This website uses cookies to ensure you get the best experience on our website.
- Table of Contents

Tween‑20 (polysorbate 20) was first developed in the 1940s as part of the “Span and Tween” series of surfactants by Imperial Chemical Industries. Originally invented for industrial use as emulsifiers (notably in food and cosmetics), these non-ionic detergents quickly became staple ingredients in various applications. Tween is actually a trademark registered by ICI Americas, Inc., and the number “20” refers to the total ethylene oxide units in the molecule. By the mid-20th century, Tween‑20 began appearing in scientific protocols – for example, early immunoassay techniques in the 1970s incorporated Tween‑20 in wash buffers to reduce background noise. As enzyme-linked immunosorbent assays (ELISAs) were pioneered in 1971, researchers adopted 0.05% Tween‑20 in the wash steps to improve specificity. One of the earliest studies to apply Tween-20 in immunoassays was published in 1985 by Spinola and Cannon. In their Western blot experiments, they demonstrated that Tween-20 effectively blocked non-specific sites on membranes, performing as well as or better than traditional blocking agents. This finding paved the way for the widespread use of Tween-20 in modern immunodetection techniques. It has since transitioned from being purely an industrial emulsifier to an indispensable reagent in life science research. Today, Tween‑20 remains ubiquitous in labs worldwide and is even formulated into many commercial kits and automated staining systems, underscoring its enduring role in modern science.
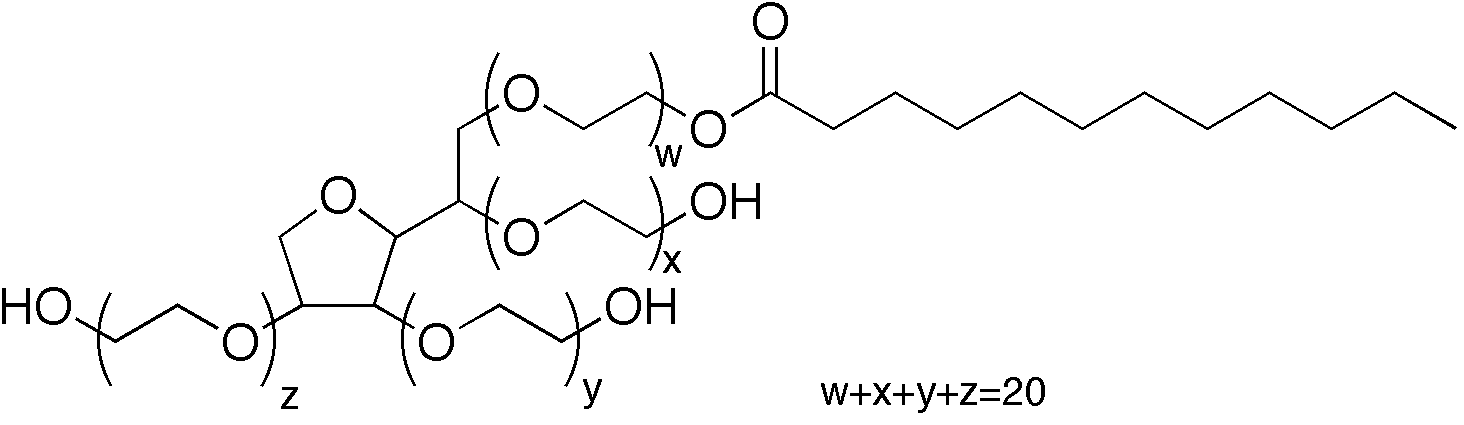
Figure: Structural representation of Polysorbate 20 (Tween‑20), showing the sorbitan (sorbitol-derived) backbone with polyoxyethylene chains (totaling ~20 ethylene oxide units across four arms) and a lauric acid fatty acyl ester tail. (image from Wikipedia)
Chemically, polysorbate 20 is defined as polyoxyethylene (20) sorbitan monolaurate. In simpler terms, it is derived from a sorbitan ring (which comes from sorbitol, a sugar alcohol) that has been esterified with a fatty acid (lauric acid, a 12-carbon saturated acid), and then ethoxylated with an average of 20 ethylene oxide units (polyethylene glycol chains). This gives Tween‑20 a molecular formula of approximately C58H114O26 and an average molar mass around 1,225–1,280 Da. Importantly, Tween‑20 is not a single uniform molecule but a mixture of species: the ethoxylation process yields a distribution of chain lengths (totaling about 20 EO units on average across 4 chains). Likewise, while laurate is the primary fatty acid, commercial Tween‑20 often contains small fractions of other fatty acids (myristate, palmitate, stearate) as impurities or byproducts (lauric acid typically ≥40% of the mixture).
