This website uses cookies to ensure you get the best experience on our website.
- Table of Contents
and ELISA kits, proteins related to Stomach Cancer,.
Stomach cancer, also known as gastric cancer, is a significant global health concern, ranking as one of the leading causes of cancer-related deaths worldwide. This aggressive disease originates in the lining of the stomach and can spread to nearby organs, making early detection and effective treatment crucial. Recent advancements in medical research have highlighted the pivotal role of antibodies in the fight against stomach cancer. By targeting specific proteins on cancer cells, antibody-based therapies offer promising avenues for more precise and personalized treatments. These innovative approaches not only aim to improve survival rates but also enhance the quality of life for patients by minimizing side effects. As scientists continue to unravel the complexities of stomach cancer, the development of antibody research stands at the forefront, offering hope for improved outcomes and a deeper understanding of this challenging disease.
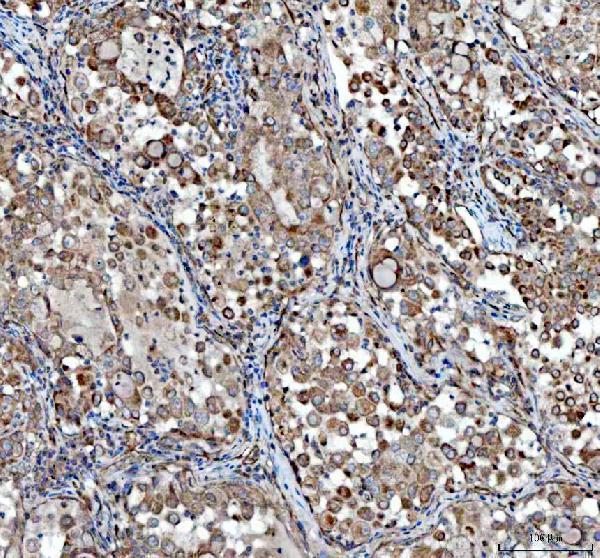
Anti-VEGF/VEGFA Antibody Picoband®, Figure 2. IHC analysis of VEGFA using anti-VEGFA antibody (PA1080).
VEGFA was detected in a paraffin-embedded section of human lung cancer tissue. Heat...
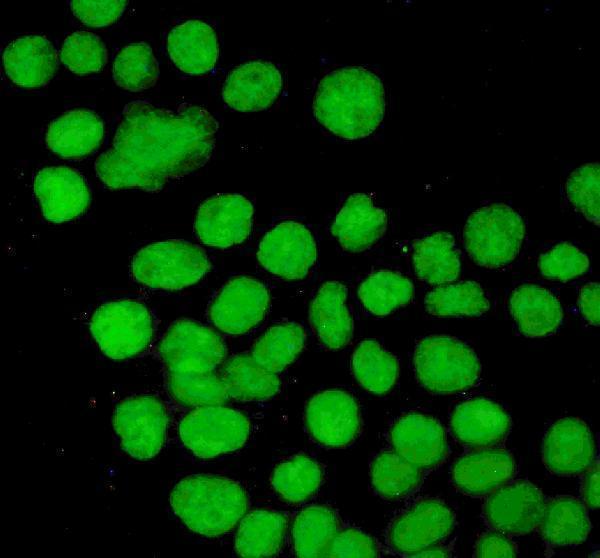
Anti-P53/TP53 Antibody Picoband®, Figure 3. IF analysis of P53 using anti-P53 antibody (PB9008).
P53 was detected in immunocytochemical section of A431 cells. Enzyme antigen...

Anti-E Cadherin 1/CDH1 Antibody Picoband®, Figure 15. IF analysis of E Cadherin using anti-E Cadherin antibody (PB9561).
