This website uses cookies to ensure you get the best experience on our website.
- Table of Contents
and ELISA kits, proteins related to asthma.
Asthma is a chronic respiratory condition affecting millions worldwide, characterized by airway inflammation and narrowing that leads to breathing difficulties. This condition not only impairs daily activities but also significantly impacts the quality of life. Recent advancements in asthma research have spotlighted the pivotal role of antibodies in understanding and managing the disease. By targeting specific antibodies, scientists aim to unravel the complex immune responses that trigger asthma attacks, paving the way for more effective and personalized treatments. Our dedicated research focuses on developing antibody-based therapies that can mitigate inflammation, reduce symptoms, and improve long-term outcomes for those living with asthma. Join us in our mission to transform asthma care through innovative antibody research, offering hope and enhanced well-being for individuals affected by this pervasive condition.
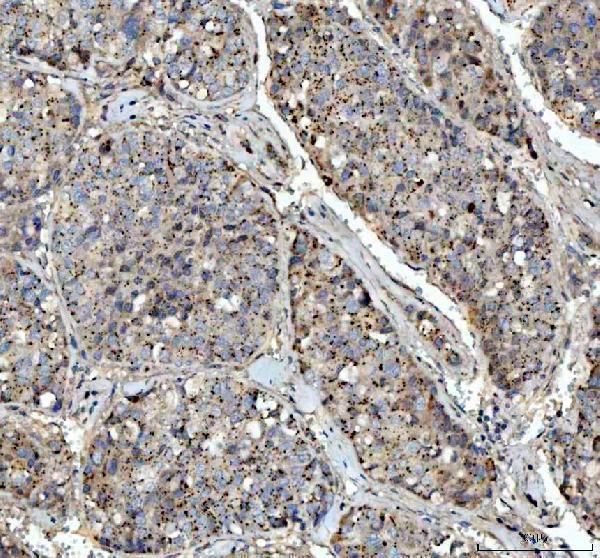
Anti-VEGF/VEGFA Antibody Picoband®, Figure 3. IHC analysis of VEGFA using anti-VEGFA antibody (PA1080).
VEGFA was detected in a paraffin-embedded section of human liver cancer tissue. Hea...
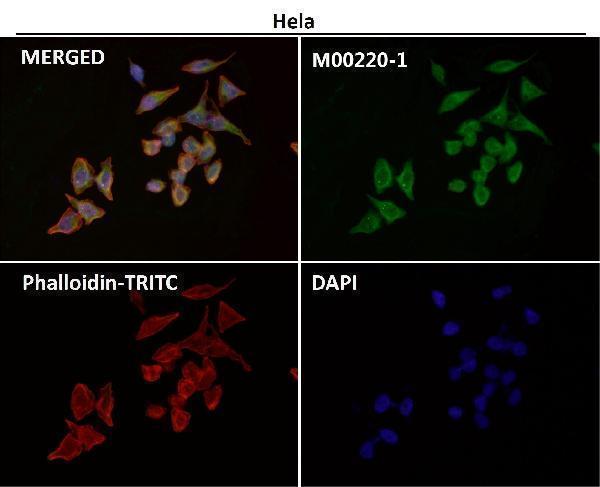
Anti-CD86/B7 2 Rabbit Monoclonal Antibody, Immunofluorescent analysis using the Antibody at 1:50 dilution....

Figure 1. IHC analysis of TGF Beta 1/TGFB1 using Anti-TGF beta 1/TGFB1 Antibody (A00019-2).
TGF Beta 1/TGFB1 was detected in a paraffin-embedded section of rat li...
| Protein Name | Gene Name | Function |
|---|---|---|
| Interleukin-4 | IL4 | Cytokine involved in the differentiation of naive T helper cells to Th2 cells, promoting IgE production. |
| Interleukin-5 | IL5 | Cytokine that promotes the growth and differentiation of eosinophils. |
| Interleukin-13 | IL13 | Cytokine involved in the inflammatory response and IgE production. |
| Periostin | POSTN | Involved in Th2-mediated inflammation and tissue remodeling. |
| Eosinophil Cationic Protein | RNASE3 | Released by eosinophils, contributing to inflammation and tissue damage. |
| Immunoglobulin E | IGHE | Antibodies involved in allergic responses and asthma pathogenesis. |
| C-Reactive Protein | CRP | Acute-phase protein serving as a marker of systemic inflammation. |
| MUC5AC | MUC5AC | Mucin protein contributing to mucus viscosity and airway obstruction. |
| Surfactant Protein D | SFTPD | Involved in innate immunity and regulation of inflammatory responses in the lungs. |
| Vascular Endothelial Growth Factor | VEGFA | Promotes angiogenesis and increases vascular permeability, contributing to inflammation. |
| Monocyte Chemoattractant Protein-1 | CCL2 | Chemokine that recruits monocytes to sites of inflammation. |
| Interleukin-17 | IL17A | Pro-inflammatory cytokine involved in neutrophil recruitment and airway inflammation. |
| OX40 Ligand | TNFSF4 | Co-stimulatory molecule involved in T-cell activation and survival. |
| CD86 | CD86 | Costimulatory molecule essential for T-cell activation and immune response modulation. |
| Interleukin-25 | IL25 | Cytokine that promotes Th2 responses and eosinophilic inflammation. |
| Interleukin-33 | IL33 | Alarmin cytokine that activates Th2 cells and innate lymphoid cells, driving inflammation. |
| ST2 | IL1RL1 | Receptor for IL-33, mediating its pro-inflammatory effects in asthma. |
| Transforming Growth Factor Beta | TGFB1 | Regulates immune responses and tissue remodeling in the airways. |
| Major Basic Protein | PRG2 | Eosinophil-derived protein contributing to airway epithelial damage and hyperreactivity. |
| Eotaxin-3 | CCL26 | Chemokine that attracts eosinophils to the airways, promoting inflammation. |
Asthma is fundamentally characterized by chronic airway inflammation, orchestrated by a complex interplay of immune cells and cytokines. Central to this inflammatory response are T-helper type 2 (Th2) cells, which produce key cytokines such as IL-4, IL-5, and IL-13. These cytokines facilitate the recruitment and activation of eosinophils, mast cells, and the production of IgE antibodies, all of which contribute to airway hyperresponsiveness and obstruction. Additionally, recent research has highlighted the role of Th17 cells and innate lymphoid cells in non-Th2 asthma phenotypes, underscoring the disease's heterogeneity. Understanding these inflammatory pathways is crucial for developing targeted therapies, such as monoclonal antibodies that inhibit specific cytokines, thereby offering more personalized and effective treatment options. Ongoing studies continue to elucidate the intricate signaling mechanisms that drive asthma pathogenesis, providing insights into novel therapeutic targets and improving disease management strategies.
Airway remodeling refers to the structural changes that occur in the bronchial walls of individuals with chronic asthma, significantly contributing to the persistence and severity of the disease. These changes include subepithelial fibrosis, increased smooth muscle mass, angiogenesis, and glandular hypertrophy, all of which lead to reduced airway elasticity and increased airway resistance. Chronic inflammation plays a pivotal role in airway remodeling by stimulating fibroblast proliferation and extracellular matrix deposition. Growth factors such as TGF-β are key mediators in this process, promoting the differentiation and activation of structural cells within the airways. Airway remodeling not only exacerbates airflow obstruction but also diminishes responsiveness to conventional asthma therapies, making it a critical focus of current research. Investigating the molecular and cellular mechanisms underlying airway remodeling offers the potential to develop interventions that can halt or reverse these structural changes, ultimately improving long-term outcomes for patients with asthma.