This website uses cookies to ensure you get the best experience on our website.
- Table of Contents
The immune system is a complex network that protects the body from harmful pathogens such as bacteria, viruses, and other foreign substances. Central to this defense mechanism are various molecular components, including antibodies, antigens, epitopes, haptens, and immunogens. Each of these components plays a unique role in the immune response, and understanding their differences is crucial for comprehending how the immune system functions.
Antibodies, also known as immunoglobulins, are specialized Y-shaped proteins produced by B cells, a type of white blood cell. These proteins are critical in the immune response as they identify and neutralize foreign objects like bacteria and viruses. The structure of antibodies allows them to specifically recognize and bind to antigens, effectively marking them for destruction by other immune cells. In research applications, this specificity can be enhanced or visualized through chemical labeling—a process typically facilitated by an antibody conjugation service. This specificity is due to the unique binding sites on the antibody that match specific parts of the antigen, known as epitopes.
As antibody research moves from basic immune recognition to reagent and therapeutic development, researchers may generate binders through approaches such as single B cell antibody discovery or phage display library screening, depending on whether the goal is to preserve native heavy/light chain pairing or to run fully in vitro, high-throughput selection.
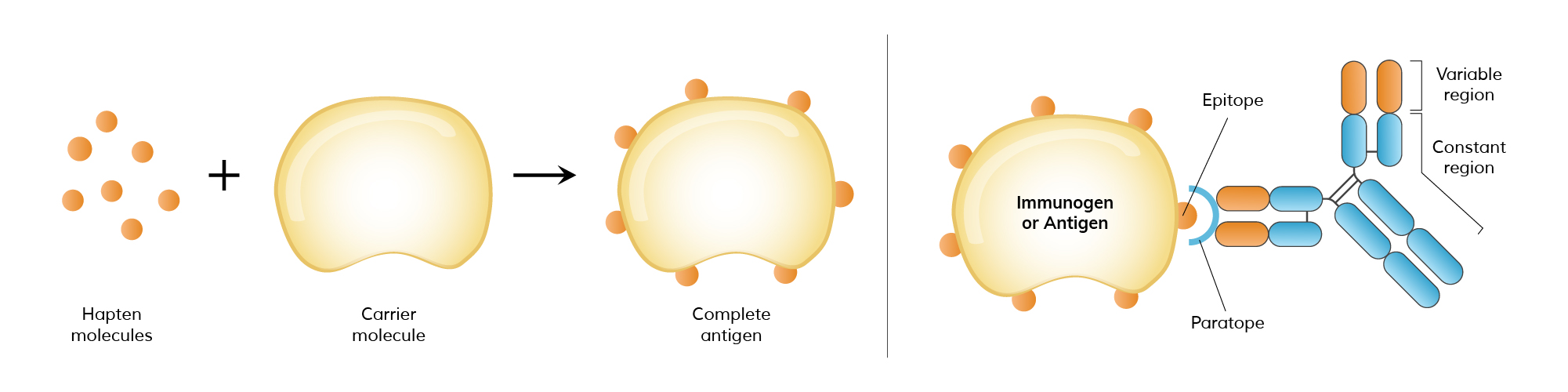
Antigens are molecules or molecular structures, often found on the surface of pathogens, that can elicit an immune response. They are typically proteins or polysaccharides and are recognized by the immune system as foreign. The interaction between antigens and antibodies is highly specific, akin to a lock and key, where the antibody binds to the antigen to neutralize or flag it for removal. This interaction is fundamental to the body’s ability to recognize and remember pathogens, forming the basis of immunity.
Epitopes, also known as antigenic determinants, are specific regions on an antigen that are recognized by antibodies. Each antigen can have multiple epitopes, allowing for the binding of different antibodies. This multiplicity enables a robust and versatile immune response, as various antibodies can target different parts of a single pathogen. The specificity of epitopes is crucial in vaccine development, as vaccines aim to introduce harmless forms of epitopes to stimulate an immune response without causing disease.
Haptens are small molecules that, by themselves, are not immunogenic, meaning they cannot independently provoke an immune response. However, when haptens bind to larger carrier proteins, they form a complex that the immune system recognizes as foreign, triggering an immune response. This phenomenon is particularly important in pharmacology and allergen studies, where certain drugs or small molecules can act as haptens, causing allergic reactions when they bind to proteins in the body.
Immunogens are a subset of antigens that can trigger an immune response on their own. While all immunogens are antigens, not all antigens qualify as immunogens. For instance, a hapten is an antigen but not an immunogen unless it is attached to a carrier protein. Immunogens must have certain properties, such as adequate size and molecular complexity, to effectively stimulate the immune system. This distinction is important in vaccine development and allergy research, where understanding what makes an antigen immunogenic can inform treatment strategies.
Depending on the downstream application, antibodies raised against these immune targets may later be reformatted for different production systems. For example, smaller binding formats such as Fab, scFv, or VHH are often well suited to microbial expression systems for antibody fragment production, especially when Fc-mediated functions or mammalian glycosylation are not required.
We’ve provided a table below that summarizes and highlights the distinct roles and interactions of these immune system components.
| Term | Description | Example |
|---|---|---|
| Antibody | Y-shaped proteins produced by B cells that specifically recognize and bind to antigens, aiding in the neutralization and destruction of pathogens. | IgG, IgM, IgA |
| Antigen | Substances, often proteins or polysaccharides on the surface of pathogens, that trigger the production of antibodies and stimulate an immune response. | Viral proteins, bacterial cell wall components |
| Epitope | Specific regions on antigens that are recognized and bound by antibodies. | Binding site on a viral protein |
| Hapten | Small molecules that are not immunogenic on their own but can elicit an immune response when bound to a larger carrier protein. | Penicillin, certain drugs |
| Immunogen | A type of antigen capable of independently provoking an immune response, possessing the necessary properties to stimulate immune cells. | Whole viruses, bacterial toxins |
The immune system's effectiveness hinges on interactions among antibodies, antigens, haptens, epitopes, and immunogens. Antibodies recognize and bind to antigens, identifying foreign molecules for destruction. Haptens, though non-immunogenic alone, provoke responses when linked to larger proteins. Epitopes are specific antigen regions targeted by antibodies, while immunogens independently trigger immune responses. Comprehending these dynamics not only advances immunological understanding but also drives the development of vaccines, therapies, and treatments for diseases.
As these concepts move into translational research, the choice of antibody generation and production platform becomes increasingly important. Early discovery may rely on phage display libraries and screening for controlled in vitro selection or single B cell antibody discovery when native B-cell-derived sequences are preferred, while larger-scale full-length antibody expression is commonly associated with recombinant antibody production in CHO cells or transgenic animals for antibody production when human-compatible properties and therapeutic development are priorities.
At Boster Bio, we offer a wide range of high-quality primary antibodies for various research applications, including custom polyclonal antibody production services for labs requiring antigen-specific sera. Our antibodies have high specificity and affinity, and have been rigorously validated for common applications such as WB, IHC, ICC/IF, ELISA, and Flow. Whether you are exploring cellular pathways, studying disease mechanisms, or developing novel therapies, browse our catalog to find the primary antibodies you need to advance your research.
For teams comparing antibody platforms more closely, CHO cell production is often favored for full-length recombinant antibodies with appropriate folding and glycosylation, whereas transgenic animal systems are especially valuable when fully human or humanized antibody repertoires are needed for advanced discovery and therapeutic programs.