This website uses cookies to ensure you get the best experience on our website.
- Table of Contents
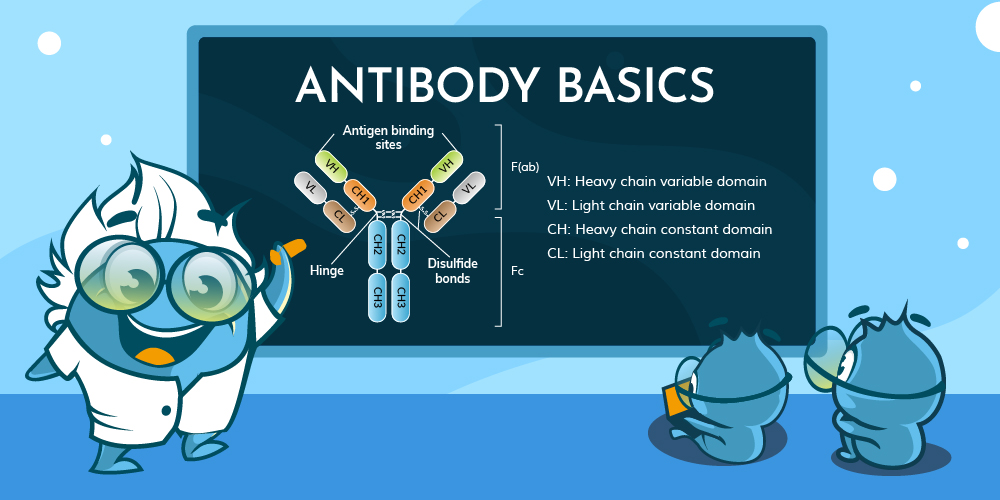
You may have heard of antibodies in everyday life—perhaps in the context of vaccines, immune responses, or even medical tests. Whether it’s a doctor explaining how your body fights off infections or a news article discussing breakthrough antibody therapies, these tiny but powerful molecules play a crucial role in health and science.
Now, as you step into the world of antibodies, we’re here to guide you through the fundamentals. Welcome to your journey into antibody biology—where science meets discovery.
Let’s find out!
1.1 Definition of Antibody
An antibody is a special type of protein in the immune
system of vertebrates, produced and secreted by B cells. Also known as Immunoglobulin
(Ig), antibodies recognize and specifically bind to antigens (such as
proteins or polysaccharides on pathogens), triggering immune responses to
eliminate foreign threats and abnormal cells. Antibodies are widely distributed
in the body and can be found in blood, lymphatic fluid, and tissue fluid.
1.2 Basic Structure of
Antibodies
Antibodies have a Y-shaped structure composed of
two identical heavy chains (H chains) and two identical light chains
(L chains), connected by disulfide bonds.
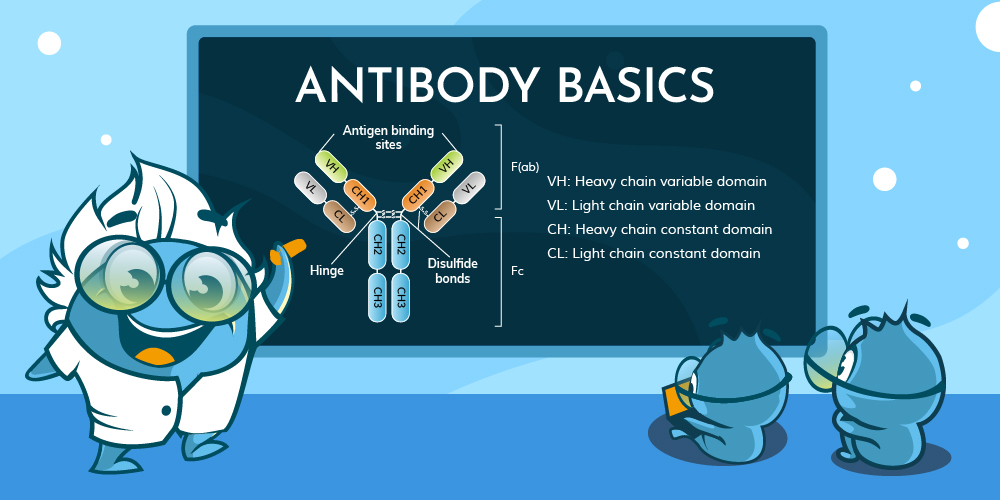
Just as different animals have developed a wide variety of external characteristics over the course of evolution, the antibodies within different organisms are also highly diverse. However, in mammals, antibodies can be classified into five major types based on their structural configuration. The amino acid sequences in the constant region of the heavy chain and the hinge region directly influence the spatial conformation of the antibody, while the number and position of disulfide bonds determine the inter-chain connections and overall folding of the antibody.
