For researchers working with non-model organisms (e.g., zebrafish, Drosophila), choosing between
custom-made antibodies and commercial catalog antibodies is not just a procurement decision—it is a
reproducibility and study-design decision. Evidence from the antibody validation community emphasizes that performance hinges on
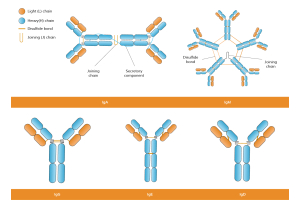
epitope-level specificity, application-matched validation, and long-term lot consistency rather than whole-protein homology alone
[Nature,
Nature Methods].
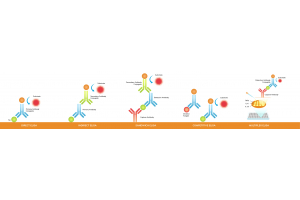
In practice, “custom antibody vs catalog antibody” often comes down to whether your ortholog’s critical epitope is conserved and
whether the reagent has been validated in your exact organism and application (WB, IF, IHC, whole-mount IF, IP/ChIP, Flow).
Recent discussions on antibody characterization and reproducibility further argue for context-specific validation and
sequence-defined supply when projects span multiple years or require publishable robustness
[eLife, 2022; 11:e72863].
This article provides a research-grade framework to decide when to prioritize a species-specific, bespoke antibody and when a
well-validated off-the-shelf reagent suffices. It also outlines pricing norms—including an
affordable custom antibody $600 entry point—so principal investigators and biotech decision-makers can weigh
time-to-data, total cost of results, and reproducibility.



.jpg)
.jpg)
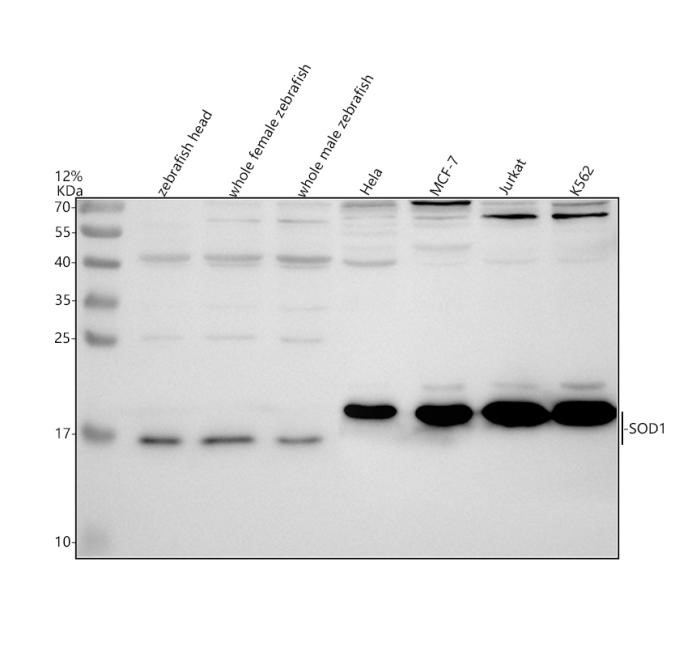
.jpg)