This website uses cookies to ensure you get the best experience on our website.
- Table of Contents
and ELISA kits, proteins related to Glioblastoma.
Glioblastoma is one of the most aggressive and devastating forms of brain cancer, originating from the glial cells that support nerve tissue. As the most common primary brain tumor in adults, glioblastoma presents significant treatment challenges due to its rapid growth and ability to infiltrate surrounding brain areas, making complete surgical removal difficult. Traditional therapies, including surgery, radiation, and chemotherapy, often provide limited success, leading to a poor prognosis for patients. In the quest for more effective treatments, recent research has focused on developing antibody-based therapies. These specialized antibodies are designed to target specific proteins on glioblastoma cells, aiming to disrupt tumor growth, enhance the immune system's response, and improve overall treatment outcomes. Our landing page delves into the latest advancements in glioblastoma research, highlighting the promising role of antibody therapies in the fight against this formidable cancer.
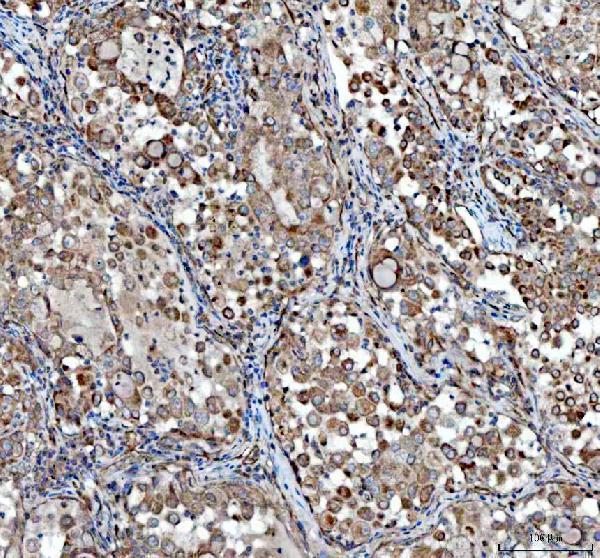
Anti-VEGF/VEGFA Antibody Picoband®, Figure 2. IHC analysis of VEGFA using anti-VEGFA antibody (PA1080).
VEGFA was detected in a paraffin-embedded section of human lung cancer tissue. Heat...
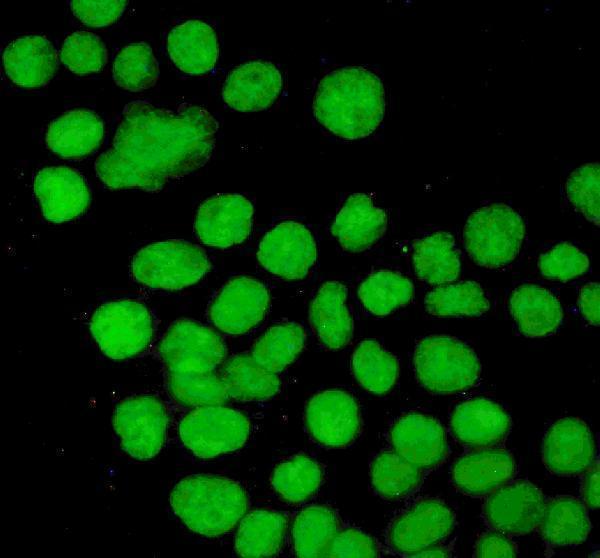
Anti-P53/TP53 Antibody Picoband®, Figure 3. IF analysis of P53 using anti-P53 antibody (PB9008).
P53 was detected in immunocytochemical section of A431 cells. Enzyme antigen...

