This website uses cookies to ensure you get the best experience on our website.
- Table of Contents
and ELISA kits, proteins related to NK-cells.
Natural Killer (NK) cells are a vital component of the innate immune system, renowned for their ability to identify and eliminate infected or malignant cells without prior sensitization. Unlike other immune cells, NK cells operate swiftly, providing the first line of defense against a wide range of threats, including viruses and cancerous transformations. Their unique mechanism of action involves recognizing stressed cells through a balance of activating and inhibitory receptors, ensuring targeted and efficient responses. Recent advancements in immunotherapy have spotlighted NK cells as promising candidates for antibody-based treatments, leveraging their natural cytotoxic abilities to enhance therapeutic efficacy. Our cutting-edge research focuses on harnessing and optimizing NK-cell related antibodies to develop innovative therapies, aiming to revolutionize treatments for various diseases and improve patient outcomes.
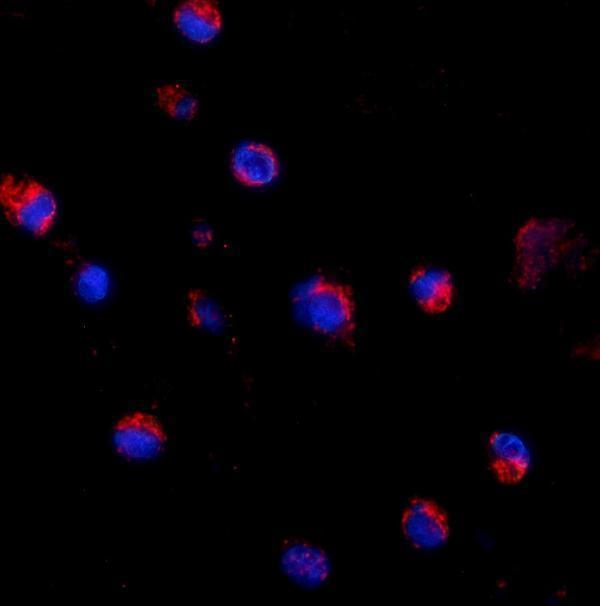
Anti-CD69 Antibody Picoband®, Figure 3. IF analysis of CD69 using anti-CD69 antibody (A00529-2).
CD69 was detected in immunocytochemical section of K562 cells. Enzyme ant...
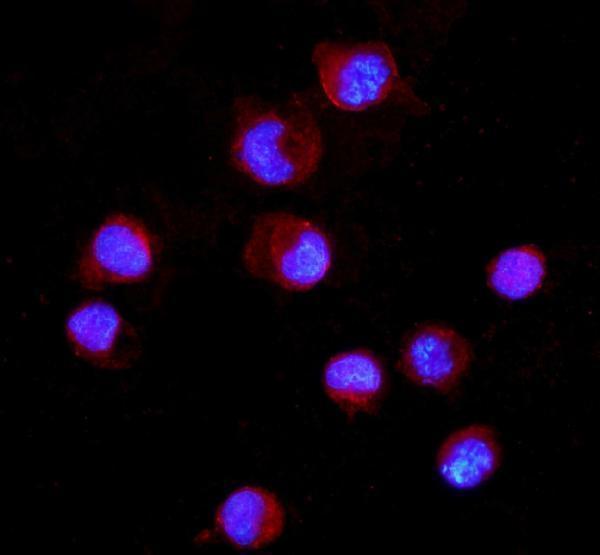
Anti-CD16/FCGR3A Antibody, Figure 4. IF analysis of CD16 using anti-CD16 antibody (A01408-1).
CD16 was detected in an immunocytochemical section of K562 cells. Enzyme antigen...

Anti-NCAM1 Antibody Picoband®, Figure 2. IF analysis of NCAM1 using anti-NCAM1 antibody (A00184-4).
