This website uses cookies to ensure you get the best experience on our website.
- Table of Contents
and ELISA kits, proteins related to Neutrophils.
Neutrophils are the most abundant type of white blood cells in humans and play a crucial role in the immune system's first line of defense against infection. As part of the innate immune system, neutrophils respond rapidly to invaders by recognizing and attacking harmful bacteria, fungi, and viruses. These cells are known for their ability to move quickly to the site of infection and perform phagocytosis, engulfing and destroying pathogens. Additionally, neutrophils release enzymes and reactive oxygen species that help to kill and digest microbes, often at the cost of their own survival – a process known as netosis, where they expel their DNA to trap pathogens. Research into neutrophils has expanded our understanding of their role not only in infection control but also in inflammation and immune regulation. Antibodies that target specific proteins and markers in neutrophils are crucial for investigating these processes, aiding in the development of diagnostic and therapeutic strategies for a wide range of diseases.
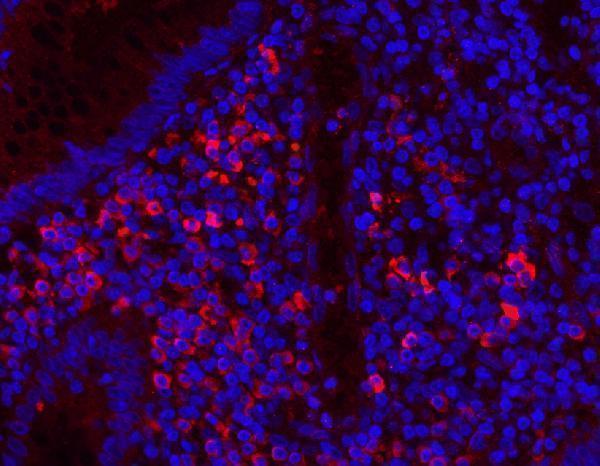
Anti-Myeloperoxidase/MPO Antibody Picoband®, IF analysis of MPO using anti-MPO antibody (PA1054).
MPO was detected in a paraffin-embedded section of human appendicitis tissue. Heat mediat...
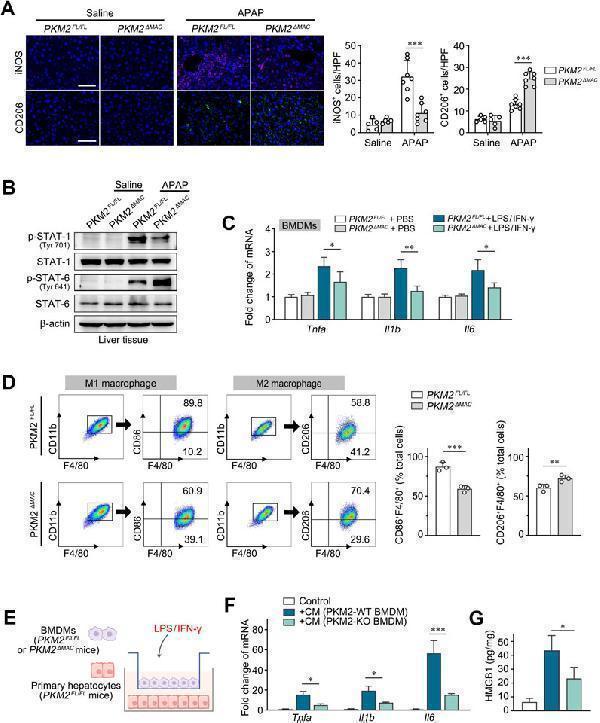
Anti-CD11b ITGAM Rabbit Monoclonal Antibody, Deletion of PKM2 in macrophages reprogrammed M1 macrophages to M2 macrophages. (A) Representative images and quantification of immu...