E Cadherin was detected in an immunocytochemi...
| Protein Name | Gene Name | Function |
|---|---|---|
| HER2 | ERBB2 | Promotes cell growth and differentiation; overexpressed in some gastric cancers, associated with poor prognosis and targeted by therapies like trastuzumab. |
| PD-L1 | CD274 | Involved in immune checkpoint regulation; overexpression can lead to immune evasion by tumor cells, targeted by immunotherapies. |
| VEGF | VEGFA | Promotes angiogenesis; overexpression supports tumor blood supply, targeted by anti-angiogenic therapies. |
| EGFR | EGFR | Regulates cell proliferation; mutations or overexpression can drive cancer growth, targeted by EGFR inhibitors. |
| p53 | TP53 | Tumor suppressor involved in DNA repair and apoptosis; mutations lead to uncontrolled cell growth. |
| MET | MET | Involved in cell growth and survival; amplification or mutations can promote tumor progression. |
| CDH1 | CDH1 | Encodes E-cadherin, important for cell adhesion; mutations associated with diffuse gastric cancer. |
| FGFR2 | FGFR2 | Regulates cell division and growth; gene amplification linked to gastric cancer proliferation. |
| KRAS | KRAS | Oncogene involved in cell signaling pathways; mutations can lead to uncontrolled growth. |
| BRAF | BRAF | Serine/threonine kinase involved in the MAPK/ERK signaling pathway; mutations can drive cancer progression. |
| PI3KCA | PIK3CA | Catalytic subunit of PI3K; mutations activate the PI3K/AKT pathway, promoting tumor growth. |
| c-MYC | MYC | Transcription factor that regulates cell cycle, apoptosis, and metabolism; overexpression linked to cancer. |
| Cyclin D1 | CCND1 | Regulates cell cycle progression; overexpression can lead to uncontrolled cell proliferation. |
| PTEN | PTEN | Tumor suppressor that inhibits the PI3K/AKT pathway; loss of function leads to increased cell survival. |
| MUC1 | MUC1 | Mucin protein involved in cell signaling and adhesion; overexpression associated with tumor progression. |
| TGF-beta | TGFB1 | Cytokine that regulates cell growth and differentiation; can promote tumor progression and metastasis. |
| APC | APC | Tumor suppressor involved in Wnt signaling pathway; mutations can lead to increased cell proliferation. |
| ALK | ALK | Anaplastic lymphoma kinase; gene rearrangements can drive oncogenesis, targetable by ALK inhibitors. |
The study of molecular and genetic mechanisms in stomach cancer is pivotal for understanding the intricate processes that drive tumor development and progression. This sub-area focuses on identifying genetic mutations, alterations in oncogenes and tumor suppressor genes, and the dysregulation of signaling pathways that contribute to carcinogenesis. Researchers employ techniques such as next-generation sequencing and genomic profiling to uncover specific genetic changes associated with gastric cancer. Additionally, epigenetic modifications, including DNA methylation and histone modification, are examined to elucidate their roles in gene expression regulation and cancer progression. By comprehensively mapping the genetic landscape of stomach cancer, scientists can identify potential biomarkers for early detection, prognosis, and therapeutic targets. This knowledge facilitates the development of personalized medicine approaches, enabling treatments tailored to the unique genetic makeup of each tumor. Furthermore, understanding molecular mechanisms aids in uncovering resistance pathways to existing therapies, guiding the design of novel interventions to overcome treatment challenges. Ultimately, advancements in this area are crucial for improving diagnostic accuracy, enhancing treatment efficacy, and increasing survival rates for patients afflicted with stomach cancer.
Immunotherapy has emerged as a groundbreaking approach in the treatment of stomach cancer, leveraging the body's immune system to fight malignancies. This sub-area explores the complex interactions between tumor cells and the immune microenvironment within the gastric tissue. Key focuses include the identification of immune checkpoints, such as PD-1/PD-L1 and CTLA-4, which tumors exploit to evade immune detection. By targeting these checkpoints with specific inhibitors, immunotherapy aims to restore the immune system's ability to recognize and eliminate cancer cells. Additionally, research delves into the role of tumor-infiltrating lymphocytes, macrophages, and other immune cells in modulating the tumor milieu. Understanding these interactions is essential for enhancing the efficacy of immunotherapeutic strategies and minimizing adverse effects. The development of personalized immunotherapies, including cancer vaccines and adoptive cell transfer, is also a significant area of investigation. Moreover, the interplay between the tumor microenvironment and other treatment modalities, such as chemotherapy and targeted therapies, is studied to optimize combination therapies. Advances in this field hold the promise of improving clinical outcomes, reducing recurrence rates, and providing durable responses for patients with stomach cancer.