NCAM1 was detected in immunocytochemical section of U20S cells. Enzym...
| Protein Name | Gene Name | Function |
|---|---|---|
| CD56 | NCAM1 | Cell adhesion molecule important for NK cell identification and cytotoxicity. |
| NKG2D | KLRK1 | Activating receptor that recognizes stress-induced ligands on target cells. |
| CD16 | FCGR3A | Fc receptor involved in antibody-dependent cellular cytotoxicity (ADCC). |
| NKp30 | NCR3 | Natural cytotoxicity receptor involved in NK cell activation and tumor cell recognition. |
| NKp46 | NCR1 | Natural cytotoxicity receptor critical for NK cell-mediated killing of virus-infected cells. |
| NKG2A | KLRC1 | Inhibitory receptor that recognizes HLA-E, regulating NK cell activity. |
| CD94 | KLRC2 | Partner of NKG2A, involved in inhibitory signaling upon binding HLA-E. |
| KIR2DL1 | KIR2DL1 | Inhibitory killer-cell immunoglobulin-like receptor recognizing HLA-C2 group alleles. |
| KIR3DL1 | KIR3DL1 | Inhibitory receptor targeting HLA-Bw4 alleles, regulating NK cell inhibition. |
| CD69 | CD69 | Activation marker indicating recent activation of NK cells. |
| CD25 | IL2RA | Alpha chain of IL-2 receptor, involved in NK cell proliferation and activation. |
| Granzyme B | GZMB | Serine protease involved in inducing apoptosis in target cells. |
| Perforin | PRF1 | Pore-forming protein facilitating entry of granzymes into target cells. |
| CD107a | LAMP1 | Marker of degranulation indicating NK cell cytotoxic activity. |
| TIM-3 | HAVCR2 | Immune checkpoint receptor regulating NK cell exhaustion and function. |
| LAG-3 | LAG3 | Immune checkpoint molecule involved in NK cell regulation and inhibition. |
| CD57 | B3GAT1 | Marker of terminal differentiation and senescence in NK cells. |
| ICOS | ICOS | Costimulatory molecule enhancing NK cell proliferation and cytokine production. |
| PD-1 | PDCD1 | Immune checkpoint receptor modulating NK cell activity in tumor environments. |
| BTLA | BTLA | Inhibitory receptor regulating NK cell function and immune tolerance. |
| Siglec-7 | SIGLEC7 | Inhibitory receptor recognizing sialic acid-containing ligands, regulating NK cell activation. |
Natural Killer (NK) cell activation and inhibition constitute a finely tuned balance that determines their responsiveness to target cells. NK cells possess a repertoire of activating and inhibitory receptors on their surface, enabling them to distinguish between healthy cells and those infected by pathogens or undergoing malignant transformation. Activating receptors, such as NKG2D and NKp30, recognize stress-induced ligands on target cells, triggering cytotoxic responses. Conversely, inhibitory receptors like KIRs (Killer-cell Immunoglobulin-like Receptors) and NKG2A bind to MHC class I molecules typically expressed on healthy cells, sending inhibitory signals that prevent unwarranted NK cell activation. This dual receptor system ensures that NK cells can effectively eliminate abnormal cells while sparing normal, healthy tissues. Understanding the molecular mechanisms governing these pathways is crucial for developing therapeutic strategies that can enhance NK cell activity against cancers and viral infections without compromising immune tolerance.
NK cells play a pivotal role in the immune surveillance system through their ability to induce apoptosis in target cells. This cytotoxic function is primarily mediated by the release of cytotoxic granules containing perforin and granzymes. Perforin forms pores in the membrane of target cells, allowing granzymes to enter and activate apoptotic pathways, leading to cell death. Additionally, NK cells can induce apoptosis via death receptor pathways, such as Fas-FasL interactions. Beyond direct killing, NK cells secrete cytokines like IFN-γ and TNF-α, which modulate the immune response and enhance the recruitment and activation of other immune cells. The efficiency and regulation of these cytotoxic mechanisms are vital for controlling tumor growth and eliminating virally infected cells. Research in this area focuses on harnessing and enhancing NK cell cytotoxicity for therapeutic interventions, including cancer immunotherapy and treatments for persistent viral infections.