesting overexpression of TNF; (C3) the DSS + B. subtilis -fermented milk group: the villus and the glands were almost integrated, and the staining of epithelial cells was similar to that of the normal group (C1) , suggesting low expression of TNF. (D) The TNF immunohistochemistry staining of the colonic mucosa: (D1) the normal colonic mucosa: the epithelium was integrated with low TNF expression (faint yellow); ( D2 ) the DSS group: the epithelium structure and the glands were destroyed and replaced by a scar, and there were a number of TNF + inflammatory cells (black brown) in the scar; (D3) the DSS + B. subtilis -fermented milk group: the recovered epithelium showed faint yellow, suggesting low TNF expression. (E) The IL-10 immunohistochemistry staining of the small intestinal mucosa: (E1) the normal small intestinal mucosa: the IL-10 staining dispersed in the villi and the crypts with faint yellow, suggesting low-level expression of IL-10; (E2) the DSS group, the residual epithelium and the crypts were light brown, suggesting mid-level of IL-10 expression; (E3) the DSS + B. subtilis -fermented milk group: the dark brown staining of the regenerative epithelium represented high-level expression of IL-10. (F) The IL-10 immunohistochemistry staining of the colonic mucosa: (F1) the normal group: the IL-10 staining dispersed in the glands with bright yellow, suggesting low-level expression of IL-10; (F2) the DSS group: there were few IL-10 + cells in the scars; (F3) the DSS + B. subtilis -fermented milk group, the dark brown staining of the epithelial cells represented high-level expression of IL-10. (G,H) Western blotting analysis for the expression of MPO, TNF, and IL-10 in the samples containing equivalent ileum and colon. The expression level of MPO, TNF, and IL-10 in the DSS group was significantly higher than that of the normal (control) group. The expression level of MPO and TNF in the DSS + B. subtilis -fermented milk (FM) group was significantly lower than that of the DSS group, while the expression level of IL-10 in the DSS + B. subtilis -fermented milk (FM) group was significantly higher than that of the DSS group ( n = 5, * represents p < 0.05, ** represents p < 0.01).
Index in PubMed under a CC BY license. PMID: 33519783
Click image to see more details
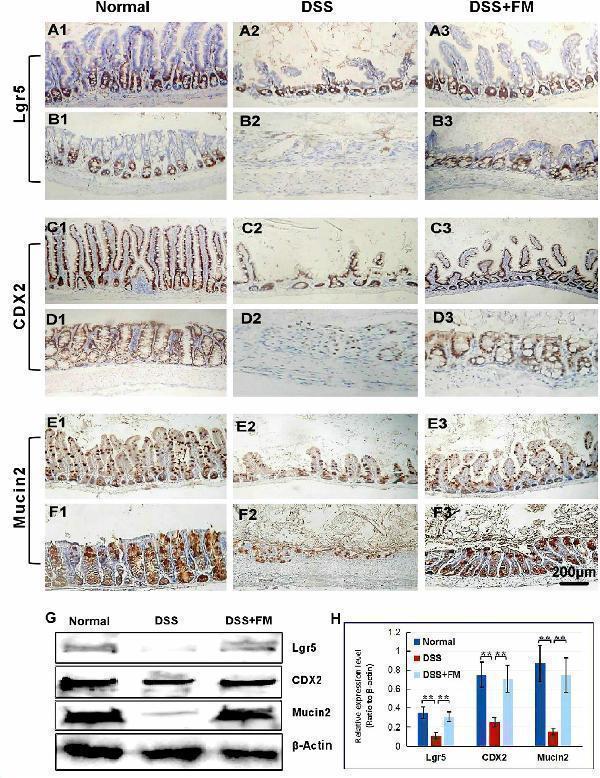
The distribution of Lgr5 + ISCs in the intestinalmucosa and the subcellular localization and relative expression level detection of epithelial function proteins CDX2 and villin in the intestinal mucosa of IBD at 7 days after termination of DSS administration. (A) The Lgr5 + ISCs (brown) in the small intestinal mucosa: (A1) the normal group, the villi and the crypts were arranged compactly, and Lgr5 + ISCs were observed in the crypts; (A2) the DSS group, the villi and the crypts were scattered, with few Lgr5 + ISCs; (A3) the DSS + B. subtilis -fermented milk group, there were more Lgr5 + ISCs in villi and crypts compared with those in the DSS group. (B) The Lgr5 + ISCs (brown) in the colonic mucosa: (B1) the normal group, the glands were arranged compactly, and there were large amounts of Lgr5 + ISCs at the bottom of the glands; (B2) the DSS group: the ulcers were replaced by scars. No Lgr5 + ISCs were observed in the scars; (B3) the DSS + B. subtilis -fermented milk group: the colonic epithelium was integrated, with some regenerated glands. A