This website uses cookies to ensure you get the best experience on our website.
- Table of Contents
Facts about Argininosuccinate synthase.
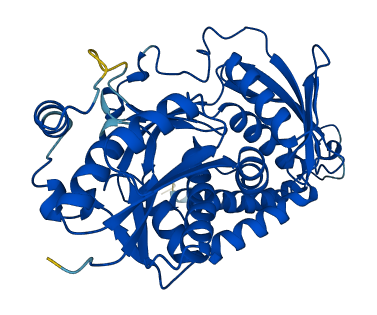
.
| Human | |
|---|---|
| Gene Name: | ASS1 |
| Uniprot: | P00966 |
| Entrez: | 445 |

| Belongs to: |
|---|
| argininosuccinate synthase family |

argininosuccinate synthase 1; argininosuccinate synthase; argininosuccinate synthetase 1; argininosuccinate synthetase; ASSCTLN1; citrulline-aspartate ligase; Citrulline--aspartate ligase; EC 6.3.4.5
Mass (kDA):
46.53 kDA

| Human | |
|---|---|
| Location: | 9q34.11 |
| Sequence: | 9; NC_000009.12 (130444707..130501274) |
Expressed in adult liver.
Cytoplasm, cytosol.





PMID: 6194510 by Bock H.-G.O., et al. Sequence for human argininosuccinate synthetase cDNA.
PMID: 6321498 by Freytag S.O., et al. Molecular structures of human argininosuccinate synthetase pseudogenes. Evolutionary and mechanistic implications.