Tween 20 is a non-ionic surfactant, which means it has hydrophilic polyethylene glycol groups and a hydrophobic fatty tail, yet carries no net charge. It is supplied as a clear, viscous, pale yellow liquid at room temperature. Key properties include a hydrophile–lipophile balance (HLB) of ~16.7 (highly hydrophilic) and a critical micelle concentration (CMC) on the order of 0.05–0.07 mM (around 60 mg/L at 20–25 °C). In practical terms, this means Tween 20 is readily soluble in water (forming micelles at very low concentrations) and in polar organic solvents like lower alcohols and dioxane, but is insoluble in hydrocarbons and natural oils. It is relatively stable and inert in neutral aqueous conditions, being mild and non-denaturing to proteins. However, it can be an irritant in pure form and will decompose if exposed to extreme conditions (see “Limitations” below). Its non-ionic nature ensures that it does not interfere with the ionic strength or pH of buffers, making it compatible with most biochemical reagents.
Tween 20 is most often encountered in the laboratory as a component of buffer solutions at low percentages. It is typically sold as a stock solution (for example, 10% or 20% v/v) and then diluted into working solutions. Table 1 below summarizes some common formulations containing Tween 20 and their typical concentrations:
| Buffer / Solution | Composition and Purpose | Typical Tween 20 Concentration |
|---|---|---|
| PBS-T (PBST) – Phosphate-buffered saline + Tween 20 | Wash buffer for ELISA, IF, etc.; prevents non-specific binding on plastic or membranes. | 0.05% Tween 20 (v/v) is common (range 0.05–0.1%). |
| TBS-T (TBST) – Tris-buffered saline + Tween 20 | Wash buffer for Western blots (nitrocellulose or PVDF membranes). Also used in IHC (especially when using alkaline phosphatase detection). | 0.05–0.1% Tween 20 (typical recipe: 0.1%). |
| Blocking Buffer (e.g., BSA or milk solution) | Blocking reagent to coat remaining binding sites on membranes or plates; Tween 20 is often included to further reduce background. | ~0.05% Tween 20 in blocking solution is standard. |
| Antibody Diluent (IHC/IF) | Dilution buffer for antibodies in immunohistochemistry or immunofluorescence; contains protein (BSA or serum) plus Tween 20 to stabilize antibodies and wet tissues evenly. | ~0.1% Tween 20 (typical range 0.01–0.2% for gentle wetting). |
| Cell Lysis/Permeabilization Buffer | Used to permeabilize or lyse cells for intracellular staining or protein extraction; often a mixed detergent buffer. Tween 20 provides mild lysis without denaturing proteins. | 0.05–0.5% Tween 20 (frequently ~0.1% with other detergents). |
| Membrane Protein Extraction Buffer | Buffer for extracting peripheral membrane proteins by washing membranes. Tween 20 can strip off loosely attached proteins without solubilizing the bilayer completely. | ~2% Tween 20 (a relatively high concentration for pre-extraction of membranes). |
Table 1: Typical buffer formulations containing Tween‑20 and
their concentrations. (PBS = phosphate-buffered saline; TBS = tris-buffered
saline; BSA = bovine serum albumin; IF = immunofluorescence; IHC =
immunohistochemistry.)
