This website uses cookies to ensure you get the best experience on our website.
- Table of Contents
and ELISA kits, proteins related to Non-Hodgkin lymphoma.
Non-Hodgkin lymphoma (NHL) is a complex group of blood cancers that originates in the lymphatic system, a crucial component of the immune system. This disease affects lymphocytes, a type of white blood cell that plays an essential role in fighting infections. Unlike Hodgkin lymphoma, which has distinct characteristics, NHL encompasses a diverse array of subtypes, each with unique features and behaviors. NHL can develop in both adults and children, varying greatly in its aggression level—ranging from slow-growing forms that require minimal treatment to aggressive types that necessitate immediate, intensive therapy. The cause of Non-Hodgkin lymphoma remains largely unknown, although factors like immune system deficiencies, exposure to certain chemicals, and genetic predispositions are believed to increase risk. Advances in medical research are continuously improving the understanding and treatment of NHL, making the development and application of specific antibodies crucial for diagnostic and therapeutic strategies. Through ongoing research, the hope is to enhance targeted therapies that can more effectively treat and potentially cure Non-Hodgkin lymphoma.
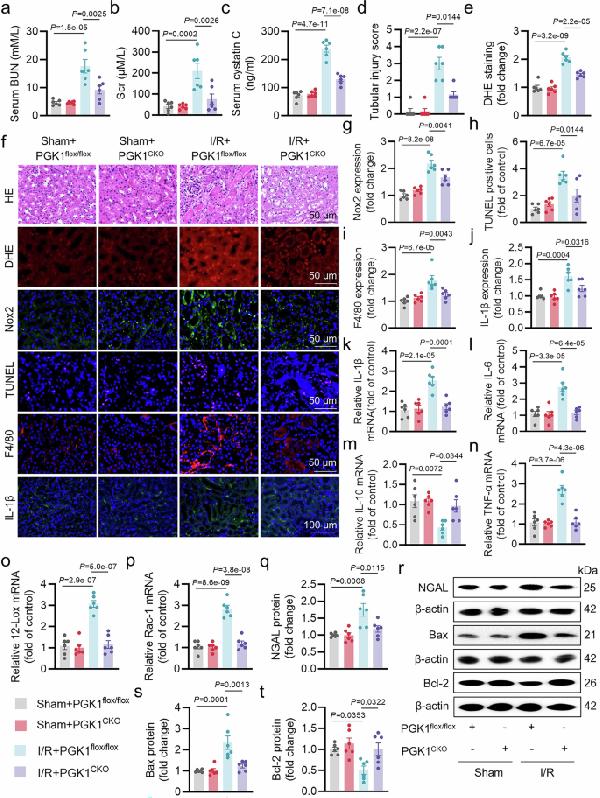
Anti-Bcl-2/BCL2 Antibody Picoband®, RTECs-specific knockout of PGK1 protected against I/R-induced AKI. After adaptation for one week, eight-week-old PGK1 flox/flox and PGK1 CK...

Anti-Ki67/MKI67 Antibody Picoband®, IF analysis of Ki67 using anti-Ki67 antibody (PB9026).
Ki67 was detected in paraffin-embedded section of human colon organoid tissue. He...

Anti-c-Myc Monoclonal Antibody, Immunofluorescent analysis using the Antibody at 1:50 dilution....
| Protein Name | Gene Name | Function |
|---|---|---|
| CD20 | MS4A1 | Commonly targeted by rituximab, involved in B-cell activation and development. |
| CD5 | CD5 | Expression indicates T-cell phenotype and some B-cell chronic lymphocytic leukemia. |
| CD10 | MME | Marker for germinal center B-cells, associated with certain aggressive B-cell lymphomas. |
| CD30 | TNFRSF8 | Expressed in Hodgkin lymphoma and some T-cell lymphomas, target for brentuximab vedotin. |
| BCL-2 | BCL2 | Anti-apoptotic protein, overexpressed in many lymphomas, associated with follicular lymphoma. |
| BCL-6 | BCL6 | Transcriptional repressor, plays role in germinal center formation, implicated in diffuse large B-cell lymphoma. |
| CD21 | CR2 | Part of the complement system, marker for follicular dendritic cells. |
| CD23 | FCER2 | Low-affinity IgE receptor, associated with B-cell chronic lymphocytic leukemia. |
| MUM1 | IRF4 | Transcription factor involved in plasma cell differentiation, expressed in multiple myeloma and some B-cell lymphomas. |
| ALK | ALK | Anaplastic lymphoma kinase, associated with anaplastic large cell lymphoma. |
| CD15 | FUT4 | Marker used in diagnosis of Hodgkin lymphoma. |
| Ki-67 | MKI67 | Nuclear protein associated with cellular proliferation, useful in grading the aggressiveness of lymphoma. |
| CD79a | CD79A | Part of the B-cell receptor complex, indicates B-cell lineage. |
| PAX5 | PAX5 | B-cell lineage specific activator protein, essential for B-cell development. |
| CD45 | PTPRC | Common leukocyte antigen, present on all hematopoietic cells except mature erythrocytes and platelets. |
| c-MYC | MYC | Transcription factor that regulates cell cycle progression, apoptosis and cellular transformation. |
| CD19 | CD19 | Critical for establishing intrinsic B cell signaling thresholds; its expression is required for B lineage development. |
| CD38 | CD38 | Marker and enzyme involved in cell activation, adhesion and calcium signaling, seen in plasma cell neoplasms. |
| CD22 | CD22 | Regulates B-cell function and B-cell receptor signaling, target for immunotherapy in B-cell malignancies. |
The immune microenvironment of Non-Hodgkin Lymphoma (NHL) plays a critical role in the development, progression, and treatment response of this diverse group of lymphoid cancers. Research in this area focuses on understanding how different components of the immune system interact with lymphoma cells and potentially support their growth and survival. Key cells investigated include T-cells, B-cells, macrophages, and various cytokines and chemokines that these cells produce. Innovations in this field are driving the development of immunotherapies such as checkpoint inhibitors, CAR T-cell therapies, and bi-specific antibodies. These treatments aim to modify the immune environment or harness the patient's immune system to more effectively target and destroy lymphoma cells. A better understanding of this sub area may lead to enhanced stratification of patients and more personalized therapeutic approaches, potentially improving prognosis and reducing the side effects of standard chemotherapy.
Molecular genetics is a pivotal research area in understanding the various subtypes of Non-Hodgkin Lymphoma, which is crucial for targeted therapy development. This research focuses on identifying and characterizing genetic mutations, chromosomal abnormalities, and gene expression profiles associated with different NHL subtypes. Techniques like whole-genome sequencing, RNA sequencing, and advanced bioinformatics tools are employed to discover genetic markers that can predict disease progression, treatment response, and patient prognosis. Such genetic insights not only enhance the understanding of lymphomagenesis but also lead to the development of precision medicine strategies. Targeted therapies, for instance, that specifically inhibit pathways altered by genetic mutations in lymphoma cells, have shown promising results in increasing treatment efficacy and reducing toxicity compared to conventional chemotherapies. Continued advancements in this field hold the promise of transforming NHL treatment paradigms, making therapy more effective and less burdensome for patients.