This website uses cookies to ensure you get the best experience on our website.
- Table of Contents

Antibody types form the foundation of adaptive immune system. In humans, IgG alone accounts for approximately 75% of circulating antibodies, while the other four classes—IgM antibodies, IgA, IgE, and IgD—fill specialized roles in blood, tissues, and secretions. Together, these Y-shaped glycoproteins create a flexible defense network that identifies, neutralizes, and clears pathogens with remarkable precision.
All antibodies share a common heterotetrametric structure consisting of heavy and lights connected by disulfide bonds in the hinge regions but variations in their constant regions define each antibody isotype. Understanding these differences is critical for selecting the right reagents for immunodiagnostic tests, designing effective vaccines, and developing therapeutic monoclonal antibodies. Each type plays a specific role in the adaptive immune response, influencing how the body detects and neutralizes threats. Learn more about the types of antibody structure and their impact on immune system. For research applications, custom monoclonal antibody production and recombinant antibodies ensures that the right antibody type is used for the intended experimental and therapeutic applications.
Beyond isotype selection, the way an antibody is discovered and produced can also shape its final performance in research and therapeutic development. For readers exploring how antibodies move from immune repertoire screening to recombinant production, methods such as single B cell antibody discovery, phage display library screening, and recombinant antibody production in CHO cells provide useful next steps.
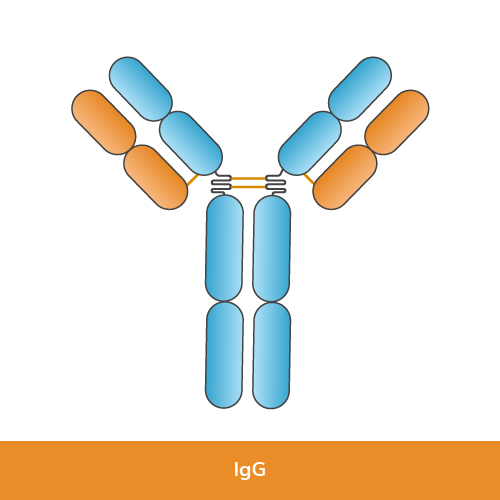
Human immunoglobulin IgG dominates serum and tissue fluids. Because of its high affinity and long half-life, it provides lasting protection after infection or vaccination. IgG also crosses the placenta, granting passive immunity to the fetus.
| Feature | Details |
|---|---|
| Structure | Two γ heavy chains, two light chains, single Y-shaped unit |
| Percentage of serum antibodies | ~75 % |
| Key functions | Neutralization, opsonization, complement activation, placental transfer |
| Common laboratory uses | Western blot, ELISA, immunohistochemistry |
Humans produce four IgG subclasses, each with distinct functions in immune defense:
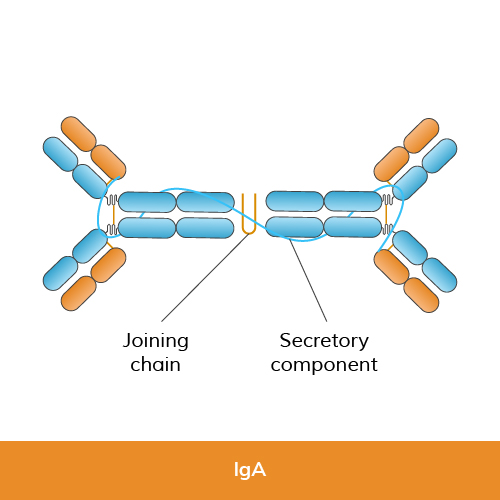
Function: IgA is primarily found in mucosal linings, such as the respiratory and gastrointestinal tracts, as well as in saliva, tears, and breast milk. It plays a key role in mucosal immunity by preventing pathogens from adhering to mucosal surfaces and facilitating their removal.
Structure: IgA has two subclasses, IgA1 and IgA2, each with distinct heavy chain structures. Both subclasses consist of two heavy chains (alpha chains) and two light chains (kappa or lambda chains).
Size: IgA can exist as monomers or dimers, where two IgA molecules are joined together by a Joining (J) chain and secretory component.