In practice, concentrations of Tween‑20
in working solutions are kept quite low (often 0.05–0.1% v/v, equivalent
to 0.5–1 mL per liter) because even tiny amounts are effective at preventing
non-specific adsorption of proteins. For instance, a classic ELISA wash buffer
might be PBS + 0.05% Tween‑20, and Western blot wash buffers are typically TBS
+ 0.05% or 0.1% Tween‑20 (TBST). In immunostaining protocols (IHC/IF), 0.1% is
a common choice for both washes and antibody diluents. Higher concentrations
(0.5–2%) are used only in special cases such as cell lysis or membrane protein
extraction where a stronger detergent effect is needed. Notably, stock
solutions of Tween‑20 can be quite viscous, so labs often prepare a 10%
solution for easier pipetting; many vendors sell 10% Tween‑20 ready-to-use for
buffer preparation.
In ELISA procedures, Tween‑20 plays a
critical role in wash buffers and sometimes in blocking solutions. After
coating an ELISA plate with antigen or antibody and performing binding steps,
the plate must be washed thoroughly to remove unbound enzyme conjugates and
reduce background. Tween‑20 (0.05–0.1%) in PBS or TBS is routinely added
to these wash buffers to prevent nonspecific protein–surface interactions.
The detergent helps to “wet” the plastic wells uniformly and dislodge
loosely bound proteins without disrupting specific antibody-antigen binding.
This yields a cleaner signal and higher signal-to-noise ratio, particularly when assays are developed using well-defined antigens generated through custom peptide synthesis. Indeed, it is
standard practice that “a detergent such as 0.05% Tween-20” be included in
ELISA wash steps to help remove nonspecifically bound material.
Tween‑20 may also be present in the blocking buffer or antibody diluent in ELISAs. For example, a blocking buffer of 1–5% BSA or milk in PBS might include ~0.05% Tween‑20 to further reduce background binding on the polystyrene well surface. Including Tween‑20 along with the blocking protein minimizes hydrophobic interactions that could cause proteins to stick nonspecifically. The outcome is decreased background staining and more reliable, low-noise readings. Overall, the addition of Tween‑20 is one reason ELISA can achieve high specificity and consistency – it ensures that only true antigen-antibody interactions (not “sticky” artifacts) generate the readout signal.
Historically, as soon as ELISA was introduced in the early 1970s (Engvall & Perlmann), the use of Tween‑20 was quickly adopted. Early reports (circa 1970s–80s) showed that washing plates with tap water plus Tween‑20 dramatically improved assay reliability. Today, virtually all ELISA protocols – whether home-brew or commercial kit – utilize Tween‑20 in the wash buffer. Its inclusion is so common that the term “PBST” or “TBST” in a protocol implicitly means PBS or TBS containing Tween‑20. Without Tween‑20, ELISAs often suffer from high background and variability due to proteins sticking to well walls; with Tween‑20 (and proper blocking), the background is kept low and uniform.
Western
blotting involves transferring proteins to a membrane (typically nitrocellulose
or PVDF) and probing with antibodies. Tween 20 is indispensable in both the blocking and washing steps. After
protein transfer, the membrane is blocked—commonly with 5% non-fat milk or BSA
in TBS containing 0.05% Tween 20—to saturate unoccupied binding sites. The
detergent helps the blocking protein distribute evenly, prevents drying, and
minimizes nonspecific binding “hot spots.”
During antibody
incubations, TBST (TBS + 0.05–0.1% Tween 20) is used between steps to wash away
loosely bound proteins and reduce antibody sticking to the membrane. This helps retain specific antigen–antibody
interactions while removing background noise. As noted in standard references,
polysorbate 20 is “a common detergent used in many buffers for washing
nitrocellulose membranes.”
If the same membrane needs to be reprobed after initial detection, this Western blot stripping buffer protocol gives a practical walkthrough for removing bound antibodies while preserving the transferred protein signal as much as possible.
Including Tween
20 results in a much cleaner blot: specific bands are sharp and distinct, while
nonspecific smears or speckles are reduced. Blotting protocols typically
include multiple 5-minute TBST washes after both primary and secondary antibody
incubations. Antibody dilution buffers often contain Tween 20 as well, to
prevent aggregation and maintain antibody stability.
