This website uses cookies to ensure you get the best experience on our website.
- Table of Contents
Affinity gauges the binding strength of a single bond, while avidity measures the combined stability of multivalent interactions. Understanding the difference enhances assay design, vaccine evaluation, and therapeutic efficacy.
Affinity vs avidity describes how tightly antibody attach to antigens, and together, these values predict real-world performance. High-affinity IgG molecules often show a low dissociation rate constants (Kd) below 10⁻⁹ M, yet even weaker binding sites can yield strong avidity when arranged on a pentameric IgM. Roughly 80% of early infection tests rely on avidity principles, whereas most monoclonal antibodies undergo affinity maturation before approval, a process supported by our antibody production services. This reflects a fundamental aspect of molecular interactions essential in diagnostics, drug development, and biomedical research.
A single antigen-antibody reaction can decide whether a pathogen infects a cell membrane or is cleared from it. Strong binding reduces the likelihood of escape, enhances diagnostic sensitivity, and can prolong drug residence time. Therefore, scientists track both affinity—the force at a single binding site—and avidity—the collective force from immune complexes formed by multiple bonds.

Fig.1 Illustrating antigen-antibody interaction, comparing affinity and avidity. Shows a monovalent antibody binding to a single epitope (affinity) alongside a multivalent antibody binding to multiple epitopes on a pathogen surface (avidity).
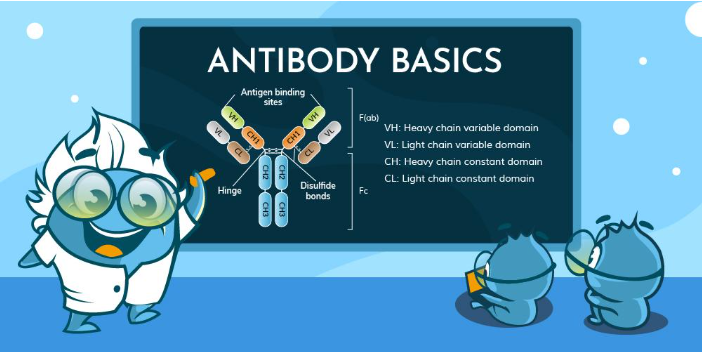
Affinity is the attraction between one antigen-binding site and one epitope. It reflects how quickly the complex forms (association rate constant)and how slowly it breaks apart (dissociation rate constant). Strong affinity implies a low Kd, often 10⁻⁹ M or lower for mature IgG. Small mutations in complementarity-determining regions can shift affinity by a thousand-fold due to changes in hydrogen bonds and hydrophobic interactions at the binding interface.
Researchers choose different tools depending on throughput and sensitivity:
High-affinity antibodies improve assay specificity because weak binders wash away during stringent steps in Western blotting or ELISA.
Avidity combines multiple affinities. An IgM pentamer, with ten identical Fab fragments, can latch onto a multivalent antigen structure even if each site binds weakly. Cooperative binding stabilizes complexes, especially when antigens display repeating epitopes. This process integrates Bond Strength with structural arrangement to resist dissociation.
Laboratories often use an avidity ELISA in which samples bound to the antigen are washed with a chaotropic agent, such as 6 M urea. The avidity index equals the signal retained after treatment divided by the signal before treatment, expressed as a %.
Clinicians apply this in diagnosing rubella, toxoplasmosis, and other infections, differentiating acute from past exposure in the humoral immune response.
Affinity and avidity describe related but distinct aspects of molecular binding, and understanding their differences is essential for interpreting experimental results.
| Factor | Affinity | Avidity |
|---|---|---|
| Definition | Strength of a single binding site | Combined strength of multiple sites |
| Unit | Dissociation rate constant (Kd) | Avidity index or functional strength |
| Influenced by | Amino-acid contacts, shape fit | Valency, epitope density, flexibility |
| Main role | Determines specificity | Enhances binding stability |
| Primary assay | SPR, BLI, equilibrium ELISA | Avidity ELISA, functional tests |
These practical examples show how affinity and avidity influence diagnostic accuracy, therapeutic design, and vaccine assessment in real-world applications.
Rapid tests for early infection often detect IgM because its high avidity yields visible agglutination. For confirmation, labs switch to IgG affinity tests that discriminate true positives from antibody cross reactivity and false signals.
Drug developers raise affinity through mutagenesis and selection cycles. Values below 10⁻¹¹ M increase target occupancy at lower doses. In contrast, bispecific antibodies sometimes leverage avidity, arranging two weaker arms to reduce off-target effects while maintaining strong overall attachment, an approach enabled by custom monoclonal antibodies.
This process considers antibody concentration, functional affinity, and how each antibody molecule binds and remains stable within antibody-antigen complexes.
After vaccination, affinity matures through somatic hypermutation, and avidity rises as antibodies switch from IgM to IgG. Monitoring the avidity index helps researchers predict the durability of protection and decide whether booster shots are necessary.
Selecting the appropriate assay depends on the research objective. When detailed binding kinetics are required, techniques such as Surface plasmon resonance (SPR) or bio-layer interferometry (BLI) provide real-time measurements of the association rate constant and dissociation rate constant. For high-throughput screening of multiple antibody clones, equilibrium ELISA is often the first step, with promising candidates later validated by SPR for precise affinity data. In infectious disease studies, avidity ELISAs that incorporate chaotropic washes help distinguish recent infections from long-term immunity by analyzing the stability of antigen–antibody complexes. For mucosal immunity, IgA assays are essential, as secretory IgA exhibits unique resistance to enzymatic digestion, offering insights into protective responses at epithelial barriers.
Affinity and avidity together describe how antibodies interact with antigens, guiding numerous decisions in diagnostics, research, and medicine. Focusing solely on one value can be misleading; a balanced evaluation ensures accurate conclusions and successful product development.

Are you looking to refine your assays, develop high-performance antibodies, or showcase new findings?
Submit an inquiry to connect with our expert team. Discover how tailored assay design, collaborative research, and exhibition opportunities can enhance your projects and foster new partnerships.