Research use: IgA is utilized in studies of mucosal immunity and secretory pathways, often in ELISA and immunofluorescence, to investigate immune responses in mucosal tissues and secretions.
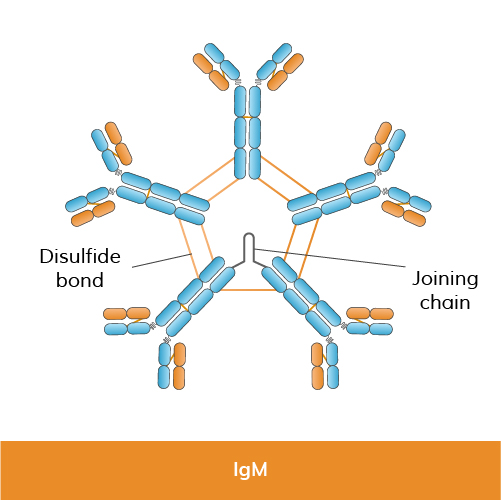
Function: IgM is the largest antibody in terms of size and is the first antibody produced in response to an infection (primary immune response). It exists mainly as a pentamer (five units joined together) in its secreted form, which enhances its ability to bind multiple antigens simultaneously (binding avidity). IgM is crucial for the initial immune response and is often associated with the clearance of pathogens during the early stages of infection.
Structure: IgM is composed of five units—four identical heavy chains (mu chains) and four identical light chains (kappa or lambda chains)—arranged in a pentameric structure.
Size: IgM molecules are large and pentameric, allowing them to bind multiple antigens simultaneously.
Research use: IgM is employed in agglutination assays and early immune response studies because of its pentameric structure, which provides strong binding to multivalent antigens.
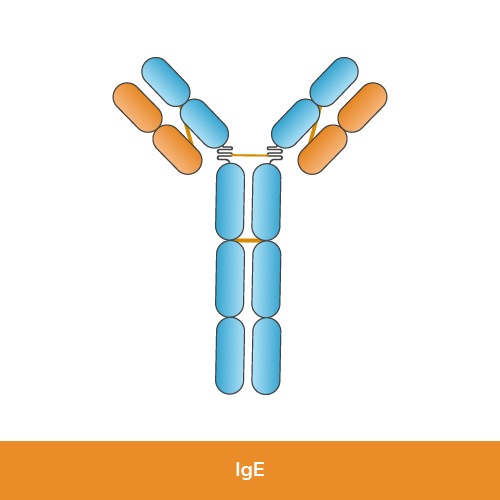
Function: IgE is present in small amounts in the bloodstream and is primarily involved in allergic reactions and defense against parasitic infections. It triggers the release of histamine and other chemicals from mast cells and basophils, leading to allergic symptoms like inflammation and bronchoconstriction.
Structure: IgE consists of two identical heavy chains (epsilon chains) and two identical light chains (kappa or lambda chains), similar to other immunoglobulin classes.
Size: IgE molecules are monomeric.
Research use: IgE is used in allergy research and studies of parasitic infections, often in ELISA, to measure and characterize immune responses related to allergic reactions and parasite defenses.
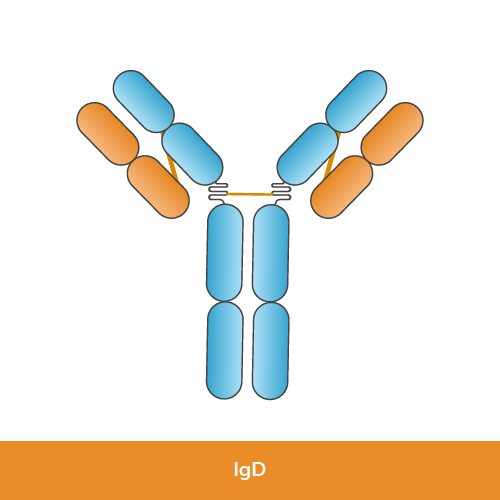
Function: IgD is found in small amounts in the bloodstream and is mainly present on the surface of immature B-lymphocytes and in mucosal areas. Its exact function is still under research, but it is believed to play a role in the activation of B cells.
