This website uses cookies to ensure you get the best experience on our website.
- Table of Contents
Antibody conjugates are essential tools in biological research, offering both specificity and sensitivity for detecting and quantifying proteins, cells, and other molecules. Below, we explore the most common types of antibody conjugates, their examples, applications, and popularity in research.
Antibody conjugation is the process of chemically linking an antibody to another molecule, such as a fluorescent dye, enzyme, biotin, or nanoparticle. This process enhances the antibody’s ability to detect specific targets by enabling visualization or measurement in various assays. Conjugated antibodies are widely used in research for applications like flow cytometry, ELISA, and immunofluorescence, where they facilitate the detection and analysis of specific proteins or cells in complex samples. In some experimental setups, especially those involving gene delivery or expression studies, related tools such as AAV Packaging Service may also be employed to introduce genetic material efficiently into target cells. These conjugates are often produced as part of comprehensive antibody production services, where antibodies are not only generated but also tailored with the appropriate labels to suit specific experimental needs.

Among the most commonly used are fluorophore conjugates, which include dyes like fluorescein isothiocyanate (FITC), cyanine dyes, DyLight® dyes, allophycocyanin (APC), phycoerythrin (PE), R-phycoerythrin (R-PE), and iFluor® dyes.
Fluorophore-conjugated antibodies are widely used in:
Below, we have provided a table comparing key characteristics and uses of some of the most common fluorophore conjugates in research.
| Fluorophore | Color | Max Excitation (nm) | Max Emission (nm) | Extinction Coefficient (M⁻¹cm⁻¹) | Advantages | Applications |
|---|---|---|---|---|---|---|
| FITC | Green | 495 | 519 | 70,000 | Bright, photostable, common filter sets | Flow cytometry, immunofluorescence, microscopy |
| Cy3 | Orange | 552 | 570 | 150,000 | Bright, used in multiplexing | Flow cytometry, immunofluorescence, FISH |
| Cy5 | Red | 650 | 670 | 250,000 | Near-infrared, high sensitivity | Flow cytometry, imaging, FRET |
| DyLight® 488 | Green | 493 | 518 | 70,000 | Bright, photostable | Flow cytometry, immunofluorescence, microscopy |
| DyLight® 550 | Orange | 562 | 576 | 150,000 | High brightness, photostable | Western blotting, fluorescence microscopy, flow cytometry |
| DyLight® 594 | Red | 593 | 618 | 115,000 | Bright, minimal spectral overlap | Multicolor fluorescence imaging, flow cytometry |
| DyLight® 650 | Far-red | 652 | 672 | 250,000 | Near-infrared, reduced background | Flow cytometry, fluorescence imaging |
| DyLight® 800 | Near-IR | 783 | 800 | 270,000 | Near-infrared, minimal autofluorescence | In vivo imaging, Western blotting, NIR fluorescence imaging |
| iFluor® 488 | Green | 491 | 516 | 70,000 | Bright, photostable, FITC alternative | Flow cytometry, immunofluorescence, confocal microscopy |
| iFluor® 555 | Orange | 555 | 565 | 150,000 | High brightness, photostable | Fluorescence microscopy, flow cytometry, multicolor applications |
| iFluor® 594 | Red | 590 | 615 | 115,000 | Bright, minimal spectral overlap | Multicolor fluorescence imaging, flow cytometry |
| iFluor® 647 | Far-red | 650 | 665 | 250,000 | High brightness, photostable | Flow cytometry, fluorescence imaging, super-resolution microscopy |
| iFluor® 750 | Near-IR | 755 | 779 | 270,000 | Near-infrared, minimal autofluorescence | In vivo imaging, NIR fluorescence imaging |
| APC | Red | 650 | 660 | 700,000 | High quantum yield, photostable | Flow cytometry, imaging |
| PE | Orange | 480-565 | 575-590 | 1,960,000 | High brightness, quantum yield | Flow cytometry, fluorescence microscopy |
| R-PE | Red-orange | 488, 546, 565 | 575-585 | 1,960,000 | Extremely bright, multiple chromophores | Flow cytometry, high sensitivity applications |
Fluorophore conjugates are very popular due to their versatility, high sensitivity, and the variety of available dyes that allow multiplexing. When searching for primary antibodies and secondary antibodies at Boster, you’ll be able to select from a range of conjugation options, such as Cy3, DyLight® dyes, FITC, APC, PE, or iFluor® dyes. You can also request custom antibody conjugation with our antibody conjugation service, which offers more conjugate labels.
Enzyme conjugates, such as those linked to horseradish peroxidase (HRP) and alkaline phosphatase (AP), are also commonly used in research. These conjugates are crucial in assays like ELISA, WB, and IHC. In particular, enzyme-conjugated antibodies are widely utilized in sandwich ELISA formats, where the precise coordination between the capture and detection antibodies is essential for achieving optimal signal development and minimizing background interference. Antibody Pair Development Service develops matched antibody pairs for these assays involving careful selection to ensure that the antibodies bind to non-competing epitopes with high affinity and stability across varying assay conditions.
