This website uses cookies to ensure you get the best experience on our website.
- Table of Contents
and ELISA kits, proteins related to Pancreatic Cancer.
Pancreatic cancer is one of the most aggressive and deadly forms of cancer, often diagnosed at advanced stages due to its subtle early symptoms. This malignancy poses significant challenges in treatment, primarily because it tends to resist conventional therapies and rapidly spreads to surrounding organs. Our research focuses on the development of specialized antibodies aimed at targeting pancreatic cancer cells with greater precision. By harnessing the body’s immune system, these antibody-based therapies seek to identify and eliminate cancerous cells while minimizing damage to healthy tissues. Through innovative approaches and cutting-edge technology, we aim to improve detection, enhance treatment efficacy, and ultimately increase survival rates for those battling pancreatic cancer. Join us in our mission to transform pancreatic cancer research and bring hope to countless individuals affected by this formidable disease.
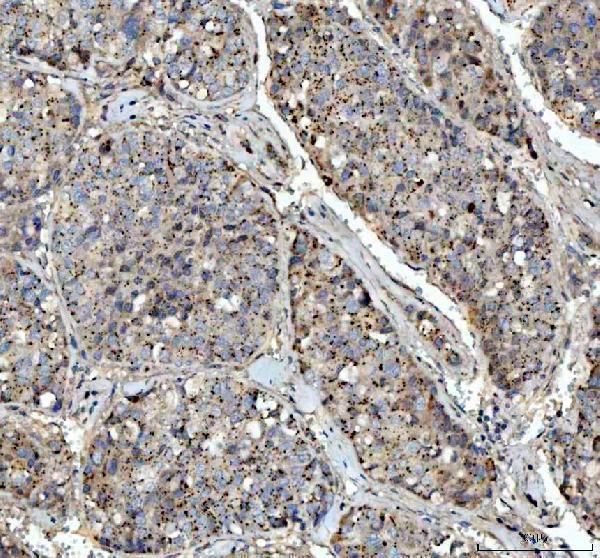
Anti-VEGF/VEGFA Antibody Picoband®, Figure 3. IHC analysis of VEGFA using anti-VEGFA antibody (PA1080).
VEGFA was detected in a paraffin-embedded section of human liver cancer tissue. Hea...
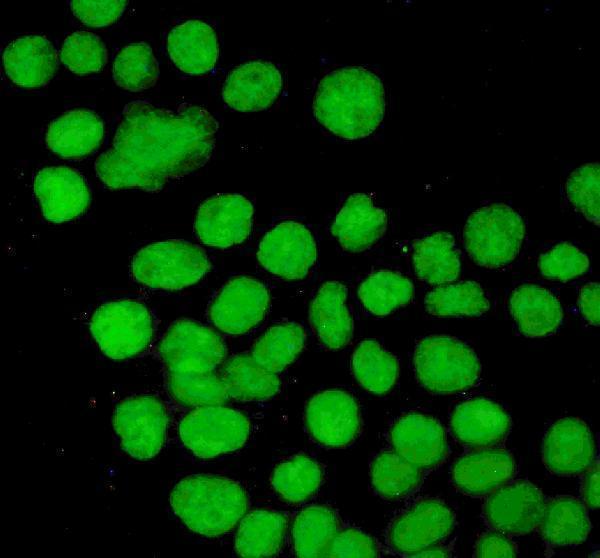
Anti-P53/TP53 Antibody Picoband®, Figure 3. IF analysis of P53 using anti-P53 antibody (PB9008).
P53 was detected in immunocytochemical section of A431 cells. Enzyme antigen...

Anti-P53 Tp53 Monoclonal Antibody, Figure 3. IF analysis of p53 using anti-p53 antibody (MA1078).
p53 was detected in immunocytochemical section of A431 cell. Enzyme antigen retrieva...
| Protein Name | Gene Name | Function |
|---|---|---|
| CA19-9 | FUT3 | Used for diagnosis and monitoring treatment response. |
| CEA | CEACAM5 | Tumor marker used in diagnosis and monitoring. |
| KRAS | KRAS | Oncogene mutation involved in cell signaling. |
| TP53 | TP53 | Tumor suppressor gene mutation linked to tumor progression. |
| CDKN2A (p16) | CDKN2A | Cell cycle regulator, tumor suppressor. |
| SMAD4 (DPC4) | SMAD4 | TGF-beta signaling, tumor suppression. |
| BRCA2 | BRCA2 | DNA repair, associated with familial pancreatic cancer. |
| EGFR | EGFR | Growth factor receptor involved in cell proliferation. |
| MUC1 | MUC1 | Cell surface mucin involved in cell signaling and adhesion. |
| HER2/neu | ERBB2 | Growth factor receptor involved in cell growth. |
| CA125 | MUC16 | Biomarker for various cancers, including pancreatic. |
| MET | MET | Receptor tyrosine kinase involved in cell growth and metastasis. |
| VEGF | VEGFA | Angiogenesis factor. |
| MIC-1 (GDF15) | GDF15 | Growth differentiation factor involved in cancer progression. |
| Osteopontin | SPP1 | Cell adhesion and signaling. |
| Mesothelin | MSLN | Cell surface protein overexpressed in pancreatic cancer. |
| S100A4 | S100A4 | Metastasis-associated protein. |
| Survivin | BIRC5 | Inhibitor of apoptosis. |
| DPC4 | SMAD4 | TGF-beta signaling. |
The tumor microenvironment (TME) in pancreatic cancer plays a pivotal role in disease progression and resistance to therapy. Unlike many other cancers, pancreatic tumors are characterized by a dense desmoplastic stroma, which consists of activated pancreatic stellate cells, extracellular matrix proteins, immune cells, and blood vessels. This unique TME fosters an immunosuppressive environment, hindering effective immune surveillance and facilitating tumor growth and metastasis. The stroma not only acts as a physical barrier, limiting the delivery of chemotherapeutic agents, but also secretes a variety of cytokines and growth factors that promote cancer cell survival, angiogenesis, and invasion. Additionally, the interaction between cancer-associated fibroblasts and cancer cells within the TME contributes to the activation of signaling pathways that drive pancreatic cancer progression. Understanding the complexities of the tumor microenvironment is essential for developing novel therapeutic strategies aimed at disrupting these interactions, enhancing drug delivery, and reactivating the immune response against pancreatic cancer cells.
KRAS mutations are a hallmark of pancreatic ductal adenocarcinoma (PDAC), occurring in over 90% of cases, and play a central role in driving tumorigenesis and disease progression. The KRAS protein is a key oncogene involved in regulating cell proliferation, differentiation, and survival. Mutations in KRAS result in its constitutive activation, leading to the persistent activation of downstream signaling pathways such as the MAPK/ERK and PI3K/AKT pathways. These signaling cascades promote uncontrolled cell growth, evade apoptosis, and enhance metastatic potential. The presence of KRAS mutations also contributes to the desmoplastic reaction characteristic of PDAC by modulating the tumor microenvironment and promoting fibrosis. Targeting KRAS-driven pathways has been challenging due to the protein's high affinity for GTP and lack of suitable binding pockets for inhibitors. However, recent advances in targeted therapies, including the development of KRAS G12C inhibitors and strategies to disrupt downstream effectors, hold promise for effective treatments. Continued research into the molecular mechanisms of KRAS signaling and its interactions within the pancreatic cancer context is crucial for the development of targeted therapeutic interventions aimed at improving patient outcomes.