Structure: IgD has two identical heavy chains (delta chains) and two identical light chains (kappa or lambda chains).
Size: IgD molecules are monomeric.
Research use: IgD is less commonly used in research but is studied in the context of B cell development and activation, primarily through flow cytometry and immunoprecipitation techniques.
Because B cell activation and repertoire biology directly influence how antigen-specific antibodies are identified, researchers interested in moving from B cell biology to antibody generation may also want to explore single B cell antibody discovery, which focuses on isolating native heavy/light chain pairs from individual B cells.
These immunoglobulin isotypes vary in their distribution, roles in immune responses, and structural compositions. Below, we’ve provided a comparison table summarizing the immunoglobulin isotypes and their subclasses, highlighting their structural characteristics, size, and primary functions.
| Isotype | Subclasses | Structure | Size | Function and Location |
|---|---|---|---|---|
| IgG | IgG1, IgG2, IgG3, IgG4 | Two gamma (γ) heavy chains + two light chains | Monomeric | Long-term immunity, crosses placenta, opsonization, complement activation |
| IgA | IgA1, IgA2 | Two alpha (α) heavy chains + two light chains | Monomeric/dimer | Mucosal immunity, found in mucosal linings, saliva, tears, breast milk |
| IgM | N/A | Five mu (μ) heavy chains + five light chains | Pentameric | First antibody in primary immune response, large size aids in antigen binding |
| IgE | N/A | Two epsilon (ε) heavy chains + two light chains | Monomeric | Allergic reactions, defense against parasites, binds to mast cells and basophils |
| IgD | N/A | Two delta (δ) heavy chains + two light chains | Monomeric | Found on surface of B cells, role in B cell activation, exact function still under study |
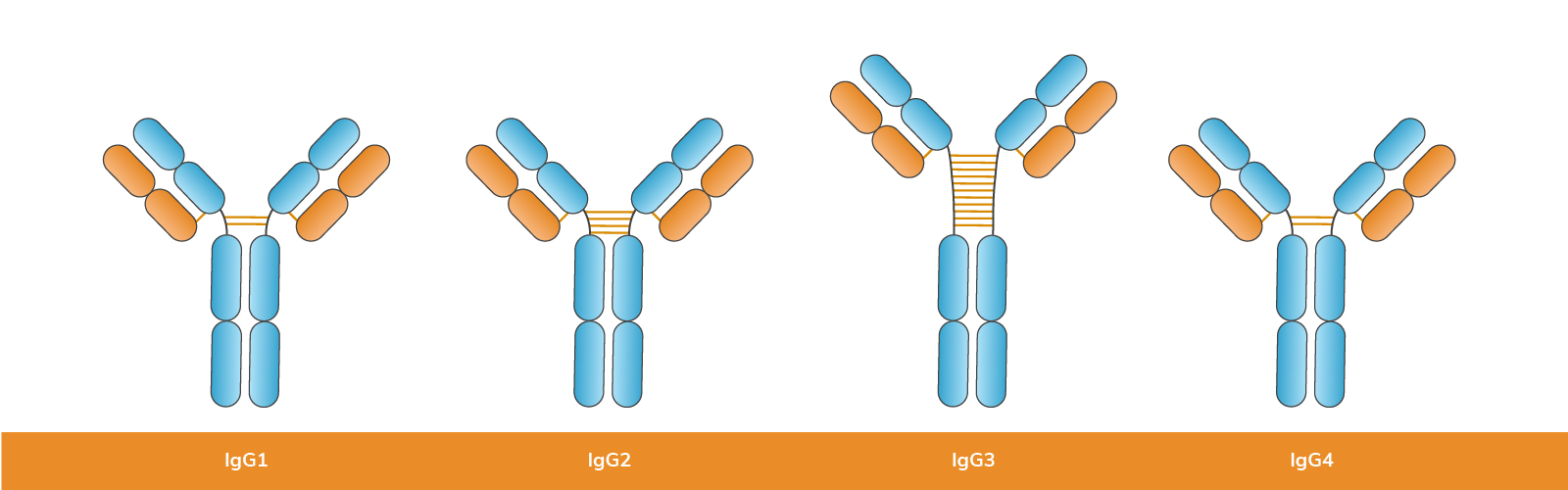
The subclasses within each immunoglobulin isotype differ primarily in their structural characteristics and functional roles within the immune system. Here’s a breakdown of the differences among the subclasses for IgG and IgA:
| Isotype | Structure | Biological Functions | Distribution and Function |
|---|---|---|---|
| IgG | IgG1, IgG3, IgG4: These subclasses have relatively similar structures with variations in their constant (C) regions. | IgG1: Predominant in secondary immune responses, effective in opsonization (marking pathogens for destruction by phagocytes) and complement activation. | IgG1 and IgG3: Found in higher concentrations in serum and tissues, involved in defense against bacterial and viral infections. |
| IgG2: Structurally distinct with longer hinge regions and differences in glycosylation patterns compared to IgG1 and IgG3. | IgG2: Less effective in complement activation but plays a crucial role in opsonization against encapsulated bacteria. | IgG2: More prevalent in mucosal areas and less abundant in serum. | |
| IgG3: Has a shorter half-life but is highly effective in activating complement and neutralizing toxins. | IgG4: Found in lower concentrations, mainly involved in chronic immune responses and allergic reactions. | ||
| IgG4: Often involved in immune tolerance and is less effective in complement activation and opsonization. | |||
