This website uses cookies to ensure you get the best experience on our website.
- Table of Contents
and ELISA kits, proteins related to macrophage.
Macrophages are vital immune cells that play a crucial role in maintaining the body's defense mechanisms. As part of the innate immune system, they are responsible for identifying, engulfing, and digesting pathogens, dead cells, and debris. Beyond their role in phagocytosis, macrophages orchestrate the immune response by releasing cytokines and presenting antigens to other immune cells, thereby bridging innate and adaptive immunity. Their ability to adapt and change function in different environments makes them key players in inflammation, tissue repair, and homeostasis. In the context of antibody research, understanding macrophage interactions with antibodies is essential for developing targeted therapies and improving immune response modulation. Advances in macrophage-related studies are paving the way for innovative treatments for various diseases, including autoimmune disorders, infections, and cancer, highlighting their significance in both health and disease.

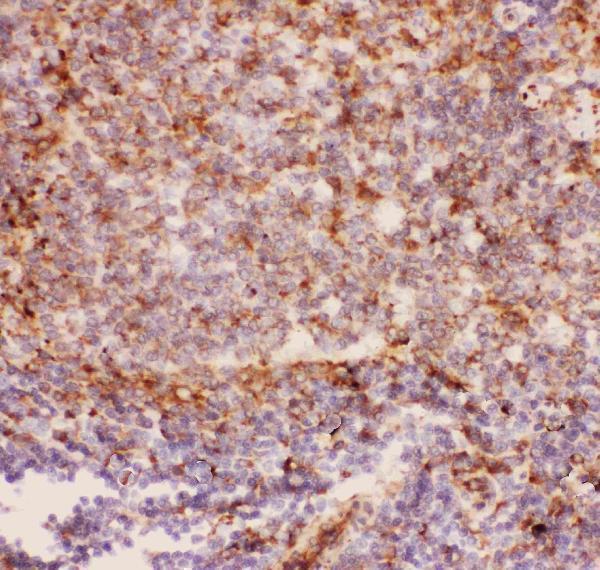
Anti-Macrosialin CD68 Antibody Picoband®, Figure 8. IF analysis of CD68 using anti-CD68 antibody (PA1518).
CD68 was detected in a paraffin-embedded section of mouse spleen tissue. Heat me...
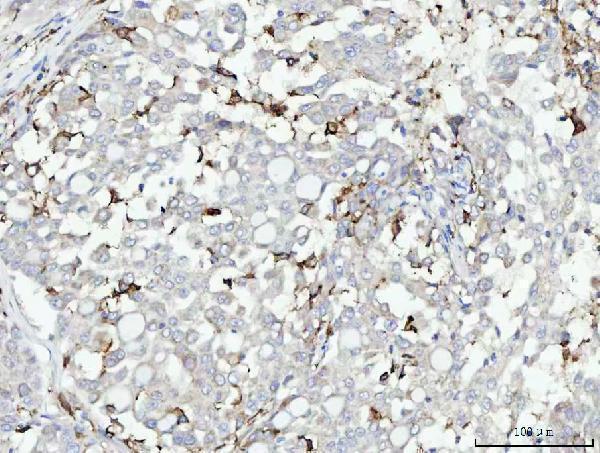
Anti-CD11b/ITGAM Antibody Picoband®, Figure 4. IHC analysis of CD11b using anti-CD11b antibody (PB9140).
CD11b was detected in a paraffin-embedded section of rat spleen tissue....
| Protein Name | Gene Name | Function |
|---|---|---|
| CD68 | CD68 | A lysosomal/endosomal marker involved in phagocytosis and antigen presentation |
| CD163 | CD163 | Scavenger receptor for haptoglobin-hemoglobin complexes, marker for anti-inflammatory macrophages |
| CD14 | CD14 | Co-receptor for the detection of bacterial lipopolysaccharide (LPS) |
| CD11b | ITGAM | Integrin involved in adhesion and migration of macrophages |
| CD206 | MRC1 | Mannose receptor involved in endocytosis and phagocytosis |
| MHCII | HLA-DRA | Major histocompatibility complex class II for antigen presentation |
| iNOS | NOS2 | Enzyme producing nitric oxide, important for microbial killing |
| ARG1 | ARG1 | Arginase involved in polyamine synthesis and wound healing |
| CCR2 | CCR2 | Chemokine receptor important for monocyte recruitment |
| CX3CR1 | CX3CR1 | Chemokine receptor involved in adhesion and migration |
| MARCO | MARCO | Scavenger receptor involved in binding and internalizing pathogens |
| SR-A | MSR1 | Scavenger receptor for modified LDL and apoptotic cells |
| CD80 | CD80 | Co-stimulatory molecule for T cell activation |
| CD86 | CD86 | Co-stimulatory molecule involved in T cell costimulation |
| TNF-alpha | TNF | Pro-inflammatory cytokine involved in systemic inflammation |
| IL-1beta | IL1B | Pro-inflammatory cytokine involved in immune response |
| Ly6C | LY6C2 | Marker for inflammatory monocytes in mice |
| TREM2 | TREM2 | Receptor involved in phagocytosis and inflammatory regulation |
| CD169 | SIGLEC1 | Sialoadhesin involved in cell-cell interactions and pathogen recognition |
Macrophage polarization refers to the ability of macrophages to adopt different functional phenotypes in response to various environmental cues. This plasticity is crucial for their role in maintaining homeostasis, responding to infections, and facilitating tissue repair. The two primary polarization states are the pro-inflammatory M1 phenotype and the anti-inflammatory M2 phenotype. M1 macrophages are typically induced by stimuli such as interferon-gamma (IFN-γ) and lipopolysaccharides (LPS), leading to the production of pro-inflammatory cytokines, reactive oxygen species, and enhanced microbicidal activity. In contrast, M2 macrophages are activated by interleukins like IL-4 and IL-13, promoting tissue remodeling, wound healing, and the resolution of inflammation. Understanding the mechanisms governing macrophage polarization is vital for developing therapeutic strategies aimed at manipulating macrophage responses in various diseases, including chronic inflammatory conditions, autoimmune disorders, and cancer. Recent research has also highlighted the existence of additional macrophage subsets beyond the classical M1 and M2 classifications, underscoring the complexity of macrophage biology and the need for a more nuanced understanding of their roles in health and disease.
Tumor-associated macrophages (TAMs) are a significant component of the tumor microenvironment and play a pivotal role in cancer progression and metastasis. Typically exhibiting an M2-like phenotype, TAMs contribute to tumor growth by promoting angiogenesis, suppressing anti-tumor immune responses, and facilitating the remodeling of the extracellular matrix, which aids in tumor invasion and dissemination. Additionally, TAMs secrete various cytokines and growth factors that support tumor cell survival and proliferation. The interaction between TAMs and cancer cells creates a dynamic environment that can enhance resistance to chemotherapy and immunotherapy, posing challenges for effective cancer treatment. Current research is focused on targeting TAMs to reprogram them from a pro-tumoral to an anti-tumoral phenotype, thereby enhancing the efficacy of existing cancer therapies. Strategies include inhibiting TAM recruitment to tumors, blocking their survival signals, and modulating their functional states through immune checkpoint inhibitors and other molecular interventions. Understanding the intricate roles of TAMs continues to be essential for the development of novel therapeutic approaches aimed at improving cancer patient outcomes.