2.1 Antibody Types in Mammals
Mammals produce five main types of antibodies, each with
distinct structures and immune functions:
| Antibody Type | Molecular Features | Typical Conformation | Functional Adaptation |
|---|---|---|---|
| IgG | Gamma (γ) chain, long hinge region, monomer | Flexible "Y" shape | Efficiently neutralizes pathogens in the bloodstream |
| IgD | Delta (δ) chain, long hinge region, monomer | Flexible "Y" shape | Functions mainly as a B cell receptor, playing a role in B cell maturation and activation |
| IgE | Epsilon (ε) chain, long hinge region, monomer | Extended "Y" shape | Mediates allergic reactions by binding to mast cells and basophils, triggering inflammatory responses |
| IgA | Alpha (α) chain, short hinge region, dimer (mainly) or monomer | "Dumbbell-shaped" dimer | Primarily found in mucosal secretions, providing mucosal immune defense |
| IgM | Mu (μ) chain, no hinge region, pentamer | "Star-shaped" pentamer | Predominantly involved in initial immune response, enhancing agglutination and complement activation |
2.2 Antibody Types in Other Vertebrates
Different vertebrates have unique antibody types:
Jawless Vertebrates (e.g., lampreys): Lack traditional immunoglobulins but utilize Variable Lymphocyte Receptors (VLRs) for immune defense.
Under natural conditions, the production of antibodies is a fundamental process in the immune system’s recognition and response to pathogens. This process is typically divided into the primary immune response and the secondary immune response, which differ significantly in terms of response speed, antibody types, and the establishment of immune memory. The primary immune response occurs when the body encounters a specific antigen for the first time, whereas the secondary immune response is triggered upon re-exposure to the same antigen, leveraging immune memory to mount a faster and stronger defense.
3.1 Primary Immune Response
When the immune system first encounters an antigen, the following immune processes are initiated in turn:
3.2 Secondary Immune Response
Upon re-exposure to the same antigen, the immune pathway in the animal becomes shorter:
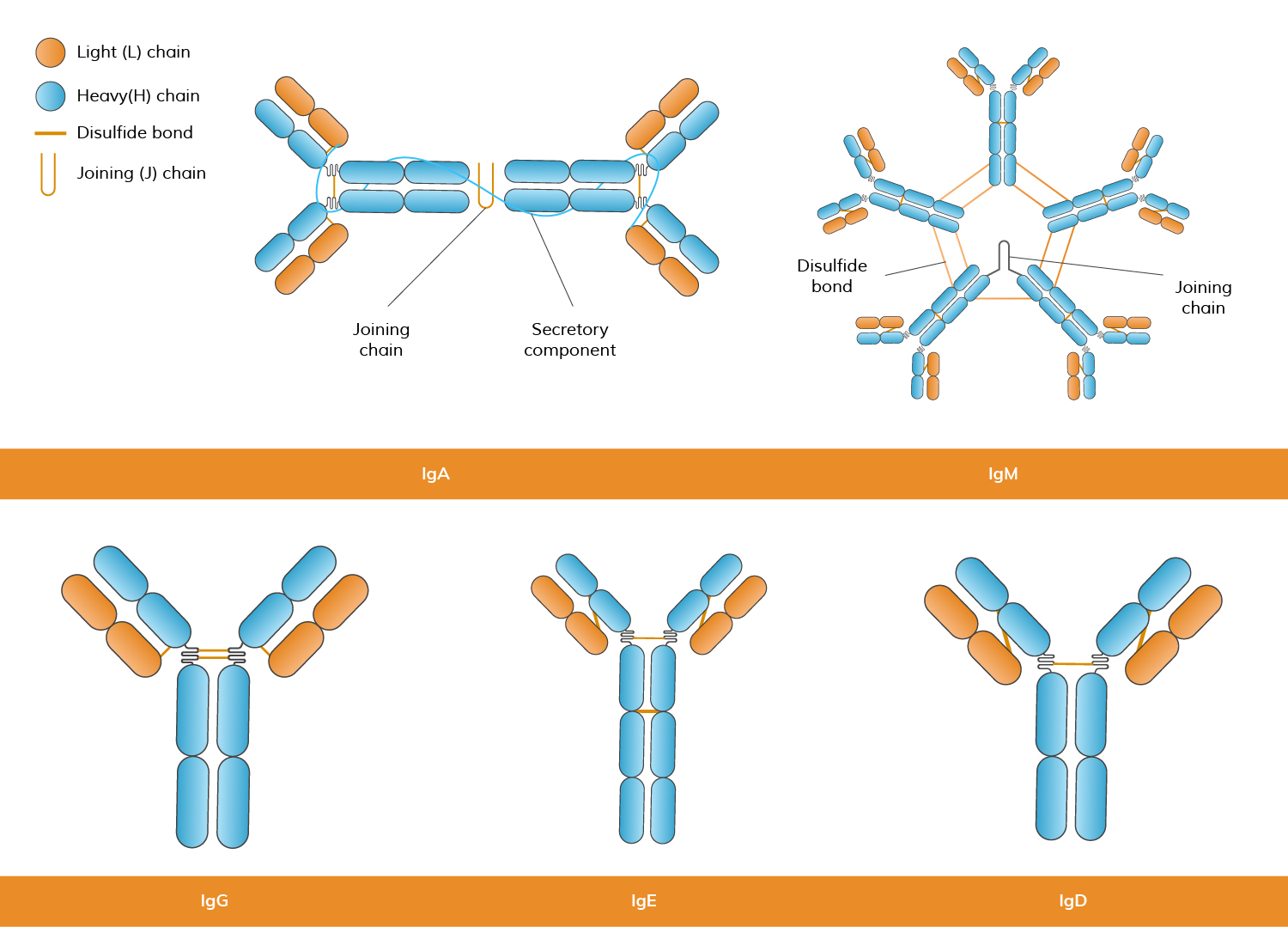
Polyclonal antibodies are produced by injecting an antigen into an animal (e.g., rabbit, goat, or sheep) to stimulate an immune response. The antibodies are then extracted from the serum.
4.1 Polyclonal Antibodies
(pAbs)
Polyclonal antibodies (pAbs) are a mixture of antibodies produced by different B-cell clones in response to a particular antigen. These antibodies recognize and bind to multiple epitopes on the same antigen, which can result in a broader immune response. Polyclonal antibodies are typically obtained by immunizing an animal (such as a rabbit or goat) and collecting the serum containing the diverse antibodies produced. For researchers who require customized reagents, polyclonal antibody production services