Tween 20 must
be used at optimal concentrations—typically
~0.05%—to effectively reduce
nonspecific binding without disrupting true antigen–antibody complexes.
Research, especially in autoimmunity, has shown that Tween 20 improves
detection specificity in immunoblots. It is also compatible with common
detection enzymes such as HRP and alkaline phosphatase, as it doesn’t interfere
with their activity at working concentrations.
The
incorporation of Tween 20 into Western blot protocols since the 1980s
significantly enhanced signal clarity and reproducibility—making it a lasting
standard in immunodetection workflows.
Immunohistochemistry on tissue sections
relies on antigen–antibody binding within fixed cells/tissues mounted on
slides. Tween‑20 is widely used in IHC workflows to promote even staining and
reduce background. Typically, IHC protocols employ PBS or TBS with ~0.1%
Tween‑20 as a rinse buffer between antibody incubations. A small amount of
Tween‑20 (0.01–0.1%) in antibody diluents and rinse buffers assists in
uniform wetting of the sample and thorough washing. This is crucial because
tissue sections can repel water in spots or dry out; Tween‑20 ensures the
reagents spread evenly over the tissue and penetrate all crevices.
Including Tween‑20 in IHC wash buffers also prevents nonspecific sticking of antibodies to tissue components, much like in Western blots. For example, an IHC wash buffer might be PBS + 0.1% Tween‑20 (often called PBST) and multiple washes are done to remove unbound antibody. The result is lower background staining and crisper contrast between specific staining and the surrounding tissue. Tween‑20 has been used as a blocking agent for membrane-based assays at ~0.05%, and similarly, in IHC it can be present in the blocking or serum buffer to saturate nonspecific binding sites on tissue.
Another benefit in IHC is that Tween 20 can enhance reagent penetration in thicker tissue sections. While it is not a strong permeabilizer (see IF section below), its surfactant action helps antibodies and detection reagents to diffuse more uniformly through the tissue surface. Manufacturers of automated IHC stainers often add Tween 20 to their buffers to improve “reagent spreading” on slides. For instance, Biocare Medical’s instructions for an IHC buffer mention that Tween 20’s purpose is to “decrease background staining and enhance reagent spreading in automated and manual procedures”. In summary, Tween 20 is a simple addition that greatly improves the reliability of IHC staining by reducing unwanted background and ensuring even, reproducible staining across the tissue section.
Immunofluorescence (IF) and immunocytochemistry on cells share
many similarities with IHC in terms of Tween‑20 usage. In IF staining (whether
on tissue sections or cultured cells), Tween‑20 is often used as a gentle
permeabilizing and washing agent. Unlike IHC, IF typically requires cell
membranes to be permeabilized (especially to stain intracellular targets).
Common permeabilization agents are Triton X-100 or Tween‑20, with Tween‑20
being the milder option. Tween‑20 at ~0.1–0.2% can permeabilize cellular
membranes to some extent while preserving morphology and protein epitopes
better than harsher detergents. Triton X-100 tends to fully solubilize membrane
lipids (risking extraction of proteins and cell shrinkage), whereas Tween‑20 creates
temporary “pore-like” defects in the membrane and has a more renaturing
effect on proteins. This gentleness can actually improve antibody access and
binding by keeping protein epitopes in a more native state.
In practice, IF protocols might use 0.1–0.5%
Tween‑20 in PBS for permeabilization/washing steps when a mild approach is
sufficient (for example, if only slight membrane perforation is needed or the
target is easily accessible). For instance, an ICC protocol for adherent cells
may recommend postfixation washes in PBS + 0.1% Tween‑20 and even use Tween‑20
as the sole permeabilizer if a gentle treatment is desired. Additionally, after
permeabilization (whether by Tween‑20 or another detergent), subsequent
antibody incubation buffers often include 0.05–0.1% Tween‑20 to keep cells wet
and reduce nonspecific staining. Including a bit of Tween‑20 in mounting or
washing steps also helps to avoid surface tension effects that can cause uneven
staining or drying artifacts on slides.
