This website uses cookies to ensure you get the best experience on our website.
- Table of Contents

Looks right doesn’t mean it is—identify and fix non-specific staining in IHC.
Immunohistochemistry (IHC) visualizes protein expression and localization within intact tissues, providing unique spatial data unavailable from
Western blotting or bulk RNA methods alone.
However, non-specific staining is a major IHC pitfall: it obscures true signals, mimics specific staining, and causes misinterpretation of protein localization—especially when the pattern seems plausible but is not target-specific.
For this reason, reducing background is only part of the solution—recognizing non-specific staining and validating signal specificity are equally critical. This guide covers:
Non-specific staining refers to signal not caused by specific antibody-antigen binding, but from tissue components, detection reagents, antibody non-specific interactions, or technical artifacts.
Since IHC relies on spatial visualization, non-specific staining can be deceptive. Reliable interpretation requires combining controls, biological logic, and validation.
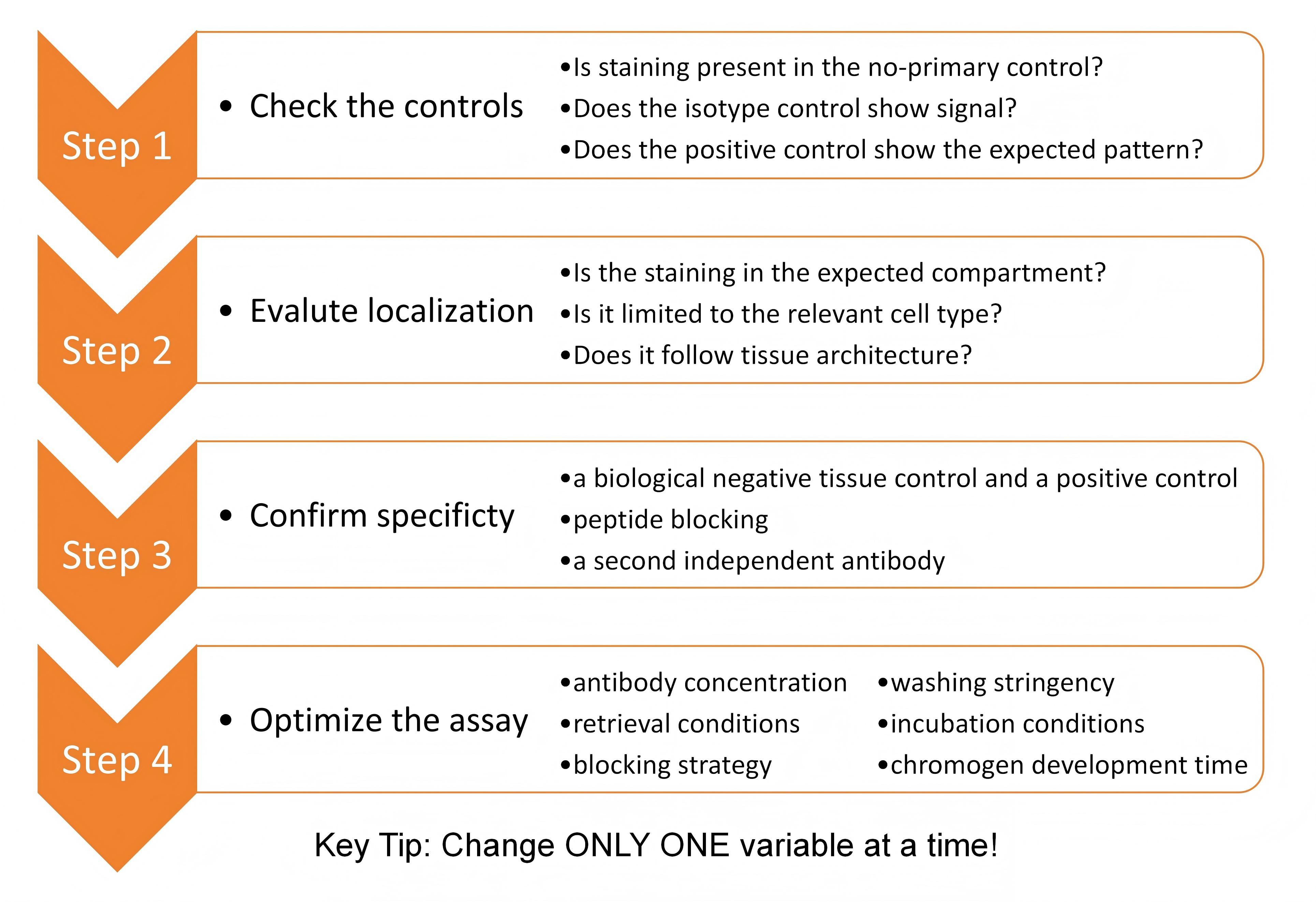
Recognizing non-specific staining is the first step in troubleshooting. A signal should not be accepted as real simply because it is visible. Instead, it should be evaluated from several angles:
Controls are the foundation of valid IHC interpretation:
Omits primary antibody to detect background from secondary antibody, endogenous enzymes, detection reagents, or chromogen artifacts. Staining here indicates the signal from the detection system, endogenous enzymes, or artifacts-not from target-specific binding.