| IgA | IgA1: Longer hinge region, more susceptible to proteolytic cleavage. | IgA1: Predominant in serum and mucosal secretions, involved in defense against pathogens by inhibiting their adherence to mucosal surfaces. | IgA1: More prevalent in serum and plays a crucial role in systemic immune responses. |
| IgA2: Shorter hinge region, more resistant to proteolysis. | IgA2: Found mainly in mucosal secretions, provides broader protection against pathogens due to enhanced resistance to proteolysis. | IgA2: Predominantly found in mucosal secretions (e.g., saliva, tears, breast milk) and provides localized immunity at mucosal surfaces. |
Immunoglobulins and their isotypes are a cornerstone of adaptive immunity, showcasing diversity in structure and function. Ongoing research into these antibodies not only enhances our understanding of immune responses but also drives innovations in diagnostic tools and therapeutic interventions.
As antibody science continues to evolve, newer discovery platforms are expanding how researchers obtain high-affinity and clinically relevant candidates. Approaches such as antibody phage display library screening and transgenic animals for antibody production have become important routes for generating antibodies beyond conventional immunization workflows.
As essential tools in biomedical research, scientists require antibodies, or immunoglobulins, to identify and quantify proteins, pathogens, and other biomolecules when performing ELISA, WB, IHC, and more applications. To enhance these applications, especially where labeled antibodies are required, researchers often rely on an antibody conjugation service for precision labeling. Each isotype, with its distinct structural and functional properties, is selected based on the specific needs of the experiment. For example, IgG antibodies are often chosen for their high affinity and specificity in detecting target proteins in tissue samples, while IgM, due to its pentameric structure, is preferred in agglutination assays. Researchers also exploit subclass differences, such as IgA's stability in mucosal environments for studying mucosal immunity or IgE for investigating allergic responses and parasite infections.
In cases where full-length antibodies are not required, researchers may also turn to fragment-based formats for improved flexibility in assay development and engineered binding applications. For example, microbial systems are often used to produce Fab, scFv, and VHH-style formats more efficiently, as outlined in this overview of microbial expression systems for antibody fragment production.
At Boster Bio, we specialize in antibody production services, offering high-quality primary antibodies validated for specificity, affinity, and several research applications, including WB, IHC, IF/ICC, ELISA, and flow cytometry. For custom-engineered antibodies based on your target isotype, explore our Recombinant Antibody Production Service. With over 20,000 antibodies in our catalog, you can find primary antibodies with high specificity and affinity for your research.
For readers comparing different antibody development routes, it may also be useful to look at how platform choice affects antibody format and downstream application, from recombinant CHO cell production for full-length recombinant antibodies to transgenic animal-based production strategies for generating human-compatible antibody candidates.