Enzyme-conjugated antibodies are used in:
Enzyme conjugates are highly popular in routine laboratory assays due to their robustness and ease of use. However, when assays demand superior specificity and minimal background noise, especially in enzyme-linked applications like ELISA and Western blotting, sourcing antibodies through specialized Rabbit Monoclonal Antibody Services can provide researchers with tailored solutions that consistently deliver reliable signal detection in complex biological samples. At Boster Bio, you can find primary antibodies and secondary antibodies conjugated to HRP, AP, and more. In addition, you can select specific conjugates for your antibodies with our custom antibody conjugation service.
Biotin, a vitamin that can be easily bound by streptavidin, has proven to be another essential antibody conjugate in research. It provides significant advantages due to its amplification capabilities. Biotin-labeled antibodies, often paired with streptavidin-HRP or AP, are used by researchers in ELISA, Western blotting, and immunohistochemistry.
In research, biotin-conjugated antibodies are frequently used in:
Biotin conjugates are widely used due to their ability to provide amplification for applications that require high sensitivity. Boster Bio's catalog contains biotin-conjugated primary antibodies and secondary antibodies, and additional conjugate options. You can also learn more about our custom antibody conjugation service and book a meeting with us to discuss your project, so we can better serve your research needs. Submit an inquiry today!
Metal conjugates, including lanthanide-chelated antibodies (e.g., Europium, Terbium) and metal isotope-tagged antibodies for mass cytometry (CyTOF), are gaining traction in advanced applications.
Metal-conjugated antibodies are used in:
Growing popularity of metal conjugates, especially in advanced applications like CyTOF, reflects their capability to provide comprehensive cellular analysis.
Quantum dot conjugates are semiconductor nanoparticles, including Qdot 525 and Qdot 655, known for their unique optical properties.
Quantum dot-conjugated antibodies are used in:
Although less common than traditional fluorophores, quantum dots (Qdots) are increasingly popular in imaging applications for their photostability and distinct spectral properties.
Gold nanoparticles (AuNPs) are widely employed in various diagnostics, biosensing, and imaging applications.
Gold nanoparticle-conjugated antibodies are used in:
Gold nanoparticle-conjugated antibodies are quite popular in diagnostics and increasingly in biosensing applications due to their practical utility and ease of detection.
Antibody conjugates play a vital role in modern research, with each type offering distinct advantages. Fluorophore and enzyme conjugates remain staples due to their broad applications and established protocols. Biotin conjugates are favored for applications requiring high sensitivity, while metal conjugates offer advanced analysis capabilities. Quantum dots and gold nanoparticles, though more specialized, are expanding in use as techniques and technologies improve. Selecting the appropriate conj...
Flow cytometry and Fluorescence-Activated Cell Sorting (FACS) are indispensable tools in biomedical research and clinical diagnostics. Despite their widespread use, confusion often arises regarding their terminology and functionalities. In this article, we identify distinctions between flow cytometry and FACS, and discuss their principles and applications.
Developed in the 1950s and 1960s, flow cytometry revolutionized cell analysis by allowing rapid, high-throughput measurement of multiple cellular characteristics. This technique analyzes the physical and chemical characteristics of particles or cells in a fluid suspension, and involves passing a cell-containing fluid stream through a laser beam, measuring the scattered and fluorescent light emitted by the cells.
Key aspects of flow cytometry include:
The avidin-biotin interaction remains one of the strongest non-covalent interactions between molecules, even surpassing the strength of antibody-antigen interactions. Interactions between these two molecules have been utilized in the laboratory for countless applications, such as immunohistochemistry (IHC), immunoprecipitation (IP), affinity purification, enzyme-linked immunosorbent assay (ELISA), and more—including advanced techniques like Multiplex Assay Services that allow simultaneous detection of multiple targets. The avidin-biotin-peroxidase complex has been used in countless assays for its simple and effective procedure, incomparable strength of binding between molecules, and ability to label and quantify lowly abundant proteins of interest in experiments. Boster Bio conveniently offers an Avidin-Biotin-Peroxidase Complex for ELISA kit (Catalog# AR1103), complete with necessary reagents, f...
What is CCK-8?
Cell Counting Kit-8 (CCK-8) is a readily available, easy-to-use cell viability and cytotoxicity assay. CCK-8 takes advantage of WST-8 [2- (2- methoxy-4-nitrophenyl)-3- (4-nitrophenyl)-5- (2, 4-disulfophenyl)-2H- tetrazolium, monosodium salt] to directly measure cell viability. Specifically, WST-8 is a water-soluble salt that can be reduced by dehydrogenases in viable cells to a water-soluble formazan dye. The biochemical reaction causes a change in color that can be easily quantified by measuring absorbance at 450nm.This simple and convenient colorimetric assay has been widely used to determine cell viability and cytotoxicity of various agents in cell culture and is also adaptable to broader workflows such as Multiplex Assay Services that assess multiple cellular parameters simultaneously, and is one of several assay services commonly employed in laboratory research. Boster Bio offers an affordable, highly cited Cell Counting Kit-8 (CCK-8) (Catalog# AR1160) for use in your cell culture experiments. CCK-8 assays are quick and easy to use since they do not require thawing or mixing any reagents before use. Simply add the reagent to cells in culture, incubate cells at standard conditions for up to four hours, then determine cell viability or cytotoxicity by measuring the colorimetric change in a cell plate reader at 450 nm.
