This website uses cookies to ensure you get the best experience on our website.
- Table of Contents
and ELISA kits, proteins related to Stem Cell.
Stem cells are the building blocks of life, possessing the remarkable ability to develop into various specialized cell types within the body. Their unique regenerative properties make them a cornerstone of cutting-edge medical research and therapies. In the realm of stem cell research, antibodies play a crucial role in identifying, isolating, and manipulating these versatile cells. By targeting specific proteins on stem cells, scientists can better understand their behavior, enhance their therapeutic potential, and advance treatments for a wide range of conditions, including degenerative diseases, cancer, and spinal cord injuries. Our comprehensive antibody solutions are designed to support your stem cell research endeavors, providing the tools necessary to unlock new possibilities in regenerative medicine and personalized healthcare. Explore the advancements in stem cell science with our high-quality antibodies tailored to accelerate your discoveries and innovations.
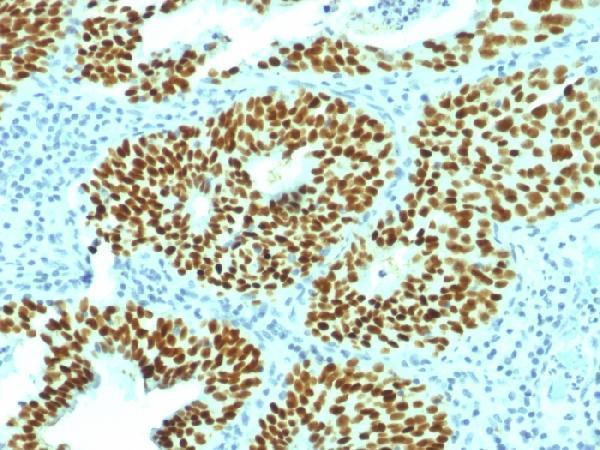
Anti-SOX2 (Transcription Factor) Monoclonal Antibody, Formalin-fixed, paraffin-embedded human Cervical Carcinoma stained with Anti-SOX2 Mouse Monoclonal Antibody (SOX2/1792)....
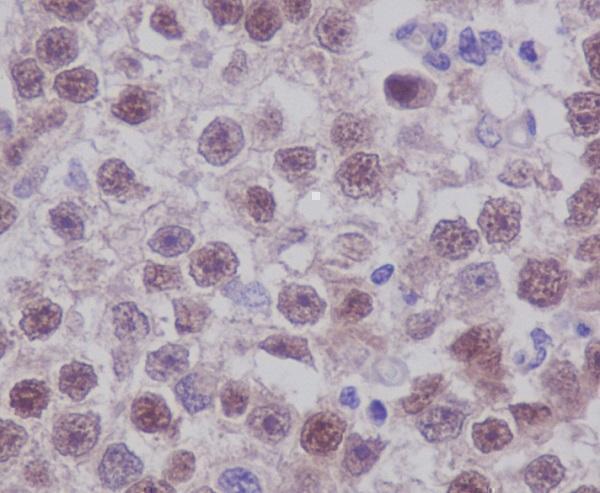
Anti-Oct4 POU5F1 Rabbit Monoclonal Antibody, Immunohistochemical analysis of paraffin-embedded human seminoma, using Oct4 Antibody. ....
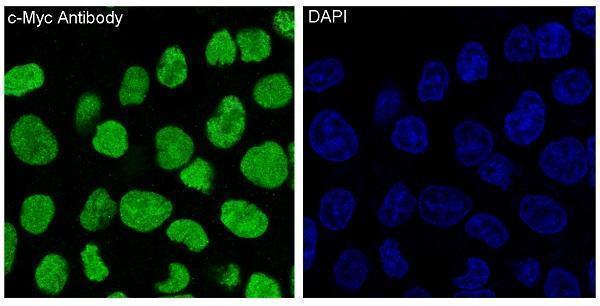
Anti-c-Myc Monoclonal Antibody, Immunofluorescent analysis of HeLa cells, using c-Myc Antibody....