number of Lgr5 + ISCs were observed at the bottom of the regenerated glands. (C) The CDX2 was localized in the epithelial cellular nuclei (brown) by immunohistochemistry staining in the small intestinal mucosa: (C1) the normal group: the villi and the crypts were arranged compactly, and CDX2 + epithelial cells were observed on the surface of the villi and the crypts; (C2) the DSS group: the villi and the crypts were scattered, and few CDX2 + epithelial cells were observed on the surface of the crypt and the villi; (C3) the DSS + B. subtilis -fermented milk group: more villi and crypts were observed in comparison with the DSS group, and there were more CDX2 + epithelial cells covering the villi and crypts. (D) The CDX2 was localized in the epithelial cellular nuclei (brown) by immunohistochemistry staining in the colonic mucosa: (D1) the normal group: the colonic glands were arranged compactly, and CDX2 + epithelial cells were observed on the surface of the glands; (D2) the DSS group: the glands were scattered, and few CDX2 + epithelial cells were observed in the scar; (D3) the DSS + B. subtilis -fermented milk group: more colonic glands were observed in comparison with the DSS group, and there were more CDX2 + epithelial cells in the glands. (E) The Mucin2 was localized in the cytoplasm of the goblet cells (brown) by immunohistochemistry staining in the small intestinal mucosa: (E1) the normal group, a number of Mucin2 + goblet cells observed in the epithelium; (E2) the DSS group: only few Mucin2 + goblet cells were observed in the remaining villi and crypts; (E3) the DSS + B. subtilis -fermented milk group: more Mucin2 + goblet cells were observed in the recovered mucosa. (F) The Mucin2 was localized in the cytoplasm of the goblet cells (brown) by immunohistochemistry staining in the colonic mucosa: (F1) the normal group, large amounts of Mucin2 + goblet cells were observed in the mucosa; (F2) the DSS group: only few Mucin2 + goblet cells were observed in the scars; (F3) the DSS + B. subtilis -fermented milk group: more Mucin2 + goblet cells were observed in the recovered colonic mucosa. (G,H) Western blotting was applied for detection of the relative expression level of Lgr5, CDX2, and Mucin2 in the samples containing equivalent ileum and colon. The expression level of Lgr5, CDX2, and Mucin2 in the DSS group was significantly lower than that of the normal (control) group. The expression level of Lgr5, CDX2, and Mucin2 in the DSS + B. subtilis -fermented milk (FM) group was significantly higher than that of the DSS group ( n = 5, ** represents p < 0.01).
Index in PubMed under a CC BY license. PMID: 33519783

Click image to see more details
IHC analysis of Villin using anti-Villin antibody (PB9457).
Villin was detected in a paraffin-embedded section of rat intestine tissue. Heat mediated antigen retrieval was performed in EDTA buffer (pH 8.0, epitope retrieval solution). The tissue section was blocked with 10% goat serum. The tissue section was then incubated with 1 μg/ml rabbit anti-Villin Antibody (PB9457) overnight at 4°C. Biotinylated goat anti-rabbit IgG was used as secondary antibody and incubated for 30 minutes at 37°C. The tissue section was developed using Strepavidin-Biotin-Complex (SABC) (Catalog # SA1022) with DAB as the chromogen.

Click image to see more details
IHC analysis of Villin using anti-Villin antibody (PB9457).
