This website uses cookies to ensure you get the best experience on our website.
- Table of Contents
and ELISA kits, proteins related to B-cells.
B-cells are integral components of the adaptive immune system, essential for defending the body against infections and diseases. Originating and maturing in the bone marrow, B-cells are responsible for producing antibodies—specialized proteins that recognize and bind to specific antigens found on the surfaces of pathogens like bacteria and viruses. Upon encountering an antigen, B-cells differentiate into plasma cells, which secrete large quantities of antibodies to neutralize and eliminate the threat. Additionally, B-cells contribute to immunological memory, enabling the immune system to respond more swiftly and effectively upon future exposures to the same pathogen. Advances in B-cell research are pivotal for developing targeted therapies, improving vaccine efficacy, and understanding autoimmune disorders, making them a focal point in the quest for innovative medical treatments.
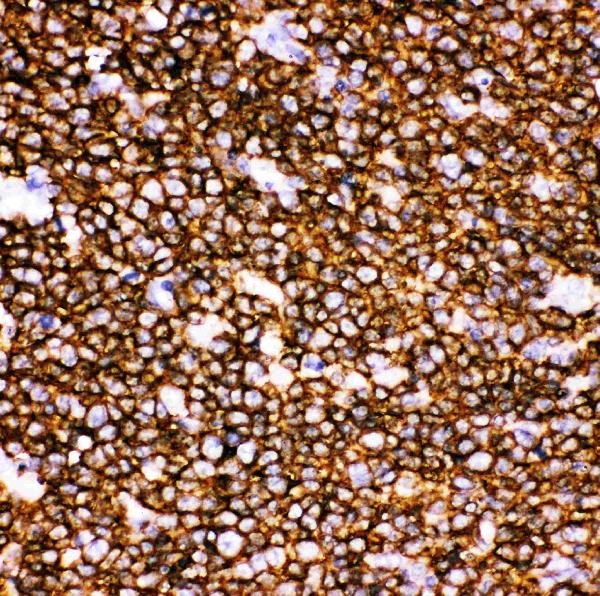
Anti-CD20/MS4A1 Antibody Picoband®, Figure 2. IHC analysis of CD20 using anti-CD20 antibody (PB9050).
CD20 was detected in paraffin-embedded section of Human Tonsil Tissue....
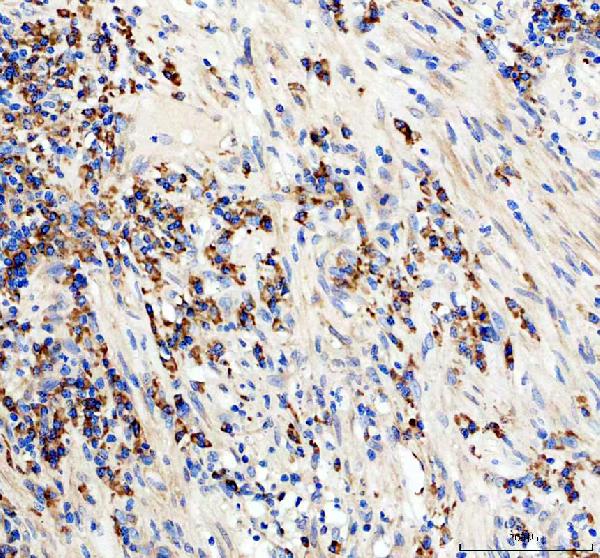
Anti-CD19 Antibody Picoband®,Figure 2. IHC analysis of CD19 using anti-CD19 antibody (PB9800). CD19 was detected in a paraffin-embedded section of human appendix tissue......