Anti-P53 Tp53 Monoclonal Antibody, Figure 3. IF analysis of p53 using anti-p53 antibody (MA1078).
p53 was detected in immunocytochemical section of A431 cell. Enzyme antigen retrieva...
| Protein Name | Gene Name | Function |
|---|---|---|
| EGFR | EGFR | Epidermal Growth Factor Receptor involved in cell proliferation and survival. |
| MGMT | MGMT | O6-methylguanine-DNA methyltransferase involved in DNA repair. |
| IDH1 | IDH1 | Isocitrate dehydrogenase 1 involved in cellular metabolism. |
| PTEN | PTEN | Phosphatase and Tensin Homolog involved in tumor suppression. |
| TP53 | TP53 | Tumor Protein p53 involved in cell cycle regulation and apoptosis. |
| NF1 | NF1 | Neurofibromin 1 involved in cell growth regulation. |
| PDGFRA | PDGFRA | Platelet Derived Growth Factor Receptor Alpha involved in cell growth signaling. |
| VEGF | VEGFA | Vascular Endothelial Growth Factor involved in angiogenesis. |
| TERT | TERT | Telomerase Reverse Transcriptase involved in telomere maintenance. |
| CDKN2A | CDKN2A | Cyclin Dependent Kinase Inhibitor 2A involved in cell cycle regulation. |
| MDM2 | MDM2 | Mouse Double Minute 2 homolog involved in p53 regulation. |
| RB1 | RB1 | Retinoblastoma 1 involved in cell cycle control. |
| ATRX | ATRX | Alpha Thalassemia/Mental Retardation Syndrome X-Linked involved in chromatin remodeling. |
| PIK3CA | PIK3CA | Phosphoinositide-3-Kinase Catalytic Subunit Alpha involved in the PI3K/AKT pathway. |
| BRAF | BRAF | B-Raf Proto-Oncogene involved in the MAPK/ERK signaling pathway. |
| NOTCH1 | NOTCH1 | Notch Receptor 1 involved in cell differentiation. |
| KIT | KIT | KIT Proto-Oncogene Receptor Tyrosine Kinase involved in cell survival and proliferation. |
| ALK | ALK | Anaplastic Lymphoma Receptor Tyrosine Kinase involved in cell growth and differentiation. |
| MET | MET | MET Proto-Oncogene involved in cell growth, motility, and differentiation. |
| FGFR3 | FGFR3 | Fibroblast Growth Factor Receptor 3 involved in cell growth and differentiation. |
Glioblastoma is characterized by a complex network of molecular pathways and genetic alterations that drive its aggressive behavior and resistance to therapy. Critical pathways involved include the PI3K/Akt/mTOR pathway, which regulates cell growth and survival; the p53 and RB tumor suppressor pathways, which are frequently mutated leading to uncontrolled cell division; and the RTK/RAS/RAF pathway, often altered through mutations in EGFR, PTEN, and other receptors promoting oncogenic signaling. Additionally, alterations in DNA repair mechanisms and the presence of specific gene mutations, such as IDH1/2 and TERT promoter mutations, contribute to tumor heterogeneity and prognosis. Understanding these molecular underpinnings is essential for identifying potential therapeutic targets, developing personalized treatment strategies, and overcoming resistance mechanisms. Ongoing research in this area aims to elucidate the intricate genetic landscape of glioblastoma, paving the way for innovative therapies that can effectively target the specific aberrations present in individual tumors.
The tumor microenvironment (TME) in glioblastoma plays a pivotal role in tumor progression, therapy resistance, and immune evasion. The TME consists of various cell types, including immune cells such as microglia, macrophages, and regulatory T cells, which interact with tumor cells and contribute to an immunosuppressive milieu. Glioblastoma cells can secrete cytokines and growth factors that modulate the TME, promoting angiogenesis, extracellular matrix remodeling, and suppression of anti-tumor immune responses. Furthermore, the blood-brain barrier presents a unique challenge for immune cell infiltration and drug delivery. Understanding the interplay between glioblastoma cells and the TME is crucial for developing immunotherapeutic strategies, such as checkpoint inhibitors, CAR T-cell therapies, and vaccine-based approaches, aimed at reactivating the immune system to recognize and eliminate tumor cells. Research in this area seeks to identify novel targets within the TME and overcome the mechanisms by which glioblastoma evades immune surveillance, ultimately improving treatment outcomes for patients.