An isotype control uses an immunoglobulin that matches the isotype of your primary antibody but does not recognize your target antigen. It helps assess non-specific binding from Fc receptors, charge interactions, or tissue components.
A positive control verifies target detection in a known positive sample. Beyond confirming signal presence, it also checks whether the staining pattern matches biological expectations. Strong yet poorly localized or widespread staining in a positive control indicates specificity or protocol issues, not a successful assay.
Whenever possible, include a tissue known to lack or minimally express the target. This can provide stronger evidence than reagent-only controls.
Specific staining usually has a pattern that is:
By contrast, non-specific staining often looks:
These patterns do not always prove non-specificity, but they should trigger caution.
A strong signal is not necessarily a correct signal. One of the most useful questions in IHC interpretation is:
Does this staining pattern make biological sense for this protein?If a known nuclear protein appears predominantly cytoplasmic, or a membrane protein appears as diffuse nuclear staining, the result should be interpreted cautiously.
A true signal is typically reproducible. Dramatic changes in staining patterns between similarly prepared sections indicate unstable assay conditions or background-driven signal.
Look for:
Large variability usually points to significant technical interference, not genuine biological signal.
A common IHC troubleshooting mistake is jumping straight to adjusting blocking, antibody dilution, or antigen retrieval without verifying antibody specificity firstly.
No amount of protocol tweaking can fix a fundamentally non-specific antibody.
For routine tissue IHC, you can quickly confirm specificity with a biological negative tissue control, a positive control, a second independent antibody, or a peptide blocking assay.
Only when you’ve confirmed your antibody binds specifically in your sample should you proceed to optimize staining conditions.
Endogenous peroxidase/biotin/alkaline phosphatase, or Fc receptors in immune tissues cause background.
How to reduce these problems?
The antibody used at an excessive concentration may bind weakly to unrelated tissue components, increasing background.
An antibody especially pAb may recognize proteins other than the intended target, especially if related proteins share homologous regions. The secondary antibody may bind endogenous immunoglobulins or tissue components, particularly if the species combination is not ideal.
How to reduce antibody-related background
Poor fixation can lead to non-specific staining or reduce signal clarity.
Under-fixation may cause:
Over-fixation can result in:
Tissue drying can lead to:
Best practices:
Antigen retrieval is one of the most important IHC-specific variables and one of the most common sources of both weak signal and excessive background.
Under-retrieval can lead to:
Over-retrieval can cause:
Blocking reduces non-specific antibody-tissue interactions.
Insufficient blocking can cause:
Common blocking methods:
Adjust blocking based on:
Poor washing and incubation conditions can lead to problematic staining, even with a good antibody.
Practical recommendations:
Sometimes the problem is not the antibody itself, but the detection chemistry.
Tissue type impacts background:
Q1.Why do I see high background in all samples, including controls?
This often suggests that the background is derived from the detection system, endogenous tissue components, or insufficient blocking rather than from target-specific antibody binding.
Q2. Why does my positive control also look messy?
A positive control should not just be positive. It should also show a pattern that is biologically sensible and reasonably clean.
If your positive control shows diffuse or widespread background, possible reasons include:
Q3. Why my antibody works in WB but not in IHC?
Possible reasons include:
WB and IHC place different demands on antibodies, so performance in one application does not guarantee performance in another.
Q4. Should I lower antibody concentration first, or change the retrieval condition first?
If the main problem is broad background, a lower antibody concentration is often one of the first variables to test. However, retrieval conditions should also be reviewed, especially if the staining is both weak and messy.
A practical approach is:
First confirm specificity as well as possible.
Then perform a small matrix of antibody dilution and retrieval conditions.
This allows you to improve signal-to-background ratio systematically.
Q5. If staining matches what I expected biologically, can I trust it?
Not automatically. Plausible-looking staining is encouraging, but it is not proof of specificity. Some background patterns can appear convincing, especially if the tissue contains many structurally complex features.
Biological plausibility should be considered supportive, not definitive, unless combined with controls and validation.
Non-specific staining in IHC is common, but it can be approached systematically.
The most important principles are:
Reliable IHC interpretation depends on both good assay design and strong antibody validation. Combined with thoughtful troubleshooting, these principles help distinguish true signal from artifacts and ensure reproducible, interpretable results.
If you're facing IHC challenges, be patient, check every step,and learn from the process.
See more IHC support