offer a reliable way to generate high-affinity antibodies tailored to specific antigens and applications. These antibodies are commonly used in research applications like Western blotting, immunohistochemistry, and ELISA, where multiple binding sites on an antigen may be beneficial.
4.2 Monoclonal Antibodies (mAbs)
Monoclonal antibodies (mAbs) are antibodies derived from a single clone of immune cells, meaning they are identical and target a specific epitope on an antigen. These antibodies are produced by hybridoma technology, where a single B-cell is fused with a myeloma cell to create a hybrid cell capable of producing large quantities of a single type of antibody. Among monoclonal antibodies, rabbit monoclonal antibodies are particularly valued for their ability to recognize epitopes that may be less immunogenic in other species. Their unique epitope binding profile and higher affinity often make them advantageous in experiments that require sensitive detection and precise target discrimination. Due to their specificity, custom monoclonal antibodies are highly effective in research, diagnostics, and therapeutic applications, such as cancer treatment, autoimmune diseases, and infectious diseases. In diagnostic assay development, particularly in formats like sandwich ELISA and lateral flow assays, the selection and optimization of antibody pairs are critical factors influencing assay sensitivity, specificity, and overall performance. Antibody pair development involves systematically screening and validating combinations of capture and detection antibodies to ensure they bind to distinct, non-overlapping epitopes, minimizing cross-reactivity and enhancing signal clarity. Antibody Pair Development Service often requires careful consideration of binding kinetics, antigen conformation, and matrix effects to achieve reliable and reproducible results.
| Category | Polyclonal Antibodies | Monoclonal Antibodies |
|---|---|---|
| Epitope Recognition | Multiple | Single |
| Stability | Good | Easily Affected |
| Sensitivity | High | Relatively high |
| Cross-reactivity | Possible | Less likely |
| Preparation Difficulty | Simple | More difficult |
| Preparation Time | Short | Long |
| Price | Relatively low | Higher |
4.3 Recombinant Antibodies
*Preparation Methods
*Characteristics
*Applications
4.4 Single Chain Antibodies
scFV
Single-chain antibodies (scFvs) are a type of antibody fragment that consist of the variable regions of both the heavy and light chains of an antibody, fused together into a single polypeptide chain. Unlike full-length antibodies, scFvs are much smaller, typically around 25 kDa, which allows for easier tissue penetration and faster clearance from the body. They maintain the ability to bind to specific antigens, making them useful in a variety of applications, including targeted therapy, diagnostic imaging, and as research tools. Due to their smaller size and versatility, scFvs offer significant advantages in drug delivery, cancer therapy, and biosensor development.