| Protein Name | Gene Name | Function |
|---|---|---|
| OCT4 | POU5F1 | Maintains pluripotency and self-renewal in embryonic stem cells |
| SOX2 | SOX2 | Regulates self-renewal and pluripotency in stem cells |
| NANOG | NANOG | Critical for maintaining pluripotency and preventing differentiation |
| KLF4 | KLF4 | Transcription factor involved in maintaining pluripotency and reprogramming |
| c-MYC | MYC | Regulates proliferation and growth; involved in stem cell self-renewal |
| CD34 | CD34 | Marker for hematopoietic stem cells |
| CD73 | NT5E | Involved in immunosuppression and stem cell maintenance |
| CD90 | THY1 | Marker for mesenchymal stem cells |
| CD105 | ENG | Endoglin, involved in angiogenesis and stem cell function |
| LGR5 | LGR5 | Marker for adult stem cells, particularly in the intestine |
| ABCG2 | ABCG2 | Efflux transporter associated with stem cell 'side population' |
| KIT | KIT | Receptor tyrosine kinase important for hematopoietic stem cell maintenance |
| ALDH1 | ALDH1A1 | Enzyme involved in detoxification, marker for stem cells |
| Nestin | NES | Intermediate filament protein used as a neural stem cell marker |
| VCAM1 | VCAM1 | Cell adhesion molecule, involved in stem cell niches |
| Integrin α6 | ITGA6 | Cell surface receptor involved in stem cell adhesion and signaling |
| E-cadherin | CDH1 | Cell-cell adhesion molecule important for maintaining pluripotency |
Induced Pluripotent Stem Cells (iPSCs) represent a groundbreaking advancement in stem cell research. These cells are generated by reprogramming adult somatic cells, such as skin or blood cells, back into a pluripotent state, which means they have the capability to differentiate into almost any cell type in the body. This technology, pioneered by Shinya Yamanaka, has revolutionized the field by providing a versatile and ethically acceptable alternative to embryonic stem cells. iPSCs hold immense potential for personalized medicine, as they can be derived from a patient's own cells, reducing the risk of immune rejection in therapeutic applications. Additionally, iPSCs serve as invaluable models for studying disease mechanisms, enabling researchers to investigate the development and progression of various genetic and acquired disorders in a controlled laboratory setting. The ability to generate patient-specific cell lines also facilitates drug screening and the discovery of novel treatments. Despite their promise, challenges such as ensuring the genetic stability and safety of iPSC-derived cells must be addressed before widespread clinical application. Ongoing research continues to refine reprogramming techniques and enhance the efficiency and reliability of iPSC technology, paving the way for transformative advancements in regenerative medicine and personalized healthcare.
Hematopoietic Stem Cells (HSCs) are a critical subfield within stem cell research, focusing on the formation and maintenance of blood cells. HSCs reside primarily in the bone marrow and possess the unique ability to self-renew and differentiate into all types of blood cells, including red blood cells, white blood cells, and platelets. This versatility makes them indispensable for the continuous replenishment of the blood and immune system throughout an individual's life. Research on HSCs has profound implications for treating a variety of hematological disorders, such as leukemia, lymphoma, and anemia, through bone marrow transplantation and gene therapy. Advances in HSC research have also enhanced our understanding of blood cell development and the regulatory mechanisms governing stem cell behavior and differentiation. Moreover, HSCs serve as a model for studying stem cell niches and the interactions between stem cells and their microenvironment, shedding light on broader principles of stem cell biology. Ongoing studies aim to improve HSC expansion techniques, enhance engraftment efficiency, and minimize complications associated with transplantation. By unlocking the full potential of HSCs, scientists hope to develop more effective and targeted therapies for blood-related diseases and contribute to the broader goals of regenerative medicine.