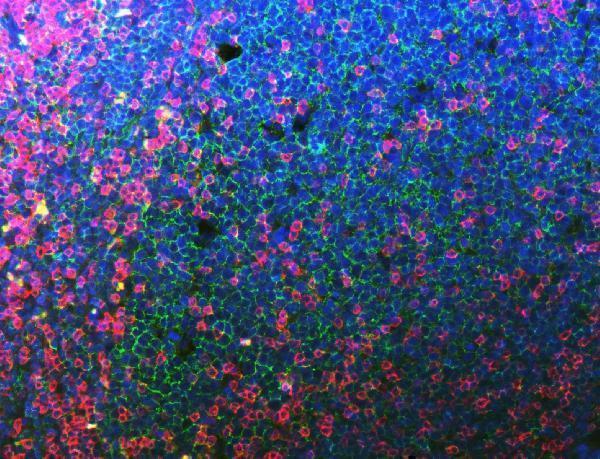
Anti-CD20 Antibody Picoband® (monoclonal, 4D11), Figure 4. IF analysis of CD3E and CD20 using anti-CD3E antibody (PB9093) and anti-CD20 antibody (M03780-5).
CD3E and CD20 ...
| Protein Name | Gene Name | Function |
|---|---|---|
| CD19 | CD19 | Essential for B-cell development and signaling |
| CD20 | MS4A1 | Target for B-cell depletion therapies |
| CD22 | CD22 | Involved in B-cell receptor signaling |
| CD79A | CD79A | Part of B-cell receptor complex |
| CD79B | CD79B | Part of B-cell receptor complex |
| CD138 | SDC1 | Marker for plasma cells |
| CD23 | FCER2 | Low-affinity IgE receptor |
| CD40 | CD40 | Important for B-cell activation and isotype switching |
| CD27 | CD27 | Marker for memory B cells |
| CD38 | CD38 | Involved in cell adhesion and signal transduction |
| BLNK | BLNK | Adapter protein in B-cell receptor signaling |
| BTK | BTK | Involved in B-cell development signaling pathway |
| PAX5 | PAX5 | Transcription factor critical for B-cell identity |
| BCL6 | BCL6 | Transcriptional repressor involved in germinal center formation |
| IL4R | IL4R | Receptor for interleukin-4, important in B-cell differentiation |
| CXCR5 | CXCR5 | Chemokine receptor guiding B-cell migration |
| CD5 | CD5 | Typically expressed on B1 cells |
| IgD | IGHD | Immunoglobulin D, surface B-cell receptor |
| IgM | IGHM | Immunoglobulin M, surface B-cell receptor |
| BAFF-R | TNFRSF13C | Receptor for BAFF, important for B-cell survival |
Class Switch Recombination (CSR) is a crucial mechanism in B-cell biology that enables a single B-cell to produce different classes of antibodies (immunoglobulins) without altering the antigen specificity. During CSR, the constant region of the immunoglobulin heavy chain undergoes genetic recombination, allowing the B-cell to switch from producing IgM antibodies to other classes such as IgG, IgA, or IgE. This process is essential for tailoring the immune response to different pathogens and bodily locations. For instance, IgG antibodies are effective in neutralizing toxins and viruses, IgA plays a vital role in mucosal immunity, and IgE is involved in allergic responses. CSR is regulated by cytokines and requires the activation-induced cytidine deaminase (AID) enzyme, which introduces mutations in the DNA to facilitate recombination. Understanding CSR is fundamental for developing targeted therapies for immunological disorders and improving vaccine efficacy by promoting the desired antibody class response.
Somatic Hypermutation (SHM) is a key process in the adaptive immune response, occurring in B-cells within germinal centers of lymphoid tissues. SHM introduces point mutations at a high rate into the variable regions of the immunoglobulin genes, specifically in the regions that determine antigen binding. This deliberate mutation process generates a diverse pool of B-cell receptors (BCRs) with varying affinities for a given antigen. B-cells with BCRs that have higher affinity for the antigen are preferentially selected for survival and proliferation, a process known as affinity maturation. SHM enables the immune system to fine-tune antibody specificity and effectiveness, enhancing the ability to combat pathogens more efficiently over time. The enzyme activation-induced cytidine deaminase (AID) plays a pivotal role in initiating SHM by deaminating cytosine bases in DNA, leading to mutations. Dysregulation of SHM can contribute to autoimmune diseases and lymphomas, making it a significant focus of research for understanding and treating immune-related disorders.