Anti-Neutrophil Elastase/ELANE Antibody Picoband®, NETs accumulate in the glomeruli and PTC of CI-AKI mice. ( A ) IF co-staining: CD31, red, vascular endothelial cell marke...
| Protein Name | Gene Name | Function |
|---|---|---|
| Lactoferrin | LTF | Involved in antimicrobial activity, iron transport and immunomodulation. |
| Myeloperoxidase | MPO | Produces hypochlorous acid for microbial killing. |
| Lysozyme | LYZ | Catalyzes the destruction of bacterial cell walls. |
| Elastase | ELANE | Serine protease that degrades various proteins including elastin during inflammation. |
| Cathepsin G | CTSG | Serine protease that regulates inflammatory processes and antimicrobial activity. |
| Neutrophil gelatinase-associated lipocalin | LCN2 | Binds small lipophilic substances such as fatty acids and steroids. |
| CD66b | CEACAM8 | Mediates cell adhesion and signaling in neutrophil activation. |
| Proteinase 3 | PRTN3 | Serine protease, involved in processing and presentation of antigens. |
| Neutrophil elastase | ELANE | Involved in tissue remodeling and degradation of extracellular proteins. |
| Neutrophil collagenase | MMP8 | Remodels extracellular matrix and degrades type I, II, and III collagens. |
| CEACAM1 | CEACAM1 | Involved in modulating inflammatory and immune responses. |
| CD 64 | FCGR1A | High affinity receptor for the Fc region of immunoglobulins. |
| Formyl peptide receptor | FPR1 | Receptor that mediates chemotaxis of neutrophils towards sites of inflammation. |
| Defensin Alpha 1 | DEFA1 | Antimicrobial peptides that disrupt microbial cell membranes. |
| CD10 | MME | Cell surface peptidase that degrades peptide hormones and modulates inflammatory responses. |
| CD11b | ITGAM | Integrin that promotes adhesion and migration of neutrophils. |
| Bactericidal/permeability-increasing protein (BPI) | BPI | Binds to bacterial lipopolysaccharides, neutralizing their toxic effects. |
| Azurocidin | AZU1 | Antibacterial protein with chemotactic properties for immune cells. |
| CD15 | FUT4 | Mediates phagocytosis and cell-cell interactions within the immune system. |
Neutrophil Extracellular Trap Formation, commonly known as NETosis, represents a crucial defense mechanism wherein neutrophils release extracellular fibers, composed of DNA, histones, and antimicrobial proteins. These structures, called Neutrophil Extracellular Traps (NETs), ensnare and neutralize pathogens. NETosis diverges from typical phagocytosis and cytokine production pathways, highlighting a unique aspect of innate immune response. While originally observed in the context of bacterial infections, recent studies have broadened the scope of NETs to anti-viral and anti-fungal efficacy, as well as implicating them in non-infectious disease processes such as thrombosis and autoimmune diseases. This bifacial role underscores its potential as a therapeutic target. Research into NETosis is pivotal as it offers insights not only into novel antimicrobial strategies but also into mechanisms underlying several chronic inflammatory and autoimmune conditions. Understanding the precise triggers and regulation of NETosis could lead to targeted treatments to modulate this process in pathological conditions where excessive or inappropriate NET formation exacerbates disease.
Neutrophil Chemotaxis is a fundamental process by which neutrophils respond to chemical signals to navigate toward sites of infection or inflammation. This targeted migration is essential for the effective functioning of the immune response, allowing neutrophils to reach and eliminate invading pathogens rapidly. The chemotactic process is mediated by a complex network of receptors on the neutrophil surface that detect chemokines, cytokines, and other signaling molecules. The ability of neutrophils to correctly interpret these chemical gradients is critical for both acute and chronic inflammatory responses. Errors in chemotaxis can lead to severe consequences, including chronic inflammatory states or failure to resolve infections. Researchers focus on the molecular pathways that control chemotaxis and how these can be adjusted, offering promising directions for therapies aimed at enhancing or dampening neutrophil function according to clinical need. Furthermore, advancements in imaging and computational biology are enabling deeper insights into the dynamics of neutrophil migration in real time and within complex biological environments.