Villin was detected in a paraffin-embedded section of human intestinal cancer tissue. Heat mediated antigen retrieval was performed in EDTA buffer (pH 8.0, epitope retrieval solution). The tissue section was blocked with 10% goat serum. The tissue section was then incubated with 1 μg/ml rabbit anti-Villin Antibody (PB9457) overnight at 4°C. Biotinylated goat anti-rabbit IgG was used as secondary antibody and incubated for 30 minutes at 37°C. The tissue section was developed using Strepavidin-Biotin-Complex (SABC) (Catalog # SA1022) with DAB as the chromogen.
Click image to see more details
IF analysis of Villi using anti-Villi antibody (PB9457)
Villi was detected in paraffin-embedded section of human rectal cancer tissues. Heat mediated antigen retrieval was performed in citrate buffer (pH6, epitope retrieval solution ) for 20 mins. The tissue section was blocked with 10% goat serum. The tissue section was then incubated with 1μg/mL rabbit anti-Villi Antibody (PB9457) overnight at 4°C. DyLight488 Conjugated Goat Anti-Rabbit IgG (BA1127) was used as secondary antibody at 1:100 dilution and incubated for 30 minutes at 37°C. The section was counterstained with DAPI. Visualize using a fluorescence microscope and filter sets appropriate for the label used.

Click image to see more details
IF analysis of Villi using anti-Villi antibody (PB9457)
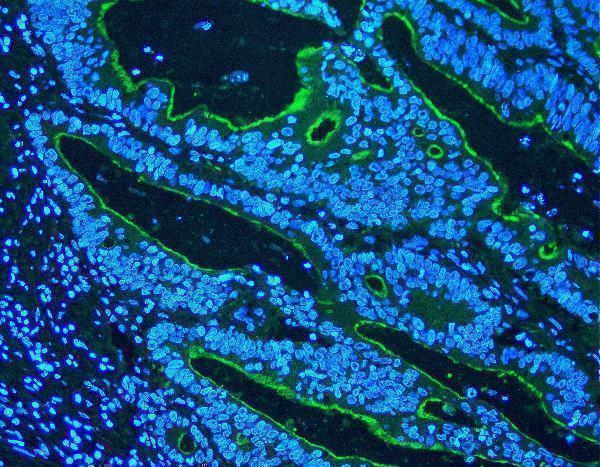
Villi was detected in paraffin-embedded section of rat intestine tissues. Heat mediated antigen retrieval was performed in citrate buffer (pH6, epitope retrieval solution ) for 20 mins. The tissue section was blocked with 10% goat serum. The tissue section was then incubated with 1μg/mL rabbit anti-Villi Antibody (PB9457) overnight at 4°C. DyLight488 Conjugated Goat Anti-Rabbit IgG (BA1127) was used as secondary antibody at 1:100 dilution and incubated for 30 minutes at 37°C. The section was counterstained with DAPI. Visualize using a fluorescence microscope and filter sets appropriate for the label used.
Click image to see more details
IF analysis of Villi using anti-Villi antibody (PB9457)
Villi was detected in paraffin-embedded section of mouse intestine tissues. Heat mediated antigen retrieval was performed in citrate buffer (pH6, epitope retrieval solution ) for 20 mins. The tissue section was blocked with 10% goat serum. The tissue section was then incubated with 1μg/mL rabbit anti-Villi Antibody (PB9457) overnight at 4°C. DyLight488 Conjugated Goat Anti-Rabbit IgG (BA1127) was used as secondary antibody at 1:100 dilution and incubated for 30 minutes at 37°C. The section was counterstained with DAPI. Visualize using a fluorescence microscope and filter sets appropriate for the label used.

Click image to see more details
IF analysis of Villin using anti-Villin antibody (PB9457).