CCK-8 measures cell viability by correlating the production of colored formazan dye to the amount of living cells in culture. Dehydrogenases from viable cells convert WST-8 into a formazan dye, which produces an easily measurable color change that can be quantified in a cell plate reader at 450 nm.
Cell viability assays often measure the amount of living, healthy cells while cytotoxicity assays typically measure the amount of dead or apoptotic cells in culture. The CCK-8 assay has been used in both types of assays though it is measuring the number of living cells. The cell counting kit-8 can be used in combination with common or novel drugs to test cytotoxicity by measuring the number of viable cells remaining after treatment...
The Fluorescence Minus One (FMO controls) are staining controls that contain all the antibodies of a panel minus 1 of them. It measures the spillover of all those other fluorophores in the channel of the missing antibody, and is used to identify and gate cells in the context of data spread due to the multiple fluorophores. It is a stronger negative control than the regular unstained control as it takes into account how the other stains in the panel impact the channel that is left out.
There are 3 situations when an FMO control is highly recommended! Click here for more FACS controls information.
With the addition of every new color, the chances of non-specific spillover increases in practically
Counting cells before the staining procedure and the analysis/live sort is not up for debate! Cell numbers affect the staining quality, the FACS instrument reading, as well as the efficacy of any downstream assay, in case the cells are live sorted. Furthermore, cells are steadily lost during the staining procedure and the recovery of most sorters is a little above 50% − it is therefore imperative that enough cells are available at the start of the experiment to compensate for the inevitable losses. There are various methods available for counting cells:
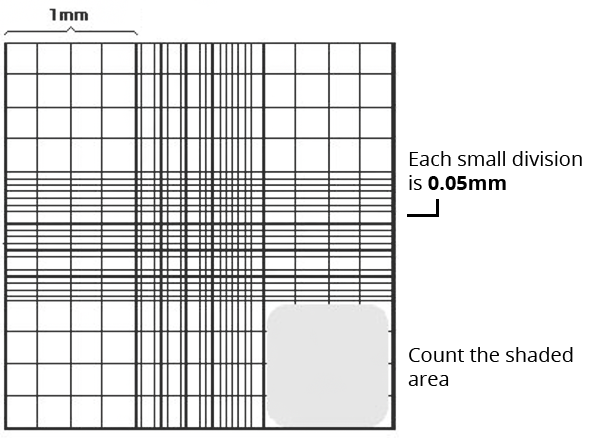
Hemocytometers are manual counting chambers and were first developed for blood cell counts. It consists of a thick glass slide onto which a gridded chamber is affixed. The four corner squares of the grid measure 1 mm x 1 mm and each square is further subdivided into 0.05 mm x 0.05 mm squares. A glass coverslip, made to specifications of the hemocytometer, can be placed 0.1 mm above the marked grid, thereby creating a space of known volume.
The cells are counted using a 4x or 10x objective lens over an area of 1 sq mm and the counting is repeated for a total of four such areas and the result is averaged. The formula for counting cells for each area is (average number of cells * 10 4 * dilution factor). To count the viable cells, use Trypan Blue exclusion method. Save some time and trouble with Boster’s ready-to-use Trypan Blue Assay Kit AR1175, see the kit below!.
Although the easiest method and least expensive, it is also labor intensive and allows for inaccuracies of the human eye. The image below depicts a typical hemocytometer counting grid.
They perform on the same principle as the hemocytometers but with the added advantages of speed and precision. Automated cell counter models have been developed by man...
Are you preparing for a FACS experiment? Here’s a 10 point checklist to help you prepare for your FACS sorting experiment:
The common problem for all fluorescence-based cell detection methods is the cells’ natural fluorescence. Cellular autofluorescence is due to the presence of various biological structures, such as collagen, elastin, NADPH, flavins, mitochondria, and lysosomes, which usually absorb in UV to blue range (355-488 nm) and emit in the blue to green range (350-550 nm). Therefore, autofluorescence interferes with analysis by reducing signal sensitivity and resolution of fluorochromes that operate in that range – FITC, GFP, and Pacific Blue to name a few.
To subtract this background and prevent false positives, it is crucial to include unstained controls and set proper PMTs and gating. However, it is even more important to minimize autofluorescence as much as possible. Below are some handy tips to follow.
Fetal calf serum (FCS) absorbs at the violet and blue spectra. Therefore, serum present in FACS buffer as well as the culture medium can increase autofluorescence. It is critical to standardize the optimum concentration of FCS in the staining buffer to strike a compromise between blocking non-specific antibody binding and minimizing autofluorescence. Most labs use FCS at 1% to 10%. From our experience, lower concentrations of FCS work as well as 10%. If low FCS concentration doe...