This website uses cookies to ensure you get the best experience on our website.
- Table of Contents

In most FFPE IHC workflows, antigen retrieval through HIER is the better first retrieval method. The more useful question is not whether antigen retrieval matters, but whether heat-based epitope retrieval is enough for your target and tissue—or whether enzymatic retrieval is the smarter next step.
That distinction matters because weak or missing staining is not always an primary antibody problem. It may reflect a retrieval method that does not match the fixation history, tissue fixation approach such as formalin fixation, tissue condition, or epitope behavior. At the same time, changing methods too early can create unnecessary trial-and-error. If heat-based retrieval still preserves tissue integrity in formalin-fixed paraffin-embedded samples, it usually makes more sense to optimize that setup before moving to enzyme digestion. If heat is already compromising morphology or section stability in tissue sections, enzymatic retrieval becomes a more logical test.
If you need a broader refresher on why retrieval is needed at all, this article should complement—not repeat—your background guide on antigen retrieval in immunohistochemical assays.
For routine FFPE IHC staining, HIER is usually the most practical place to start because it is easier to standardize within repeatable IHC protocols. If the initial stain is weak, you can still optimize several variables within the same retrieval strategy, including buffer choice such as sodium citrate, pH, heating strength, and retrieval time.
That makes HIER especially useful when you are building a stable workflow across multiple tissues, projects, or staining runs in molecular biology applications. A weak result after HIER does not automatically mean heat retrieval was the wrong choice. In many cases, it simply means the conditions were too mild, too short, or not well matched to the target.
This is also why HIER fits naturally into a broader IHC sample preparation and immunoassay protocols workflow. If heat retrieval is directionally correct, refining the setup is often more productive than switching method classes too early.
Enzymatic retrieval becomes more worth testing when heat is part of the problem rather than part of the solution.
In these cases, enzymatic retrieval is not just an alternative for the sake of variety. It is a different strategy for antigen unmasking while reducing thermal stress on the sample.
This is also where upstream sample conditions matter more. Retrieval choice is easier to judge when you consider the broader context of fixatives used in IHC and ICC and overall pre-staining conditions. If fixation or sample handling is already working against epitope accessibility, retrieval performance can be harder to interpret and may contribute to a broader staining issue.
One of the most common mistakes in IHC optimization is treating a weak first result as proof that the retrieval method itself was wrong.
If you used HIER and the tissue still looks intact, do not jump to enzyme digestion immediately. First ask a narrower question: did you test HIER broadly enough to make that call? If not, optimizing within HIER is often the faster and cleaner next step, including adjusting antibody dilution, PBS buffer conditions, or detection reagents.
By contrast, if HIER is clearly reducing morphology quality, weakening section stability, or making the workflow harder to reproduce, that result tells you more. In that situation, the next move should not be “more heat, but different.” It should be a different retrieval logic.
This is also where retrieval should remain part of a broader troubleshooting mindset. A weak or negative stain may involve retrieval, but it may also reflect secondary antibody performance, signal amplification limits, or blocking issues such as endogenous peroxidase or endogenous biotin interference. These factors can contribute to background staining or poor signal clarity.
In most routine FFPE workflows, optimizing HIER first is still the more practical path. But not every project has the time for a broad HIER matrix covering multiple buffers, pH conditions, and retrieval windows.
If you are working under time pressure and already have strong literature support, prior validation data, or known tissue-specific reasons to avoid heat-heavy optimization, testing enzymatic retrieval earlier may be the more efficient choice. This is often relevant in studies involving autoimmune diseases or targets like the estrogen receptor, where prior validation may guide method selection. The same applies when sample history already suggests that heat is unlikely to be the best fit.
In other words, the most economical decision is not always to exhaust every HIER variation. Sometimes it is to move earlier toward the retrieval method with the strongest existing rationale. That is not cutting corners. It is making a time-aware optimization decision.
