This website uses cookies to ensure you get the best experience on our website.
- Table of Contents
and ELISA kits, proteins related to Endometrial Cancer.
Endometrial cancer, primarily affecting the inner lining of the uterus (the endometrium), stands as a significant concern in women's health worldwide. This cancer type typically manifests in postmenopausal women, though it can also appear at younger ages. Characterized by symptoms like abnormal vaginal bleeding, pelvic pain, and weight loss, early detection and diagnosis are crucial for effective treatment outcomes. Research into endometrial cancer has increasingly focused on the role of biological markers and antibodies in both detection and therapeutic strategies. Scientists aim to understand the molecular and genetic underpinnings of the disease to develop targeted therapies that improve survival rates and quality of life for patients. The development and validation of specific antibodies that can reliably detect endometrial cancer cells or related biomarkers are at the forefront of this research. These antibodies not only hold the potential for early diagnostic applications but also pave the way for innovative treatment approaches, including immunotherapy and personalized medicine. As research progresses, the hope is to transition these scientific advancements from the laboratory to the clinic, offering new hope to those battling this challenging disease.

Anti-VEGF/VEGFA Antibody Picoband®, YJD affected the VEGF/VEGFR-2/FAK pathway in vivo. ( A – B ) Germ cell markers MVH and Oct4 were detected by IF. ( C ) The expression of VEGF, VEGFR-2, and...
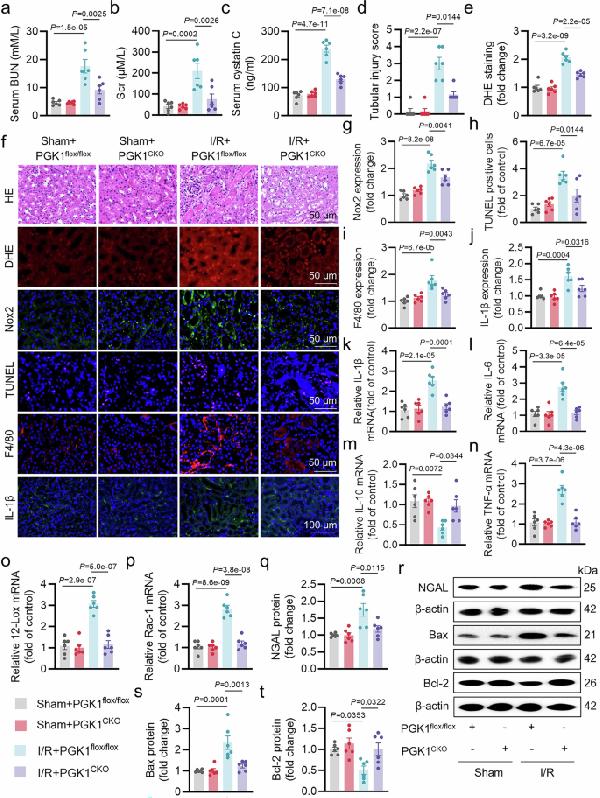
Anti-Bcl-2/BCL2 Antibody Picoband®, RTECs-specific knockout of PGK1 protected against I/R-induced AKI. After adaptation for one week, eight-week-old PGK1 flox/flox and PGK1 CK...
Anti-P53/TP53 Antibody Picoband®, IF analysis of P53 using anti-P53 antibody (PB9008).
P53 was detected in immunocytochemical section of A431 cells. Enzyme antigen retrieva...
| Protein Name | Gene Name | Function |
|---|---|---|
| PTEN | PTEN | Tumor suppressor gene involved in cell cycle regulation and apoptosis. |
| TP53 | TP53 | Regulates cell cycle and functions as a tumor suppressor. |
| PIK3CA | PIK3CA | Involved in the PI3K/AKT pathway, influencing cell growth and survival. |
| ARID1A | ARID1A | Chromatin remodeler that influences DNA repair and transcriptional regulation. |
| KRAS | KRAS | Plays a crucial role in cell signal transduction. |
| CTNNB1 | CTNNB1 | Key regulator in the Wnt signaling pathway, involved in cellular adhesion and gene transcription. |
| MLH1 | MLH1 | DNA mismatch repair protein, helps maintain genomic stability. |
| MSH2 | MSH2 | Part of the DNA mismatch repair system, preventing genomic mutations. |
| MSH6 | MSH6 | Works with MSH2 in the DNA repair process to maintain genomic integrity. |
| PMS2 | PMS2 | DNA repair protein that interacts with MLH1 in the mismatch repair pathway. |
| HER2 | ERBB2 | Oncogene important in cell growth and differentiation. |
| FGFR2 | FGFR2 | Fibroblast growth factor receptor involved in cell growth, differentiation, and repair. |
| VEGF | VEGFA | Stimulates vascular endothelial growth and angiogenesis. |
| EGFR | EGFR | Receptor tyrosine kinase involved in cell growth and division. |
| MET | MET | Hepatocyte growth factor receptor involved in cell growth, motility, and morphogenesis. |
| CCNE1 | CCNE1 | Regulates the cell cycle by controlling the transition from G1 to S phase. |
| CDK2 | CDK2 | Cyclin-dependent kinase important for cell cycle G1/S transition. |
| BCL2 | BCL2 | Anti-apoptotic protein, promotes cell survival. |
| KI67 | MKI67 | Nuclear protein that is associated with cellular proliferation. |
| CA125 | MUC16 | Tumor marker used in monitoring treatment and progression of ovarian cancer that can also be elevated in endometrial cancer. |
The study of molecular genetics in endometrial cancer is crucial for understanding the pathogenic processes underpinning the disease, as well as for developing targeted therapies. This sub-area scrutinizes the genetic and epigenetic alterations that trigger endometrial cancer, including mutations in genes such as PTEN, PIK3CA, ARID1A, and KRAS. Additionally, much attention is given to the role of the microsatellite instability (MSI) pathway and the interplay between estrogen-driven pathways and genetic predispositions. By mapping these genetic landscapes, researchers not only decipher the heterogeneity of endometrial cancer but also pave the way for personalized medicine, where treatments are tailored based on individual genetic profiles, potentially leading to better prognosis and survival rates.
Immunotherapy has emerged as a revolutionary treatment modality in endometrial cancer, particularly with the implementation of immune checkpoint inhibitors that target PD-1/PD-L1 and CTLA-4 pathways. As endometrial cancer often presents a high mutational burden, immunotherapy offers an effective treatment alternative that modulates the immune system to target and kill cancer cells. Crucially, research in this area also involves the identification of biomarkers to predict responsiveness to such therapies and studying the immune microenvironment of the tumor to further understand the mechanisms of immune resistance and tolerance. Comprehensive exploration in this field aims to not only extend the clinical benefits to a broader range of patients but also integrates with other treatments to reduce the recurrence rate and improve long-term outcomes.