One caveat: Tween‑20, while milder than
Triton, still can affect cells at higher concentrations or longer exposures.
Studies have shown that even 0.03% Tween‑20, if left on cells for extended
periods (24 h), can induce temporary morphological changes and reduce cell
viability – though these effects were reversible upon washing out the Tween.
For example, in a virology experiment, treating kidney cells with 0.03% Tween‑20
for 24 hours caused cell membranes to swell and microvilli to decrease, along
with a rise in apoptosis markers, but normal morphology returned after the
Tween was removed. This indicates that in live-cell contexts, Tween‑20 must be
used sparingly. In fixed-cell IF assays, however, these viability concerns are
moot, and the focus is on balancing permeabilization with epitope preservation.
Researchers often choose Tween‑20 when a “less harsh” permeabilization is
needed or in combination with other detergents (e.g., a small amount of Tween‑20
plus saponin).
Beyond immunostaining, cell culture applications for Tween‑20 include using it as a solubilizing agent or to prevent cell clumping. Tween‑20 is generally considered biocompatible at low concentrations and is even tested for cell culture suitability (Sigma’s cell-culture grade Tween‑20 is tested at 0.15 mg/mL ≈ 0.015% for lack of cytotoxicity). In some cases, a tiny percentage of Tween‑20 is added to culture media to help dissolve a hydrophobic drug or compound being delivered to cells. For example, it has been reported that Tween‑20 can reverse multidrug resistance in tumor cells by increasing drug uptake – low concentrations of Tween‑20 enhance the accumulation of chemotherapy drugs in resistant cancer cells. This is attributed to Tween’s ability to mildly perturb the cell membrane or inhibit efflux pumps, thereby allowing more drug to enter and stay in the cells. Similarly, in microbiology, Tween‑20 (or the related Tween‑80) is added to broth to prevent microbial cells from clumping. A small amount (≤0.1%) of Tween can reduce the surface tension and disperse hydrophobic microbial spores or mycobacterial cells that would otherwise aggregate. For instance, preparing spore suspensions of fungi or Bacillus often involves Tween‑20 in water to achieve a uniform single-cell suspension.
In summary, within cell-based experiments, Tween‑20 serves as a gentle detergent that can be used to permeabilize fixed cells for IF, to reduce nonspecific binding in cell staining protocols, and even to aid in delivering compounds to living cells (with caution to avoid toxicity). Its mild nature compared to other detergents makes it valuable when preservation of fine cellular structures or antigen epitopes is important.
Over the decades of Tween‑20’s use,
several improvements and optimizations have been made to the reagent itself and
how it’s used:
In essence, the tweaks and
improvements have focused on enhancing Tween‑20’s stability and purity, and
adapting its use for new technology. These efforts allow Tween‑20 to continue
being a reliable component of experiments without introducing artifacts. For
example, adding a preservative (like ProClin) to Tween‑20-containing wash
buffers in IHC helps prevent microbial growth in the buffer reservoir over
time– an issue recognized and fixed in modern labs. Thanks to such refinements,
a bottle of Tween‑20 today is more consistent and long-lasting than those in
the past, and protocols are optimized to exploit its benefits fully while
avoiding pitfalls (like avoiding excessive concentrations or heat exposure).
• Non-Ionic & Mild: Tween 20’s non-ionic nature means it does not disrupt protein structure or protein–protein interactions as harshly as ionic detergents (like SDS). It preserves antigenicity in assays (antibodies retain their binding) and generally does not denature enzymes or ligands at working concentrations. This makes it ideal for assays where maintaining biological activity is crucial.
• Reduces Nonspecific Binding: Perhaps the greatest advantage is its ability to block nonspecific adsorption of proteins to surfaces. By coating well walls, membrane pores, and other surfaces, Tween 20 prevents “sticky” background and improves assay specificity. This leads to clearer results in ELISAs, blots, and histology, as discussed earlier.