Villin was detected in paraffin-embedded section of human ileum tissue. Heat mediated antigen retrieval was performed in citrate buffer (pH6, epitope retrieval solution) for 20 mins. The tissue section was blocked with 10% goat serum. The tissue section was then incubated with 5μg/mL rabbit anti-Villin Antibody (PB9457) overnight at 4°C. DyLight®488 Conjugated Goat Anti-Rabbit IgG (BA1127) was used as secondary antibody at 1:100 dilution and incubated for 30 minutes at 37°C. The section was counterstained with DAPI. Visualize using a fluorescence microscope and filter sets appropriate for the label used.

Click image to see more details
IF analysis of Villin using anti-Villin antibody (PB9457).
Villin was detected in paraffin-embedded section of human colon organoid tissue. Heat mediated antigen retrieval was performed in citrate buffer (pH6, epitope retrieval solution) for 20 mins. The tissue section was blocked with 10% goat serum. The tissue section was then incubated with 5μg/mL rabbit anti-Villin Antibody (PB9457) overnight at 4°C. DyLight®488 Conjugated Goat Anti-Rabbit IgG (BA1127) was used as secondary antibody at 1:100 dilution and incubated for 30 minutes at 37°C. The section was counterstained with DAPI. Visualize using a fluorescence microscope and filter sets appropriate for the label used.

Click image to see more details
IF analysis of Villin using anti-Villin antibody (PB9457).
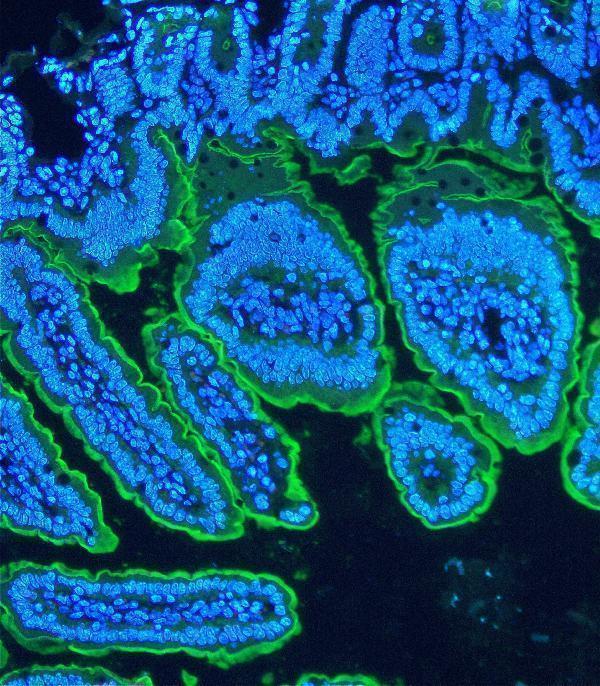
Villin was detected in paraffin-embedded section of mouse ileum tissue. Heat mediated antigen retrieval was performed in citrate buffer (pH6, epitope retrieval solution) for 20 mins. The tissue section was blocked with 10% goat serum. The tissue section was then incubated with 5μg/mL rabbit anti-Villin Antibody (PB9457) overnight at 4°C. DyLight®488 Conjugated Goat Anti-Rabbit IgG (BA1127) was used as secondary antibody at 1:100 dilution and incubated for 30 minutes at 37°C. The section was counterstained with DAPI. Visualize using a fluorescence microscope and filter sets appropriate for the label used.
Click image to see more details
IF analysis of Villin using anti-Villin antibody (PB9457).
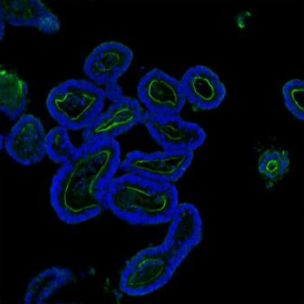
Villin was detected in paraffin-embedded section of mouse ileum organoid tissue. Heat mediated antigen retrieval was performed in citrate buffer (pH6, epitope retrieval solution) for 20 mins. The tissue section was blocked with 10% goat serum. The tissue section was then incubated with 5μg/mL rabbit anti-Villin Antibody (PB9457) overnight at 4°C. DyLight®488 Conjugated Goat Anti-Rabbit IgG (BA1127) was used as secondary antibody at 1:100 dilution and incubated for 30 minutes at 37°C. The section was counterstained with DAPI. Visualize using a fluorescence microscope and filter sets appropriate for the label used.
