This website uses cookies to ensure you get the best experience on our website.
- Table of Contents
and ELISA kits, proteins related to Epilepsy.
Epilepsy is a chronic neurological disorder characterized by recurrent, unprovoked seizures resulting from abnormal electrical activity in the brain. Affecting millions worldwide, epilepsy manifests in various forms, ranging from brief moments of confusion to intense convulsions. While many individuals with epilepsy lead active and fulfilling lives, the condition can pose significant challenges, including impacts on learning, memory, and daily functioning. Recent advancements in research have spotlighted the role of antibodies in the development and progression of epilepsy. By exploring how the immune system interacts with neural pathways, scientists aim to uncover novel therapeutic targets that can offer more effective and personalized treatments. This innovative approach holds promise for improving the quality of life for those affected by epilepsy, paving the way for breakthroughs in managing and potentially overcoming this complex neurological condition.

Anti-BDNF Rabbit Monoclonal Antibody, Immunofluorescent analysis of HeLa cells, using BDNF Antibody....

Anti-Glucose Transporter GLUT1/SLC2A1 Antibody Picoband®, Figure 14. IF analysis of SLC2A1 using anti-SLC2A1 antibody (PB9435).
SLC2A1 was detected in immunocytochemical ...
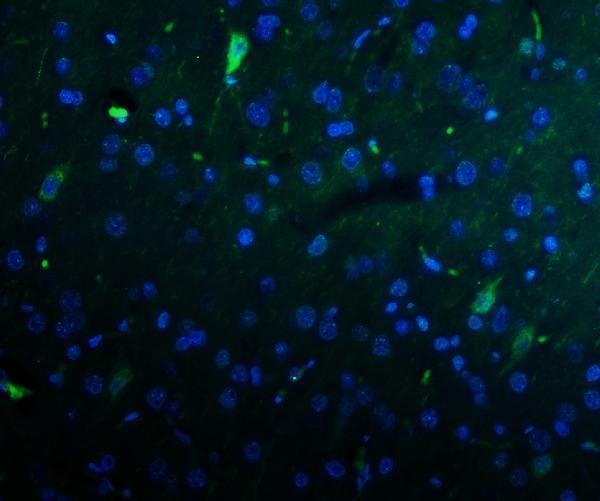
Anti-Kv1.1 potassium channel/KCNA1 Antibody Picoband®, Figure 5. IF analysis of KCNA1 using anti-KCNA1 antibody (A01813-1).
KCNA1 was detected in paraffin-embedded secti...
| Protein Name | Gene Name | Function |
|---|---|---|
| Nav1.1 | SCN1A | Encodes sodium channel involved in action potential initiation |
| Nav1.2 | SCN2A | Encodes sodium channel involved in generation and conduction of action potentials |
| GABA-A alpha1 | GABRA1 | Encodes subunit of GABA receptor involved in inhibitory neurotransmission |
| Kv1.1 | KCNA1 | Encodes potassium channel important for regulating neuronal excitability |
| P/Q-type Ca channel | CACNA1A | Encodes calcium channel involved in neurotransmitter release |
| DEPDC5 | DEPDC5 | Part of mTOR pathway implicated in focal cortical dysplasia and epilepsy |
| MTHFR | MTHFR | Involved in homocysteine metabolism; variants associated with seizure susceptibility |
| CDKL5 | CDKL5 | Involved in neuronal development; mutations associated with epileptic encephalopathy |
| Kv7.2 | KCNQ2 | Encodes potassium channel involved in setting neuronal resting potential |
| nAChR alpha4 | CHRNA4 | Encodes subunit of nicotinic acetylcholine receptor linked to epilepsy |
| BDNF | BDNF | Brain-derived neurotrophic factor involved in neuron survival and synaptic plasticity |
| Syntaxin-binding protein 1 | STXBP1 | Involved in synaptic vesicle release; mutations linked to epileptic encephalopathy |
| Tuberin | TSC1 | Part of tuberous sclerosis complex involved in cell growth; mutations associated with epilepsy |
| Hamartin | TSC2 | Part of tuberous sclerosis complex involved in cell growth; mutations associated with epilepsy |
| NMDA receptor NR2A | GRIN2A | Encodes NMDA receptor subunit involved in synaptic plasticity |
| Protocadherin 19 | PCDH19 | Protein mutated in epilepsy in females |
| GLUT1 | SLC2A1 | Glucose transporter; deficiency leads to seizures |
| LGI1 | LGI1 | Involved in synaptic function; mutations linked to temporal lobe epilepsy |
| UBE3A | UBE3A | Encodes ubiquitin ligase involved in synaptic function |
| PRRT2 | PRRT2 | Associated with paroxysmal kinesigenic dyskinesia and epilepsy |
Genetic research in epilepsy explores the hereditary aspects and molecular mechanisms that contribute to seizure disorders. Advances in genomics have enabled the identification of specific genes and mutations associated with various forms of epilepsy, including both common and rare epileptic syndromes. Understanding the genetic underpinnings helps in diagnosing specific epilepsy types, predicting disease prognosis, and developing targeted therapies. Additionally, studying genetic factors aids in uncovering the biological pathways involved in neuronal excitability and network stability, which are critical in the onset of seizures. This subarea also examines the role of gene-environment interactions and epigenetic modifications, providing a comprehensive view of how genetic predispositions manifest in epilepsy. The insights gained from genetic research not only enhance our fundamental understanding of epilepsy but also pave the way for personalized medicine approaches, improving treatment efficacy and patient outcomes.
Neuroinflammation has emerged as a pivotal mechanism in the pathophysiology of epilepsy. This subarea investigates how inflammatory processes within the central nervous system contribute to the development and progression of seizures. Chronic inflammation can alter neuronal excitability, disrupt synaptic transmission, and lead to neuronal damage, all of which can exacerbate epileptic activity. Research focuses on identifying inflammatory cytokines, immune cells, and signaling pathways that are involved in epileptogenesis. Understanding the link between inflammation and epilepsy opens new avenues for therapeutic interventions, such as anti-inflammatory treatments, which could potentially reduce seizure frequency and severity. Additionally, this area explores the role of blood-brain barrier dysfunction and how systemic inflammation may influence neuronal health and seizure susceptibility. By elucidating the complex interactions between the immune system and neural circuits, studies on neuroinflammation provide critical insights into novel strategies for managing and treating epilepsy.