Antibodies are known for their ability to bind specifically to antigens. This kind of one-to-one corresponding structure similar to the key and lock makes the antibody in addition to fighting against foreign invaders in the biological body, but also has an immeasurable role in scientific research.
5.1
Mechanism of Action
Antibodies function by specifically binding to target
antigens. The key mechanisms include:
5.2 Direct Detection and Indirect Detection
In both experimental and clinical
settings, the role of antibodies goes beyond immune responses and is widely
used to detect the presence of target molecules or proteins. Common detection
methods include:
5.3 The Role of Secondary Antibodies
Secondary antibodies are
antibodies generated against another antibody (usually the primary antibody).
In biological experiments, secondary antibodies are typically conjugated with
enzymes (e.g., horseradish peroxidase, HRP, or alkaline phosphatase, AP),
fluorescent dyes (e.g., FITC, Cy3), or other markers. Their main functions
include:
5.4 The Diversity of Antibody Binding
The functions of antibodies are
not limited to simple binding and clearance. Using conjugation and label, such as those provided through our antibody conjugation service, they can extend to more complex biological processes, such as:
Immunotherapy: Monoclonal antibody drugs that block specific receptors or target and eliminate cancer cells for cancer treatment.
Finally, let's talk about how to judge the quality of antibodies. Specificity, Affinity and Batch-to-Batch Consistency are three common indicators of antibody specificity. Antibody performance may vary under different experimental conditions too. Boster provides high-quality antibody testing services, and also performs its own antibody testing for the antibodies produced by our company. Recently, we launched a free test service for some antibodies on the test method and sample specified by customers. Click the link at the end of the article to enjoy the free test service before purchasing antibodies.
6.1 Specificity Validation:
Specificity is one of the most
critical indicators of an antibody, referring to its ability to accurately
recognize and bind to the target antigen without cross-reacting with non-target
proteins. Specificity is typically validated across multiple experimental
platforms, such as Western Blot, immunohistochemistry (IHC), and
immunoprecipitation (IP). Ideally, high-quality antibodies should exhibit
clear, unique signals with low background noise in these applications. Many
manufacturers provide validation data across different cell lines, tissue
samples, and animal models, helping users determine the antibody's
applicability and guiding experimental design. For new users, understanding the
specificity data and references can prevent misinterpretation due to
non-specific binding.
6.2 Affinity Measurement:
Affinity reflects the strength
of the binding between the antibody and antigen. High-affinity antibodies can
maintain effective binding at low concentrations, improving detection
sensitivity and reliability. Common methods for measuring affinity include
surface plasmon resonance (SPR), ELISA, and competitive binding assays. The
affinity value (e.g., Kd value) provided by manufacturers serves as an
important reference for evaluating antibody performance. High-affinity
antibodies not only help reduce background signals but also make it easier to
capture trace amounts of target proteins in complex samples, laying a solid
foundation for subsequent quantitative analysis.
6.3 Batch-to-Batch Consistency:
Consistency between antibody
production batches is crucial for ensuring the stability of long-term
experimental results. Minor variations between batches can affect the
comparability of experimental data. High-quality manufacturers typically
establish strict quality control systems, thoroughly validate each batch, and
provide detailed technical documentation. Users should pay attention to batch
reports and quality control data when purchasing and, if necessary, conduct
internal pre-experiments to verify batch consistency, ensuring data reproducibility
and reliability.
6.4 Validation Data and Experimental Conditions:
Antibody performance may vary
under different experimental conditions. Factors such as fixation methods,
choice of blocking reagents, antigen modification status, and sample type can
all affect antibody binding. Therefore, understanding and referencing the
validation data provided by manufacturers is crucial. Ideally, antibodies
should perform well under multiple conditions. Researchers can use these data
to compare the performance of different antibodies and select the most suitable
product for their experimental system. Additionally, conducting pre-experiments
or small-scale validation before formal use is recommended to ensure
consistency between experimental conditions and validation data.
6.5 Common Challenges and Solutions:
Common challenges during
antibody validation include cross-reactivity, high background signals, and
antibody instability with target proteins in certain samples. To address these
issues, researchers should pay attention to appropriate antibody dilution,
optimize blocking and washing conditions, and select suitable secondary
antibody systems.
.jpg)