Click image to see more details
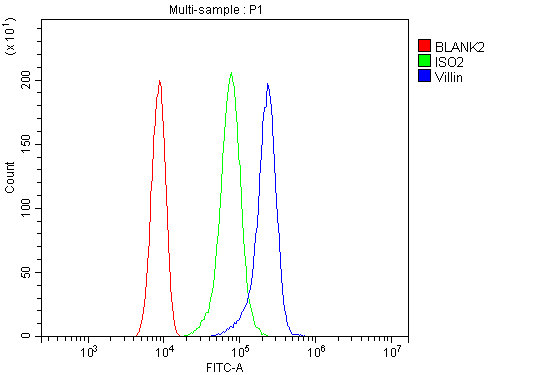
Flow Cytometry analysis of CACO-2 cells using anti-Villin antibody (PB9457).
Overlay histogram showing CACO-2 cells stained with PB9457 (Blue line). To facilitate intracellular staining, cells were fixed with 4% paraformaldehyde and permeabilized with permeabilization buffer. The cells were blocked with 10% normal goat serum. And then incubated with rabbit anti-Villin Antibody (PB9457, 1 μg/1x106 cells) for 30 min at 20°C. DyLight®488 conjugated goat anti-rabbit IgG (BA1127, 5-10 μg/1x106 cells) was used as secondary antibody for 30 minutes at 20°C. Isotype control antibody (Green line) was rabbit IgG (1 μg/1x106) used under the same conditions. Unlabelled sample without incubation with primary antibody and secondary antibody (Red line) was used as a blank control.
Specific Publications For Anti-Villin/VIL1 Antibody Picoband® (PB9457)
Loading publications
Recommended Resources
Here are featured tools and databases that you might find useful.
- Boster's Pathways Library
- Protein Databases
- Bioscience Research Protocol Resources
- Data Processing & Analysis Software
- Photo Editing Software
- Scientific Literature Resources
- Research Paper Management Tools
- Molecular Biology Software
- Primer Design Tools
- Bioinformatics Tools
- Phylogenetic Tree Analysis
Customer Reviews
Have you used Anti-Villin/VIL1 Antibody Picoband®?
Share your experimental results or join a short interview to earn up to $1,000 in product credits or other rewards.
0 Reviews For Anti-Villin/VIL1 Antibody Picoband®
Customer Q&As
Have a question?
Find answers in Q&As, reviews.
Can't find your answer?
Submit your question
3 Customer Q&As for Anti-Villin/VIL1 Antibody Picoband®
Question
Is PB9457 conjugated with colors and what color is it conjugated with?
Verified customer
Asked: 2022-06-13
Answer
The Anti-Villin/VIL1 Antibody Picoband™ (PB9457) is unconjugated. Please see the FCM image and image editing on the product page. https://www.bosterbio.com/anti-villin-picoband-trade-antibody-pb9457-boster.html
Boster Scientific Support
Answered: 2022-06-14
Question
How much concentration of PB9457 is used to generate the IHC(P) images in the product page?
Verified customer
Asked: 2019-02-27
Answer
The concentration of Anti-Villin/VIL1 Antibody Picoband™ PB9457 used to generate the IHC(P) images is 1ug/mL.
Boster Scientific Support
Answered: 2019-02-28
Question
We are currently using anti-Villin/VIL1 antibody PB9457 for mouse tissue, and we are happy with the IHC-P results. The species of reactivity given in the datasheet says human, mouse, rat. Is it likely that the antibody can work on horse tissues as well?
Verified Customer
Verified customer
Asked: 2019-01-25
Answer
The anti-Villin/VIL1 antibody (PB9457) has not been tested for cross reactivity specifically with horse tissues, but there is a good chance of cross reactivity. We have an innovator award program that if you test this antibody and show it works in horse you can get your next antibody for free. Please contact me if I can help you with anything.
Boster Scientific Support
Answered: 2019-01-25





