This website uses cookies to ensure you get the best experience on our website.
- Table of Contents
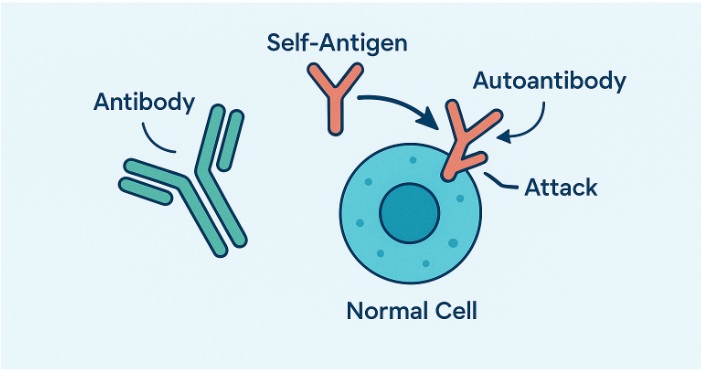
Roughly 10% of people worldwide live with an autoimmune condition, and more than 80 of these disorders trace at least part of their pathology to antibodies that have turned on the body. These rogue molecules—often referred to as autoantibodies—trigger inflammation, degrade healthy tissue, and keep the immune system in a state of overdrive. Understanding antibody function, how they arise, damage organs, and respond to treatment opens doors to sharper diagnostics, targeted therapies, and the development of specialized tools through antibody production services.
Antibodies usually patrol the blood, tagging microbes so other white blood cells and effector cells can finish the job. Autoantibodies appear nearly identical, yet they bind to self-antigens. Several checkpoints typically delete or re-educate B lymphocytes that make self-reactive antibodies, but these safeguards can fail. To study these processes and develop more precise treatments, researchers often use custom monoclonal antibodies to specifically detect and target immune molecules involved in autoimmunity.
The immune system relies on self-tolerance mechanisms to prevent attacks on the body’s own tissues.
Researchers investigating these checkpoints often use tools from Recombinant Antibody Production Service to track and study the behavior of self-reactive B cells with precision.
Specific HLA alleles, regulatory T-cell defects, viral mimicry, or toxins can weaken these checkpoints, allowing autoreactive clones to mature and expand. The resulting autoimmune response often involves chronic inflammatory disease and progressive tissue damage. In some organ-specific autoimmune diseases, such as Hashimoto thyroiditis or type 1 diabetes, localized antibody attacks target one organ, whereas systemic conditions affect multiple tissues.
Autoantibodies emerge through the same genetic tinkering that creates healthy antibody diversity, but with minor deviations.
Autoantibodies harm in several overlapping ways:
| Disease | Hallmark Autoantibodies | Diagnostic Value |
|---|---|---|
| Systemic lupus erythematosus | Anti-dsDNA, anti-Sm | High specificity; titres mirror flares |
| Rheumatoid arthritis | Anti-CCP, rheumatoid factor | Predict disease years before joint damage |
| Type 1 diabetes | Anti-GAD65, insulin autoantibodies, anti-ZnT8 | Appear months to years pre-symptom |
| Multiple sclerosis | Anti-MOG, anti-MBP | Useful in paediatric diagnoses |
| Hashimoto thyroiditis | Anti-TPO, anti-TG | Correlate with gland destruction |
| Graves disease | TSHR-stimulating antibodies | Direct cause of hyperthyroidism |
Multiplex panels that read dozens of specificities at once now catch overlapping syndromes that single autoantibody tests or isolated blood tests might miss.
Since sensitive ELISAs are often the first step in detecting autoantibodies, using an ELISA troubleshooting guide can help improve test accuracy and reliability. Western blots confirm specificity, while peptide arrays map epitope spreading, often using peptides for ELISA development to improve assay specificity and consistency across testing platforms. Serial measurements guide therapy—rising titers usually warn of an impending flare in autoimmune disorders.
Plasmapheresis quickly lowers antibody levels by filtering plasma, and IVIg floods Fc receptors with harmless IgG, dampening inflammation. For a longer-term solution, clinicians often turn to B-cell depletion with anti-CD20 antibodies, such as rituximab or ocrelizumab. New drugs that block the neonatal Fc receptor (FcRn) accelerate the breakdown of IgG, further reducing titers.
In parallel, researchers are exploring bispecific antibodies that can engage both autoreactive B cells and regulatory dendritic cells to restore tolerance. Other innovative therapies, including mRNA-lipid nanoparticles and peptide-coupled nanoparticles, may teach the immune system to ignore self-antigens without suppressing defense against infections.
Not every autoantibody is harmful. Natural IgM binds dying cells and cellular debris, preventing secondary inflammation. Memory B cells and regulatory B lymphocytes producing IL-10 may release low-affinity antibodies that reduce overactive immune responses. These protective mechanisms are crucial in maintaining balance.
Integrating B-cell receptor sequencing, transcriptomics, and proteomics promises the development of patient-specific autoantibody atlases. Early detection tools may spot individuals at risk long before symptoms start. Gene editing now enables scientists to replace autoreactive B-cell receptors with benign versions by reprogramming disulfide bond patterns in antibodies, suggesting durable cures.
Researchers continue to explore the differences between antibody structure, antibody function, and antibody responses in both organ-specific autoimmune diseases and systemic conditions, such as systemic lupus.
Autoantibodies sit at the core of many autoimmune disorders, steering everything from the first misfired antibody responses to chronic organ damage. Mapping their origins, tracking their rise and fall, and blocking their effects gives clinicians and scientists new leverage. As research refines every step—from B-cell selection to Fc receptor biology—treatments are moving from broad immunosuppression to precise, personalized intervention.
Ready to accelerate your autoimmune research?
Boster Bio supplies validated antibodies, ELISA kits, and custom antibody development services. Ask our scientific team how we can support your next experiment or diagnostic assay.