This website uses cookies to ensure you get the best experience on our website.
- Table of Contents
and ELISA kits, proteins related to Acute myeloid leukemia.
Acute myeloid leukemia (AML) is a rapid-developing form of blood and bone marrow cancer that predominantly affects adult populations, though it can occur at any age. Characterized by an overproduction of immature white blood cells, known as myeloblasts or leukemic blasts, AML disrupts normal hematopoiesis—the process by which the body produces blood cells. These abnormal cells crowd out healthy cells, leading to severe symptoms such as infections, anemia, and bleeding disorders. Research in AML is pivotal as it explores avenues for better diagnostic markers, targeted therapies, and understanding of genetic mutations related to the disease. Antibodies, vital components in immune response, hold potential in both diagnostic and therapeutic capacities. They can be engineered to target specific AML cells, delivering treatments directly and sparing healthy cells, thus offering hopes for increased survival rates and reduced side effects. As research progresses, understanding these molecular mechanisms deepens, paving the way for innovative treatments and ultimately, a cure.
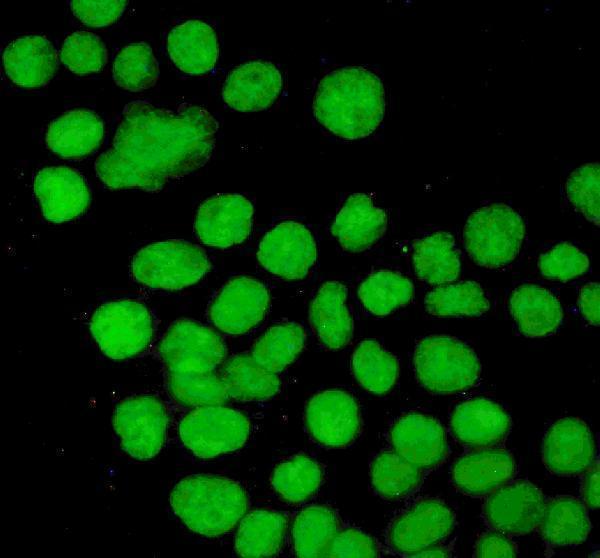
Anti-P53/TP53 Antibody Picoband®, IF analysis of P53 using anti-P53 antibody (PB9008).
P53 was detected in immunocytochemical section of A431 cells. Enzyme antigen retrieva...

Anti-P53 Tp53 Monoclonal Antibody, IF analysis of p53 using anti-p53 antibody (MA1078).
p53 was detected in immunocytochemical section of A431 cell. Enzyme antigen retrieval was per...

Anti-JAK2 Monoclonal Antibody, IF analysis of JAK2 using anti-JAK2 antibody (M00027) and anti-Beta Tubulin antibody (M01857-3).
JAK2 was detected in immunocytochemical section of A549 cell. E...
| Protein Name | Gene Name | Function |
|---|---|---|
| FLT3 | FLT3 | Regulates cell survival, growth, and differentiation and is commonly mutated in AML. |
| NPM1 | NPM1 | Involved in ribosome biogenesis and export, frequently mutated; mutations often lead to aberrant cytoplasmic dislocation. |
| TP53 | TP53 | Tumor suppressor gene involved in cell cycle regulation and apoptosis, mutations are associated with poor prognosis in AML. |
| CEBPA | CEBPA | Transcription factor essential for hematopoietic stem cell differentiation, biallelic mutations are often seen. |
| KIT | KIT | Tyrosine kinase receptor critical for stem cell growth; mutations are associated with progression and poor outcomes. |
| RUNX1 | RUNX1 | Transcription factor critical for hematopoietic differentiation, frequently mutated. |
| ASXL1 | ASXL1 | Regulates chromatin remodeling, mutations are associated with adverse outcomes in AML. |
| WT1 | WT1 | Transcription factor involved in cell growth and survival, mutations are common in AML. |
| DNMT3A | DNMT3A | Involved in DNA methylation and gene regulation, mutations are frequent and associated with prognosis. |
| IDH1 | IDH1 | Metabolic enzyme, mutations lead to metabolic shifts and epigenetic changes influencing AML pathogenesis. |
| IDH2 | IDH2 | Enzyme in the citric acid cycle, mutations result in abnormal cell differentiation. |
| TET2 | TET2 | Involved in DNA demethylation, mutations contribute to clonal hematopoiesis and AML progression. |
| PTPN11 | PTPN11 | Protein tyrosine phosphatase, mutations disrupt normal hematopoietic differentiation. |
| NRAS | NRAS | GTPase involved in signal transduction pathways, mutations lead to uncontrolled proliferation. |
| KRAS | KRAS | Involved in signaling pathways controlling cell division, mutations are influential in AML development. |
| MPL | MPL | Thrombopoietin receptor, mutations can contribute to myeloproliferation disorders. |
| JAK2 | JAK2 | Tyrosine kinase involved in signaling pathways, mutations associated with a variety of hematological malignancies. |
| BCOR | BCOR | Corepressor for BCL6 transcription repression, mutations associated with poor outcome in AML. |
| SF3B1 | SF3B1 | Spliceosomal complex component, mutations are linked to myelodysplastic syndromes progressing to AML. |
| SRSF2 | SRSF2 | Serine/arginine-rich splicing factor, mutations affect mRNA splicing and are related to myelodysplasia and secondary AML. |
In acute myeloid leukemia (AML), understanding leukemic stem cell (LSC) biology is paramount, as these cells are considered the root of leukemia initiation and persistence. LSCs are a subpopulation of leukemic cells with stem cell-like properties, including self-renewal and differentiation, akin to normal hematopoietic stem cells, but leading to the malignant production of abnormal blood cells. Research in this area focuses on the identification and characterisation of these cells, investigating their unique molecular and genetic signatures. Insights into LSC biology not only aid in revealing the pathogenesis of AML but are also crucial for developing targeted therapies. Eliminating LSCs is challenging, as they often exhibit resistance to conventional chemotherapy treatments. Advances in understanding the microenvironmental factors sustaining LSCs and their immunophenotypic markers are crucial for designing precision therapies that specifically target these cells and potentially lead to improved patient outcomes and reduced relapse rates.
The molecular genetics of acute myeloid leukemia involves the investigation of genetic mutations and their roles in the pathogenesis of the disease. This research area is critical as it informs the development of targeted therapies that can specifically inhibit the activity of the mutated genes driving leukemia. Key mutations often studied include FLT3, IDH1, IDH2, and TP53 among others, which are implicated in AML's heterogeneous nature. Emerging therapies that target these mutations offer new hope for more effective and less toxic treatments compared to traditional chemotherapy. This also includes the use of precision medicine approaches, such as tyrosine kinase inhibitors and monoclonal antibodies, aimed at specific genetic alterations in a patient’s leukemia cells. By tailoring treatment based on individual genetic profiles, much progress has been made towards providing personalized care that improves outcomes while minimizing adverse effects.