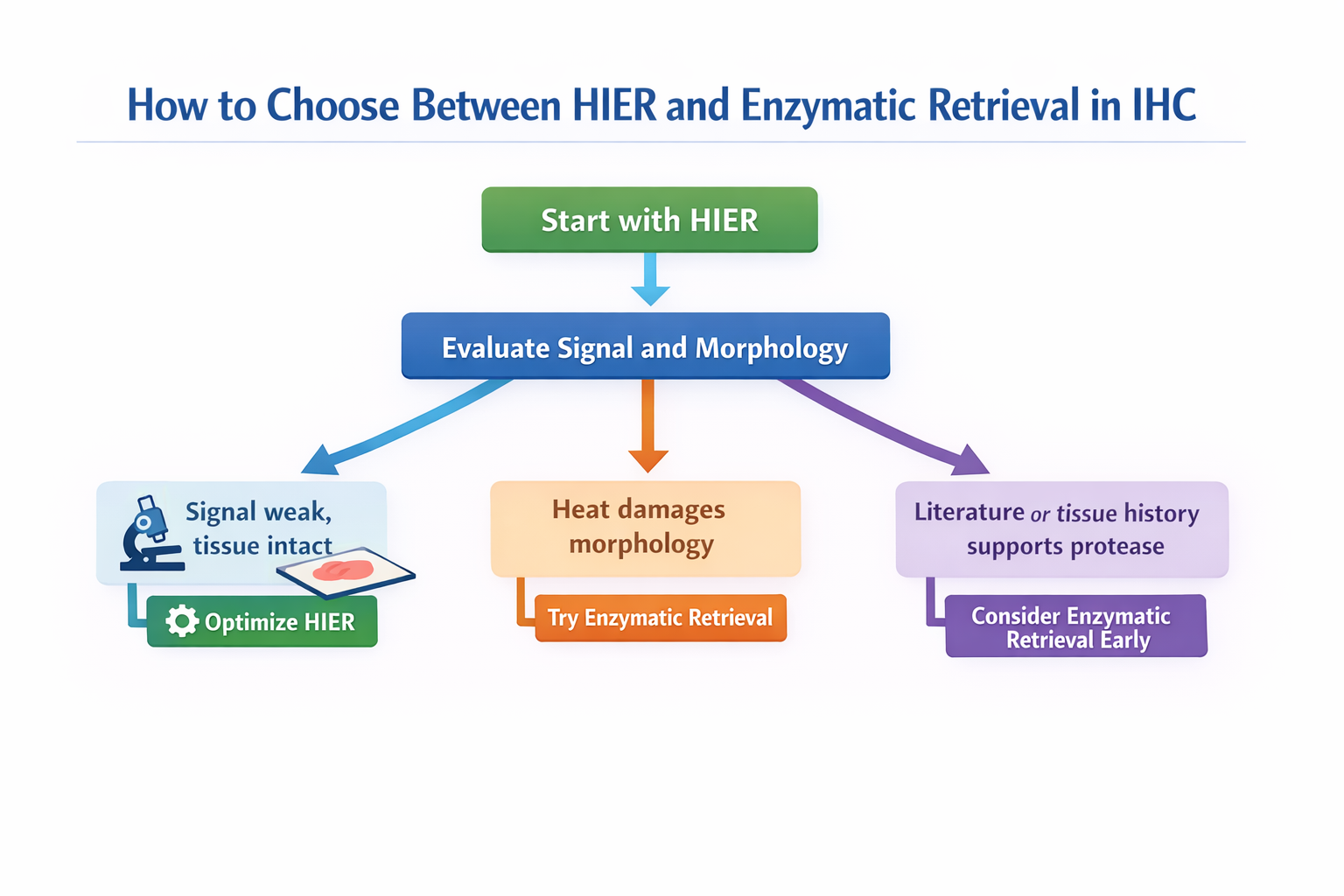
A simple decision path for choosing between HIER and enzymatic retrieval in IHC.
| If your result looks like this | Better next move |
|---|---|
| Routine FFPE tissue, first-round setup | Start with HIER |
| Weak signal, but tissue remains intact | Optimize HIER first |
| Heat damages morphology or section stability | Test enzymatic retrieval |
| Literature or antibody guidance supports protease digestion | Consider enzymatic retrieval earlier |
| Enzyme treatment over-digests tissue | Reduce digestion strength or reassess method fit |
This kind of decision guide is often more useful than a longer theory section because it matches how people actually troubleshoot in the lab.
Antigen retrieval is not an isolated step. It interacts with fixation, sample preparation, controls, and interpretation. A poor retrieval choice can create downstream problems that look like monoclonal antibody failure, weak antibody staining, or inconsistent staining quality. It can also introduce tissue artifacts or issues related to sectioning tissue and slide storage.
That is why retrieval decisions should be made in the context of the full IHC workflow, not as a standalone technical preference. Readers who want to step back and review the bigger picture can move from here into 6 IHC controls you should know, the broader IHC troubleshooting hub, or the full Immunohistochemistry IHC Technical Resource Center. Many labs, including those using platforms from Leica Biosystems, emphasize consistency across the entire workflow rather than optimizing a single step in isolation.
Is HIER always the default choice for FFPE IHC?
Not always, but it is usually the better starting point for routine FFPE IHC because it is easier to standardize and optimize. Enzymatic retrieval becomes more attractive when heat damages tissue quality or when strong prior evidence supports protease digestion.
When should I try enzymatic retrieval earlier instead of optimizing HIER further?
Consider it earlier when you are under time pressure and already have strong literature support, prior validation data, or sample-specific reasons to think heat-based retrieval is a poor fit.
What does “heat damages morphology” usually look like in practice?
It often shows up as easier section lifting, less distinct cell boundaries, reduced nuclear detail, or poorer preservation of overall tissue architecture after retrieval.
If staining is weak after HIER, should I switch methods immediately?
Not necessarily. If tissue integrity is still good, optimizing HIER first is often the better next step.
If you need a reliable default answer, start with HIER in most FFPE IHC workflows. Move toward enzymatic retrieval when heat damages tissue integrity, fails to recover the target after reasonable testing, or is already a poor fit for the sample.
The right retrieval method is not the harsher one or the more specialized one. It is the method that improves antigen access without creating a new problem somewhere else in the assay.