• Broad Compatibility: Tween 20 works in a variety of buffer systems (PBS, TBS, etc.) and across a pH range typically used in biology (near neutral pH). It’s compatible with common blocking reagents (serum, BSA, milk), enzymes (HRP, AP), and does not chelate metal ions. It can be used in conjunction with other detergents (e.g., NP-40, Triton X-100) to fine-tune lysis strength. Also, it mixes well with alcohols for special procedures and is soluble in both water and many organic.
• Low Toxicity: Tween 20 is regarded as relatively non-toxic to cells and organisms at the concentrations used. It is even approved as a food additive (emulsifier E432) and used in pharmaceuticals (as an excipient in vaccine and drug formulations). In cell culture, low doses of Tween 20 (≪0.1%) usually do not harm mammalian cells significantly – indeed, some company certifies certain lots for cell culture use. This is an advantage when residual detergent might come into contact with live cells or when considering biocompatibility.
• Cost-Effective and Accessible: Tween 20 is inexpensive and widely available from multiple suppliers. A small volume goes a long way (a liter of 0.05% PBST contains only 0.5 mL of Tween 20). Its long history means it’s referenced in countless protocols, and most labs keep a bottle on hand. The longevity of those bottles is also good; if kept properly, Tween 20 stock can remain effective for years.
• Enhances Reagent Spreading: By reducing surface tension, Tween 20 ensures even coverage of reagents over surfaces and samples. This is an often underappreciated benefit – it prevents “ring effects” when liquids dry and helps washes rinse uniformly. In automated staining, this property helps avoid uneven staining or drying artifacts.
• Versatility: Outside classical immunoassays, Tween 20 finds use in diverse lab techniques – e.g. as a solubilizing agent for membrane proteins, in enzyme assays to keep substrates in solution, in PCR to reduce enzyme sticking, and even in nanoparticle synthesis as a stabilizer. This versatility means one detergent can serve many functions across protocols.
In summary, Tween‑20’s advantages
– gentle action, reduction of nonspecific binding, and broad compatibility –
are why it’s so widely used in labs. Its limitations are generally
manageable with good practices (fresh solutions, proper concentrations,
complementary detergents when needed). Researchers have learned through
experience where Tween‑20 works best and where it doesn’t, allowing them to
capitalize on its strengths in myriad applications.
Polysorbate 20 has firmly established
itself as a workhorse reagent in modern science. Virtually every molecular
biology or biochemistry laboratory uses Tween 20 as a standard component in
protocols. Its role today spans routine techniques and cutting-edge
applications alike: from ELISA, Western blotting, IHC, and IF to
high-throughput systems, microfluidic platforms, and biopharmaceutical
manufacturing. Its ability to reduce nonspecific binding and surface adsorption
makes it indispensable in clinical diagnostics—such as clean IHC staining in
hospitals and default inclusion in automated ELISA platforms. In the
pharmaceutical industry, Tween 20 helps stabilize protein therapeutics like
monoclonal antibodies and vaccines by preventing aggregation at air-liquid or
container interfaces. It also finds use in nanoparticle synthesis and mass
spectrometry prep, where its gentle surfactant properties preserve protein
function. The widespread adoption of standardized buffers like PBST and TBST
reflects its central role in assay reproducibility, while in educational
settings, it remains one of the first reagents used to illustrate the
importance of surface chemistry in biological systems.
In conclusion, Tween‑20 remains
deeply integrated in contemporary scientific workflows. It may not be
flashy or novel – in fact, its chemistry is over 75 years old – but it is
precisely this track record and reliability that keep it in use. Researchers
continue to rely on Tween‑20 because it consistently improves assay
performance, whether in a basic research lab doing a Western blot or a biotech
company formulating a new drug. While alternatives and tweaks are always being
explored, polysorbate 20’s unique balance of properties ensures it has a
lasting place on the lab shelf. In many ways, Tween‑20 exemplifies a “timeless”
reagent: invented in the mid-20th century, yet still indispensable in the 21st-century
laboratory for enabling accurate, low-background, and reproducible
experiments.